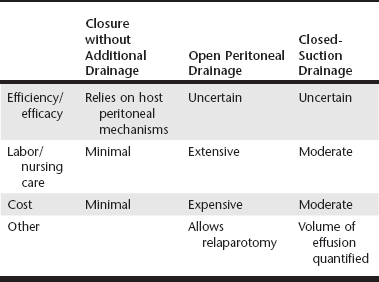
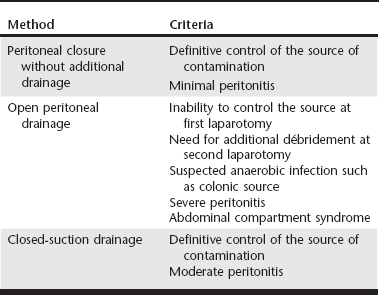
Web Chapter 2 John L Yates wrote in 1905: “There is probably no detail in modern surgical pathology that deserves more thorough comprehension, but which is less definitively understood….than the nature of the reaction of the peritoneum to drainage.” Since Yates’s classic paper, there has been substantial investigation into the indications for and best methods of peritoneal drainage. However, universal guidelines for techniques of peritoneal drainage and consistent indications for their application have not emerged. Prophylactic drainage following routine, uncontaminated intraabdominal procedures has been largely abandoned in human surgery. In veterinary medicine, postoperative abdominal drainage is now generally reserved for cases with generalized septic peritonitis. Septic peritonitis is a severe, life-threatening condition that poses many challenges for the small animal veterinarian. Obtaining an accurate and timely diagnosis; understanding peritoneal fluid and protein loss, hypovolemia, sepsis, and the systemic inflammatory response syndrome; and effective resuscitation are vital to successful treatment of peritonitis. Control of the source of peritoneal contamination remains the primary goal of exploratory laparotomy in cases of generalized septic peritonitis. More recently, the benefits of copious intraoperative peritoneal lavage and the need for postoperative peritoneal drainage have been questioned. Although a detailed discussion of peritoneal lavage is beyond the scope of this chapter, current recommendations include the judicious use of lavage to remove gross contamination and aspiration of the lavage fluid from the peritoneal cavity before closure (Platell, Papadimitriou, and Hall, 2000). This chapter focuses on the indications for and use of various drainage techniques in septic peritonitis. Postoperative drainage is indicated in septic peritonitis because a large volume of effusion has detrimental effects on peritoneal defense mechanisms and organ function. Web Table 2-1 shows a comparison of the most commonly used peritoneal drainage techniques, including closure of the peritoneal cavity without additional drainage, open peritoneal drainage, and closed-suction drainage. Patient factors to consider in selecting a method of peritoneal drainage include the success of source control and the severity of the peritonitis (Web Table 2-2). Cost and the availability of intensive nursing care may also influence the technique selected. Effective drainage of the peritoneal cavity in generalized septic peritonitis requires an understanding of intraperitoneal fluid circulation, normal intraperitoneal pressures, and the response of the peritoneal cavity to insertion of a foreign body such as a drain. Fluid injected into the peritoneal cavity disperses throughout the cavity within 15 minutes to 2 hours, depending on the site of injection (Hosgood et al, 1989). The clinical implication is that although fibrin, the omentum, the viscera, and the mesentery may try to localize a focus of contamination, it is likely that contaminated fluid can spread rapidly throughout the peritoneal cavity. Within the peritoneal cavity, the gastrointestinal tract has a luminal pressure exceeding atmospheric pressure, whereas the pressure within the peritoneal space is subatmospheric. Even with experimental insufflation of air, the intraperitoneal pressure never exceeds atmospheric pressure (Gold, 1956). Unless either the intraperitoneal pressure becomes higher than atmospheric pressure or air can enter the peritoneal cavity after surgery through a vent, drainage from the peritoneal cavity will not occur without the use of a vacuum system. Drains inserted into the peritoneal cavity are rapidly encased by the omentum and viscera (Yates, 1905). Thus in some studies it is not clear if drainage occurs from the peritoneal cavity or, more likely, from the encased area around the drain. In a previous experimental study in dogs, sump-Penrose drains were found to be encapsulated and isolated from the peritoneal cavity at necropsy after 48 hours (Hosgood et al, 1989). Despite this isolation, the drains continued to remove radiopaque contrast material from the peritoneal cavity. The use of closed-suction silicone drains for septic peritonitis has been reported (Mueller, Ludwig, and Barton, 2001). Drains in the cases described continued to accumulate fluid for up to 8 days, seeming to indicate adequate function. However, it is not clear if closed-suction drains are encased to the same extent as sump-Penrose drains and are draining a localized area or if they retain functional drainage of the peritoneal cavity despite being encased. The anticipation of significant postoperative fluid production caused by inability to control the source of contamination, generalized peritonitis, or severe local peritonitis is an indication for postoperative drainage. The efficacy of the peritoneal defense mechanisms may be limited by a large volume of fluid, either ongoing effusion or residual lavage. Phagocytosis of bacteria within fluid depends on the presence of opsonins in the fluid, which can become depleted. Therefore the presence of fluid may allow rapid bacterial proliferation. A large volume of fluid may also limit the localization of the contamination and speed the systemic absorption of bacteria and endotoxins (Platell, Papadimitriou, and Hall, 2000). A large volume of effusion also increases intraabdominal pressure. In some cases, intraabdominal hypertension is sufficient to cause cardiopulmonary dysfunction, anuria or oliguria, and intestinal ischemia (abdominal compartment syndrome) (Drellich, 2000; Conzemius et al, 1995). In some cases peritonitis may not be generalized at the time of exploratory surgery. The two most common examples of localized peritonitis in small animal surgery are found with prostatic and pancreatic abscesses. Local peritoneal drainage has been used to treat these conditions and generally involved placement of one or more Penrose drains in the abscess cavity once débridement and local lavage were completed. These techniques have largely been replaced by omentalization. The abscess cavity is located by palpation, intraoperative ultrasound, or aspiration. It is opened, drained, and gently débrided with a moistened gauze sponge. After thorough local lavage with a warm, balanced electrolyte solution, the omentum is packed loosely into the cavity, and the abdominal incision is closed. The immunologic and angiogenic properties of the omentum promote local infection control and healing. The technique has been associated with long-term success in 19 of 20 dogs with prostatic abscesses (White and Williams, 1995) and 6 of 12 dogs with pancreatic abscesses (Johnson and Mann, 2006). The decision to close the peritoneal cavity without drainage is based on control of the source of contamination and adequate decontamination of the peritoneal cavity. The clinician then relies on the body’s peritoneal drainage and immune defense systems. Fluid absorption occurs primarily by passing through gaps (stomata) in the mesothelial cells into lymphatics and thereby the circulation. More microvilli are present on the mesothelial cells of the visceral peritoneum than the parietal peritoneum to promote movement of fluid toward the diaphragm for absorption. The number of mesothelial microvilli increases and lymphatics dilate in response to peritoneal inflammation. In addition to mesothelial cells, numerous immune cells are present in the peritoneal membrane and omentum, including macrophages, lymphocytes, and mast cells. The immune cells function in bacterial phagocytosis, antigen presentation, and antibody and cytokine production in cases of peritonitis. Finally, production of plasminogen activator inhibitor 1 by peritoneal mesothelial cells promotes the organization of fibrous adhesions, which facilitate phagocytosis and localize contamination (Yao, Platell, and Hall, 2003). Lanz and colleagues (2001) reported 54% survival in a cohort of 28 cases of canine septic peritonitis managed with source control, intraoperative lavage, and closure of the peritoneal cavity without a means of additional postoperative drainage. As discussed, this method of surgical management relies on peritoneal drainage and may be appropriate when the source of contamination can be controlled definitively and peritonitis is not severe.
Drainage Techniques for the Septic Abdomen
Indications for Postoperative Drainage
Local Peritonitis
Generalized Peritonitis
Primary Abdominal Closure without Drainage
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree