Web Chapter 10 Veterinarians in both small and large animal practices are frequently called on to treat known or suspected animal toxicosis. A definitive diagnosis is not always established at the time of clinical presentation because many of these cases pose diagnostic or therapeutic challenges. Yet an early diagnosis and specific treatment are often keys to successful outcomes. In these efforts, veterinarians should avail themselves of diagnostic and treatment information from a variety of sources (see Web Chapter 9). The prevalence of toxicoses in dogs and cats is reasonably well known (see Chapters 20 and 21). Consequently, the majority of drugs needed to treat toxicoses in companion animals are known and indirectly provide information for stocking emergency drugs in practices and emergency/critical care centers. Prevalence data of animal toxicoses also may be useful to pharmaceutical companies when considering which therapies to develop and what types and amounts of drug inventories may be needed for treating animal toxicoses. This chapter includes information on the treatment of both companion animal toxicoses and food animal toxicoses because the authors anticipate that some readers work in a mixed animal practice. The use of extralabel drugs and compounding drug considerations are particularly pertinent when treating food animals. The Federal Food, Drug, and Cosmetic Act (FFDCA) (Section 201 [21 U.S.C. 321(g) (1)]) defines a drug as*: Drugs administered to animals to treat (diagnose, cure, mitigate, treat, or prevent a disease or to affect the structure or any function of the body) a toxicosis are regulated by the FDA under the FFDCA and its implementing regulations, published in Title 21 of the Code of Federal Regulations (available on-line at http://www.gpoaccess. gov/cfr/). Practicing veterinarians often place drugs used in treating an animal toxicosis into three categories—general treatment, supportive treatment, and specific treatment. This chapter does likewise. Some drugs used to treat toxicoses are not approved by the FDA under the FFDCA but are licensed by the U.S. Department of Agriculture (USDA) under the Virus-Serum-Toxin Act (VSTA). The VSTA is implemented by the Center for Veterinary Biologics of the USDA Animal and Plant Health Inspection Service. Examples of such products include antivenins and antitoxins. Animal drugs produced and distributed in full conformance with VSTA have a USDA code rather than an FDA-assigned New Animal Drug Application [NADA] or Abbreviated New Animal Drug Application [ANADA] number. Examples include botulinum antitoxin (USDA Code 6400), tetanus antitoxin (USDA Code 6302.00 & 6302.01), and Crotalidae antivenin (USDA Code 6101.00). Antivenins for animals that are licensed as biologics by the USDA are a unique form of legally marketed animal drugs.*† More information about the USDA Center for Veterinary Biologics and a listing of licensed veterinary biologics are available at http://www.aphis.usda.gov/animal_health/vet_biologics/. The limited availability of drugs approved to treat toxicoses is a long-standing problem in veterinary medicine. The drugs listed in Web Table 10-1 are approved by the FDA for use in animals for treatment of toxicoses. Use of these drugs, as indicated on labels, does not give rise to the extralabel drug use issues (discussed later in this chapter). Other than the approved uses of the seven drugs listed in Web Table 10-1, any drug used to treat animal toxicoses is likely done so in an extralabel, or an unapproved, manner. Consequently, review of extralabel drug use regulations may be of value to practicing veterinarians (see Web Table 10-1). WEB TABLE 10-1 Drugs Approved by the US FDA/CVM to Treat Animal Toxicoses *21 CFR § 522.147 = Title 21 of the Code of Federal Regulations, part 522, section 147. **NADA = New Animal Drug Application;
Treatment of Animal Toxicoses
Regulatory Points to Consider
Drugs Used to Treat Animal Toxicoses
Animal Drugs That Are Biologics
Drugs Approved to Treat Toxicoses
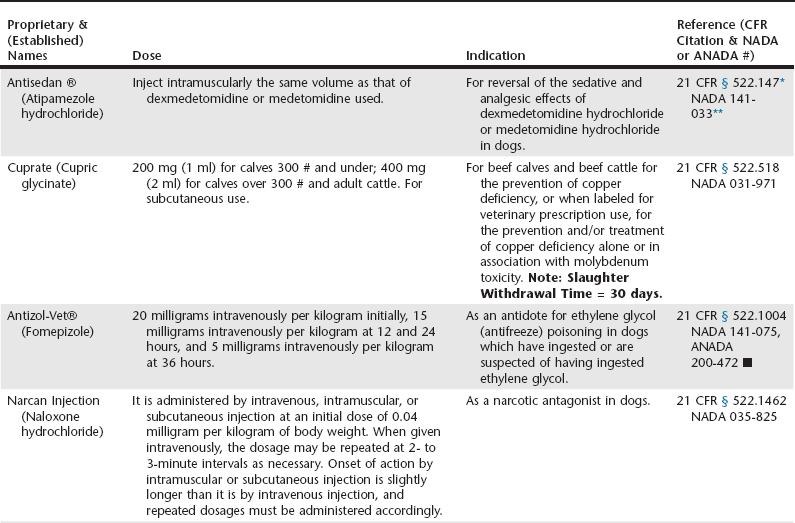

 = Abbreviated New Animal Drug Application.
= Abbreviated New Animal Drug Application.
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


