CHAPTER 10 Development of the Heart and Great Vessels The heart is a conical, muscular organ that in mammals has evolved into a four-chambered pump with four valves. During early fetal development, it is converted from an elongated muscular tube into a C-shaped structure by a process termed looping (Fig. 10-1). Subsequently, septation occurs to produce the right and left atrial and ventricular chambers and separation of the common truncus arteriosus into the aorta and pulmonary artery, respectively. The heart is interposed as a pump into the vascular system, with the right side supplying the pulmonary circulation and the left side the systemic circulation (Web Fig. 10-1; also see Chapter 2). The vascular system is subdivided into arterial, capillary, venous, and lymphatic segments. The arteries are classified into three types: elastic arteries, muscular arteries, and arterioles. The venous vessels are termed venules and veins. The lymphatic vasculature includes lymphatic capillaries and lymphatic vessels. Interposed between the arterial and venous segments are the capillary beds. A vascular segment termed the microcirculation (systemic capillary beds) includes arterioles, capillaries, and venules and is the major area of exchange between the circulating blood and the peripheral tissue (see Web Fig. 10-1; also see Chapter 2). Fig. 10-1 Heart development, emphasizing the three sources of cells. Web Fig. 10-1 Schematic diagram showing serially connected pulmonary and systemic circulatory systems and how to trace the flow of blood. The heart lies within a fibroelastic sac called the pericardium, and the wall of the heart is composed of three layers: the epicardium, the myocardium, and the endocardium (Fig. 10-2). Structurally, the heart contains in order of blood flow four major blood vessels (vena cava, pulmonary artery, pulmonary vein, and aorta), four chambers (right atrium/auricle, right ventricle, left atrium/auricle, and left ventricle), and four valves (tricuspid, pulmonic semilunar, mitral, and aortic semilunar) (Fig. 10-3). Fig. 10-2 Wall of the heart. Fig. 10-3 Normal heart, pig. The pericardium, which normally contains a small amount of clear, serous fluid (see Fig. 10-2), is composed of an outer fibrous component and an inner serous layer, which form the sac surrounding the heart. The outer component is continuous with the mediastinal pleura. The base of the fibrous pericardium surrounds and blends with the adventitia of the greater arteries and veins exiting and entering the heart. The serous pericardium forms a closed sac surrounding the heart. The heart is a muscular four chamber pump that simultaneously supplies blood to the pulmonary and systemic circulatory beds (see Web Fig. 10-1 and Fig. 2-1). Mechanical pumping is composed of sequential contraction (systole) and relaxation (diastole) that must be preceded by an electrophysiologic process that triggers a coordinated chronologic sequence of electrical events that result in muscle contractions. This electrophysiologic process is made possible by a network of special conducting fibers that are collectively referred to as the cardiac conduction system. Blood Vessels: The aorta originates from the left ventricle and provides oxygenated blood to the entire body via arteries. In a treelike manner, arteries branch and become smaller arterioles as they approach capillary beds (see Web Fig. 10-1; also see Chapter 2). These beds and postcapillary venules provide the site for exchange of oxygen, carbon dioxide, nutrients, and waste. Small venules return the exchanged fluid and blood to larger veins and eventually the postcava and precava drain into the right atrium. The poorly oxygenated blood enters the pulmonary artery from the right ventricle. Oxygen exchange occurs in the capillaries of the lung, and oxygenated blood is returned to the heart via the pulmonary veins into the left atrium. Lymphatic Vessels: Lymphatic vessels are thin walled, endothelial-lined channels that originate near the capillary beds and serve as a drainage system for returning interstitial tissue fluid and inflammatory cells to the blood. Afferent lymphatic vessels drain lymph into regional lymph nodes, which then filter and provide immunologic surveillance of the lymph, its cells, and the foreign matter it contains. The filtered lymph continues into larger efferent lymphatic vessels, which eventually drain into the caval blood via the thoracic duct. Both lymphatic vessels and veins have valves to prevent backflow of fluid. A more complete description can be found in Chapter 2. The myocardium consists of cardiac muscle cells surrounded by interstitial components that include blood and lymphatic vessels, nerves, and connective tissue cells, such as fibroblasts, histiocytes, mast cells, pericytes, primitive mesenchymal stem cells, and extracellular matrix elements of connective tissue, including collagen fibrils, elastic fibers, and acid mucopolysaccharides. Cardiac muscle cells can be divided into two populations: the contracting myocytes and the specialized fibers of the conduction system. The contracting myocyte is a cross-striated branching fiber of an irregular cylindric shape that measures 60 to 100 µm in length and 10 to 20 µm in diameter, with centrally located, elongated nuclei. Myocytes in young animals are smaller and have small amounts of sarcoplasm. Atrial myocytes are smaller than ventricular myocytes. Adjacent myocytes are joined end-to-end by specialized junctions known as intercalated disks and less frequently by side-to-side connections termed lateral junctions. Multinucleated fibers with nuclei arranged in central rows are frequently seen in hearts of growing pigs (Fig. 10-4). The myocytes of old animals commonly have large polyploid nuclei. The cytoplasm (sarcoplasm) of myocytes is largely occupied by the contractile proteins that are highly organized into sarcomeres, the repeating contractile units of the myofibril (see Figs. 15-3 and 15-8). Myofibrils are formed by end-to-end attachment of many sarcomeres. The cross-striated or banded appearance of myocytes is the result of sarcomere organization into A bands composed of myosin in the form of “thick” filaments (12 to 16 nm in diameter), I bands composed of actin in the form of “thin” filaments (5 to 8 nm in diameter), and dense Z bands at the end of each sarcomere. Thick and thin filaments interdigitate and provide the basis for the sliding mechanism of muscle contraction. Myocytes are enclosed by the sarcolemma, which consists of the plasma membrane and the covering basal lamina (external lamina). Other important components of cardiac muscle cells are generally only apparent in electron micrographs and include abundant mitochondria, a highly organized network of intracellular tubules termed the sarcoplasmic reticulum, cylindric invaginations of the plasma membrane called T tubules, ribosomes, cytoskeletal filaments, glycogen particles, lipid droplets, Golgi complexes, atrial granules (contain atrial natriuretic factor), lysosomes, and residual bodies (Web Fig. 10-2). Fig. 10-4 Normal cardiac muscle. Web Fig. 10-2 Normal cardiac muscle. The morphologic features of the cardiac muscle cells that form specialized conduction tissues, including the SAN, AVN, AV bundle (bundle of His), and bundle branches, vary greatly at different sites and among animal species but generally are thin, branching nodal muscle cells with scarce myofibrils separated by highly vascularized connective tissue (Fig. 10-5; Web Fig. 10-3). Autonomic nerve fibers are contained within the SAN. The Purkinje fibers (cardiac conduction fibers) are distinguished by their large diameters (in horse, ox) and abundant pale eosinophilic sarcoplasm rich in glycogen and poor in myofibrils. Fig. 10-5 Cardiac conduction system. Web Fig. 10-3 Heart, fibrous stroma of the cardiac conduction system, goat. Sinoatrial Node: The SAN is positioned adjacent to the epicardial adipose tissue and is often centered around a branch of the right coronary artery (Fig. 10-5, A). Several large autonomic nervous system ganglions can occasionally be seen clustering in the epicardium adjacent to the node. The SAN lacks discrete structure, and its ill-defined borders merge with the adjacent atrial wall. It structurally consists of a collection of haphazardly oriented myofibers that appear as a pseudosyncytium and are embedded within abundant loose collagenous and elastic connective tissue, with rare cores of epicardially oriented dense collagen fibers (Fig. 10-5, A1). The nodal myofibers have discrete cell borders, a moderate amount of wavy sarcoplasm with sparse myofibrils, and an elongated nucleus that contains clumps of coarse chromatin (Fig. 10-5, A2). Atrioventricular Node, Atrioventricular Bundle, and Bundle Branches: The AVN lies within the right atrial subendocardium and consists of a discrete, compact to loose mass of interconnecting myofibers that are often embedded within adipose tissue. A small nodal artery, parasympathetic ganglia, and large myelinated autonomic nerves are often present adjacent to the AVN. The nodal myofibers that have characteristic pale eosinophilic and thin sarcoplasm generally run parallel to each other but occasionally have an interweaving pattern with intervening loose collagen fibers. These myofibers contain a moderate amount of sarcoplasm with abundance of distinct striations and a short oval to elongated nucleus with dispersed chromatin. The AV bundle (Fig. 10-5, B) emerges from the cranial pole of the AVN (Fig. 10-5, B1) and pierces through the CFB, approximately at the level of the annuli of the aortic and mitral valves (Fig. 10-5, B2), to become the left and right bundle branches. The size of an AV bundle myofiber progressively enlarges and its cytomorphology transitions from a pale eosinophilic, small and thin (AVN-like morphology) myofiber to a pale eosinophilic, foamy to waxy, large, and somewhat rectangular myofiber that lacks cross striations (a Purkinje-like cellular morphology) (Fig. 10-5, C). Autonomic Nervous System: The nerve supply to the heart is autonomic and includes sympathetic, parasympathetic, and nonadrenergic noncholinergic innervation. Histologically, large nerves can be seen in the epicardium and adjacent to the coronary blood vessels, whereas special staining techniques are required for demonstration of neural tissue elsewhere. Electron microscopy and immunohistochemistry allow differentiation between sympathetic and parasympathetic nerves that are otherwise indistinguishable with H&E stain. Preganglionic parasympathetic fibers pass to the heart through the cardiac branch of the vagal nerve and synapse with parasympathetic ganglionic neurons. Postganglionic neurons are distributed to the SAN and AVN, as well as to atrial and, to a much lesser extent, ventricular myocardium (however, the ventricular conduction system is well supplied by cholinergic innervation). Postganglionic sympathetic fibers arising from the cervicothoracic and middle cervical ganglia intensely innervate the SAN and AVN and to lesser extent the AV bundle. The atrial endocardium, myocardium, and epicardium are evenly innervated, whereas the ventricles are considerably less innervated with epicardium more densely populated by neural tissue than the endocardium. Information on this topic including Web Appendix 10-1 and Web Figs. 10-4 and 10-5 is available at evolve.elsevier.com/Zachary/McGavin/. Web Fig. 10-4 Postmortem “chicken fat” arterial cast, dog. Web Fig. 10-5 Focal hemorrhage and discoloration, intracardiac injection, euthanasia solution, left ventricle, dog. Information on this topic including Web Appendix 10-2 and Web Fig. 10-6 is available at evolve.elsevier.com/Zachary/McGavin/. Web Fig. 10-6 Schematic diagram of the gross and microscopic examination of the heart. Cardiovascular dysfunction is the result of one or more of six basic pathophysiologic mechanisms (Box 10-1). Compensatory mechanisms operate in both normal and diseased hearts in an attempt to meet both the short- and long-term demands for adequate cardiac output. These mechanisms include cardiac dilation, myocardial hypertrophy, increase in heart rate, increase in peripheral resistance, increase in blood volume, and redistribution of blood flow. These compensatory mechanisms can maintain cardiac output that is adequate for some time, even in animals with severe cardiac disease sufficient to compromise cardiac function from loss of myocardial contractility, sustained pressure overload, or sustained volume overload. Cardiac dilation can occur as a terminal lesion in many cardiac diseases (Fig. 10-6, A). As a compensatory response to achieve increased cardiac output, dilation allows stretching of cardiac muscle cells to increase contractile force according to the Frank-Starling phenomenon and increased stroke volume is the result. However, stretching beyond certain limits decreases contractile strength. Myocardial hypertrophy is an important long-term compensatory response of the heart to maintain adequate cardiac output in the face of increased pressure or volume overload (see the discussion on the myocardium in the section on Responses to Injury) (Fig. 10-6, B). Fig. 10-6 Cardiac dilation and hypertrophy, heart, transected ventricles, dog. WEB TABLE 10-1 Experimental Models of Heart Failure Modified from Smith HJ, Nuttall A: Cardiovasc Res 19:181-186, 1985. A wide variety of experimental animal models of heart failure exist (see Web Table 10-1). The models have been used to develop an understanding of human cardiac diseases. Congestive Heart Failure: Congestive heart failure usually develops slowly from gradual loss of cardiac pumping efficiency coupled with either pressure or volume overload or myocardial damage (Figs. 10-7 and 10-8; also see Figs. 8-29, 8-30, and 9-39). Pathophysiologically, congestive heart failure is initiated by development of cardiac disease (e.g., myocardial, valvular, congenital) or increased workload associated with pulmonary, renal, or vascular disease leading to loss of cardiac reserve and development of decreased blood flow to peripheral tissue (forward failure) and accumulation of blood behind the failing chamber (backward failure). Reduced renal blood flow creates hypoxia in the kidneys and increases renin release from the juxtaglomerular apparatus, resulting in stimulation of aldosterone release from the zona glomerulosa of the adrenal cortex. Sodium and water retention are the results of the action of aldosterone on the renal tubules; increased plasma volume follows, as does accumulation of edema fluid (mainly in body cavities). Hypoxia also stimulates increased erythropoiesis in bone marrow and extramedullary organs, such as the spleen, causing polycythemia and thus increased viscosity of the blood. The hypervolemia from aldosterone-induced water retention increases the workload on the already failing heart. Thus a vicious cycle of cardiac decompensation is initiated that eventually leads to death from cardiac failure unless therapeutic intervention occurs. Cardiac dilation, hypertrophy, and increased heart rate can provide some compensation for the increased workload. Fig. 10-7 Ascites, congestive heart failure, furazolidone cardiotoxicity, heart and liver, duckling. Fig. 10-8 Subcutaneous edema, high altitude disease with congestive heart failure (“brisket disease”), presternal, sternal, and caudal sternocephalic regions (brisket), cow. Acute and Chronic Left-Sided Heart Failure: Acute left-sided heart failure is manifested by pulmonary congestion and edema, whereas chronic left-sided heart failure is manifested by chronic passive pulmonary congestion, chronic pulmonary edema, hemosiderosis (“heart failure cells”; see Fig. 9-39), and fibrosis. The most common causes are (1) myocardial contractility loss associated with myocarditis, myocardial necrosis, or cardiomyopathy; (2) dysfunction of the mitral or aortic valves; and (3) several congenital heart diseases. Acute and Chronic Right-Sided Heart Failure: Acute right-sided heart failure results in acute passive congestion (see Fig. 2-35) leading to hepatomegaly and splenomegaly (see Figs. 2-35 and 13-50, A), whereas chronic right-sided heart failure (see Fig. 2-36) results in hepatic congestion (nutmeg liver) (see Fig. 8-30) and more severe sodium and water retention than in left-sided heart failure. Edema is evident predominantly as ventral subcutaneous edema in horses and ruminants (see Fig. 10-8), ascites in dogs, and hydrothorax in cats. Causes of right-sided failure include (1) pulmonary hypertension, (2) cardiomyopathy, and (3) disease of the tricuspid and pulmonary valves. (See Chapters 8 and 9 for further details on hepatic and pulmonary lesions, respectively, associated with congestive heart failure.) Cells of the cardiac conduction system are modified cardiac myofibers that are able to spontaneously depolarize, which is also called autoexcitation, and function to (1) coordinate the sequence of events required for efficient ventricular filling during diastole and ejection during systole and (2) maintain the pressure in the pulmonary and systemic circuits (see Web Fig. 10-1). Depolarization of the membrane of these pacemaker cells is due to a transiently increased rapid permeability to sodium ions and a slightly longer lasting increase in permeability to calcium ions that results in their influx into the myofiber sarcoplasm, thus changing the membrane potential (see Chapter 14). As this transient permeability is lost, membranal potassium channels open, and rapid outward potassium flux results in myofiber membranal hyperpolarization (along with sodium and calcium efflux), ultimately bringing the membrane potential back to a “normal” steady state (resting potential). During this period of time (refractory period), the myocyte cannot depolarize again because of a special conformation of membrane sodium channels that is transiently lost with depolarization and regained only with hyperpolarization. Intrinsically, the resting membrane potential of pacemaker cells is more positive than that of contracting cardiomyocytes and slightly more negative than the membrane threshold potential. This difference is due to leakiness of the pacemaker cell membrane to sodium and calcium ions and a steady low-grade influx of these ions. The endocardium lines the myocardium and contains Purkinje nerve fibers, which transmit a rhythmic action potential throughout the myocardium leading to contraction. The endocardium is lined by endothelial cells, which modulate many aspects of normal hemostasis. In normal states, the endothelial cells are antithrombotic preventing circulating cells from attaching and thus allowing normal flow of blood through the heart and blood vessels. The endocardium is continuous with the endothelium of blood and lymphatic vessels. Normal flow of blood through the heart depends on functional valves (see Chapter 2). Properly functioning valves serve as one-way valves, allowing blood to flow either from one chamber to another (AVVs) or exiting from the heart to either the pulmonary circulation (pulmonic valve) or the systemic circulation (aortic valve).
Cardiovascular System and Lymphatic Vessels
Structure
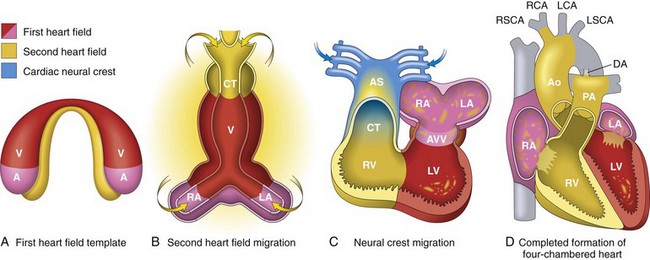
A, Day 15. First heart field (FHF) cells (shown in red) form a crescent shape in the anterior embryo with second heart field (SHF) cells (shown in yellow) near the FHF. B, Day 21. SHF cells lie dorsal to the straight heart tube and begin to migrate (arrows) into the anterior and posterior ends of the tube to form the right ventricle (RV), conotruncus (CT), and part of the atria (A). C, Day 28. Following rightward looping of the heart tube, cardiac neural crest cells (shown in blue) also migrate (arrow) into the outflow tract from the neural folds to septate the outflow tract and pattern the bilaterally symmetric aortic arch arteries (III, IV, and VI). D, Day 50. Septation of the ventricles, atria, and atrioventricular valves (AVV) results in the appropriately configured four-chambered heart. Ao, Aorta; AS, aortic sac; DA, ductus arteriosus; LA, left atrium; LCA, left carotid artery; LSCA, left subclavian artery; LV, left ventricle; PA, pulmonary artery; RA, right atrium; RCA, right carotid artery; RSCA, right subclavian artery; V, ventricle. (Modified from Srivastava D: Making or breaking the heart: from lineage determination to morphogenesis, Cell 126:1037, 2006. In Kumar V, Abbas A, Fausto N et al: Robbins & Cotran pathologic basis of disease, ed. 8, Philadelphia, 2009, Saunders.)
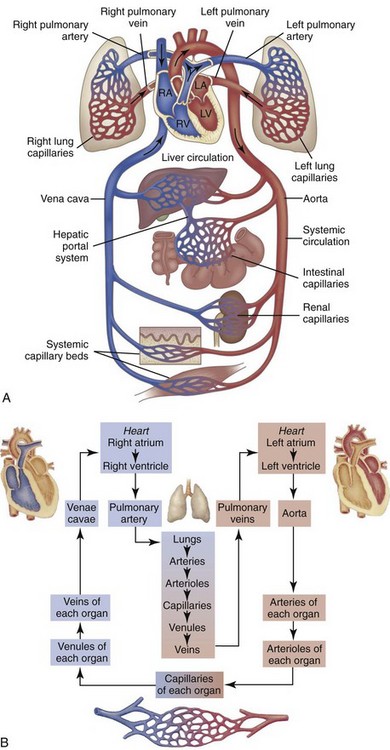
A, Right heart chambers propel unoxygenated blood through the pulmonary circulation, and the left heart propels oxygenated blood through the systemic circulation. B, The direction of blood flow begins at the left ventricle of the heart, flows to the arteries, arterioles, capillaries of each body organ, venules, veins, right atrium, right ventricle, pulmonary artery, lung capillaries, pulmonary veins, left atrium, and then goes back to the left ventricle. RA, Right atrium; RV, right ventricle; LA, left atrium, LV, left ventricle. (From McCance, K: Pathophysiology: The biologic basis for disease in adults and children, ed 6, St Louis, 2009, Mosby.)
Macroscopic Structure
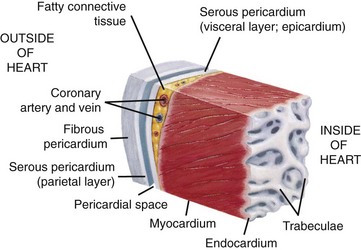
This section of the heart wall shows the fibrous pericardium, the parietal and visceral layers of the serous pericardium (with the pericardial space between them), the myocardium, and the endocardium. Note the fatty connective tissue between the visceral layer of the serous pericardium (epicardium) and the myocardium. Note also that the endocardium covers beamlike projections of myocardial muscle tissue called trabeculae. (From Huether SE, McCance KL: Understanding pathophysiology, ed 4, St Louis, 2007, Mosby.)
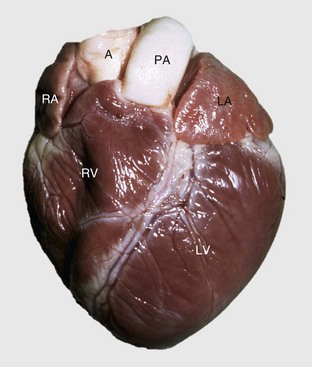
A, Aorta; LA, left atrium; LV, left ventricle; PA, pulmonary artery; RA, right atrium; RV, right ventricle. (Courtesy School of Veterinary Medicine, Purdue University.)
Pericardium and Epicardium
Cardiac Conduction System
Blood and Lymphatic Vascular Systems
Microscopic Structure
Myocardium
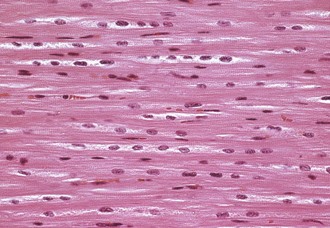
Left ventricular myocardium, longitudinal section, normal young pig. The multiple nuclei in a myocyte are readily seen and evaluated in a longitudinal section. H&E stain. (Courtesy School of Veterinary Medicine, Purdue University.)
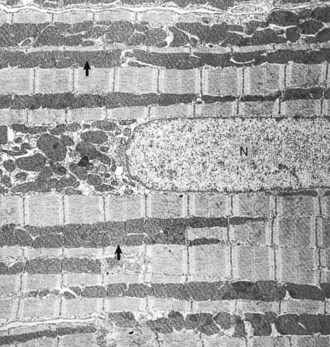
Heart, left ventricular myocytes, longitudinal section, normal rat. Numerous dense mitochondria (arrows) lie between myofibrils, which have prominent bands. N, Nucleus. TEM. Uranyl acetate and lead citrate stain. (Courtesy School of Veterinary Medicine, Purdue University.)
Cardiac Conduction System
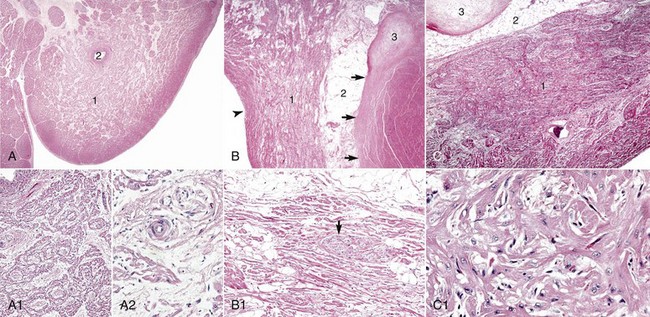
A, Sinoatrial (SA) node, foal. The center of the SA node (1) contains a nodal artery (2). H&E stain. A1, Higher magnification. Haphazardly oriented myofibers are embedded within abundant loose collagenous and elastic connective tissue. H&E stain. A2, Higher magnification. Nodal myofibers have discrete cell borders, a moderate amount of wavy sarcoplasm and an elongated nucleus. H&E stain. B, Atrioventricular (AV) node, goat. The AV node (1) is composed of interconnecting nodal myofibers that are supported by loose collagenous and elastic fibrous stroma. The node is embedded in adipose tissue (2). Note that in this illustration the AV node (1) is a poorly demarcated region (see B1 for greater detail) that is elongated (flattened) from top to bottom and that it and its surrounding adipose tissue are positioned adjacent to the cardiac fibrous skeleton (arrows) that has undergone focal chondroid metaplasia (3). The position and overall shape of the AV node in a histologic section is dependent upon the plane of section. Endocardium (arrowhead). H&E stain. B1, AV node, goat. In this higher magnification of B, AV nodal myofibers have a characteristic pale eosinophilic and thin sarcoplasm, with abundance of distinct striations and a short oval to elongated nucleus with dispersed chromatin. An autonomic myelinated nerve is present in the AV node (arrow). H&E stain. C, AV bundle, goat. The AV bundle (1) travels diagonally through the center of the figure from the lower left to the upper right margins. It is formed by an interweaving pseudosyncytium of cardiac myofibers supported by a loose to dense intervening collagenous stroma (see C1 for greater detail) and may be surrounded by adipose tissue (2). Cardiac cartilaginous skeleton (3). H&E stain. C1, Higher magnification. The AV bundle myofibers of the pseudosyncytium have moderate to large, pale eosinophilic sarcoplasm with prominent striations and large nuclei with fine stippled chromatin. H&E stain. (Courtesy Drs. A. Gal and J.F. Zachary, College of Veterinary Medicine, University of Illinois.)
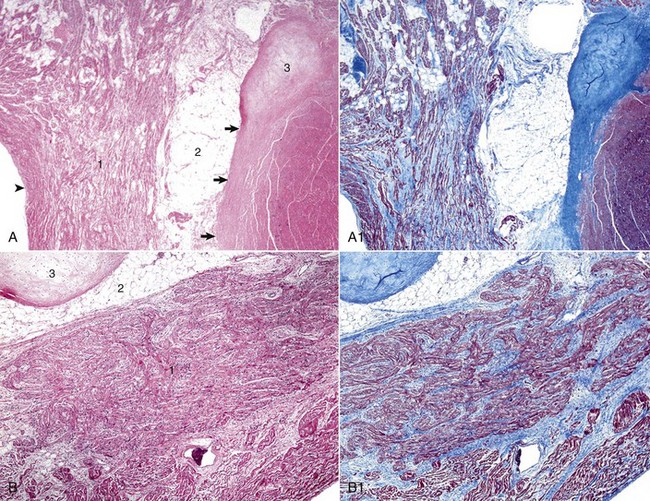
A, Atrioventricular (AV) node. The AV node (1) is composed of interconnecting nodal myofibers that are supported by loose collagenous and elastic fibrous stroma. The node is embedded in adipose tissue (2) that is adjacent to the cardiac fibrous skeleton (arrows) that has undergone focal chondroid metaplasia (3). Endocardium (arrowhead). H&E stain. A1, AV node, goat. Serial section of A. Note the overall deposition of collagen fibers (blue) in the structures labeled 1 and 3 in Web Fig. 10-3, A. Masson’s trichrome stain. B, AV bundle. In this illustration, the AV bundle (1) travels diagonally through the center of the image from the lower left to the upper right margins. It is supported by a loose to dense intervening interstitial collagenous stroma and is surrounded by adipose tissue (2). Cardiac cartilaginous skeleton (3). H&E stain. B1, AV bundle, goat. Serial section of B. Note the overall deposition of collagen fibers (blue) in the structures labeled in Web Fig. 3, B. Masson’s trichrome stain. (Courtesy Drs. A. Gal and J.F. Zachary, College of Veterinary Medicine, University of Illinois.)
Necropsy Assessment of Heart and Vascular Structures
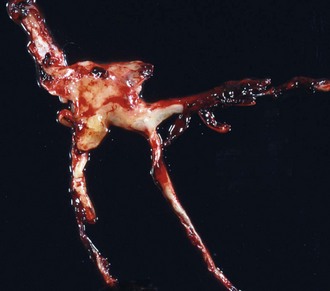
Note how the clot conforms to the shape of the lumens of the vessels from which it was removed. Chicken fat clots consist primarily of clotted plasma and fibrin and other proteins of the coagulation cascade. They are often indicative of anemia; however, in all animals their formation by the separation of the red blood cells from the rest of the components of blood depends on the erythrocyte sedimentation rate (ESR). Separation can occur in all animals in response to systemic inflammation, which increases the ESR, but the horse normally has a high ESR because equine erythrocytes clump together in rouleau formation, which increases the ESR. Thus depending on the ESR, postmortem clots may be pale white to yellow (“chicken fat” clot) or shiny red (“currant jelly” clot) or sometimes a mixture. (Courtesy Dr. R.K. Myers, College of Veterinary Medicine, Iowa State University.)
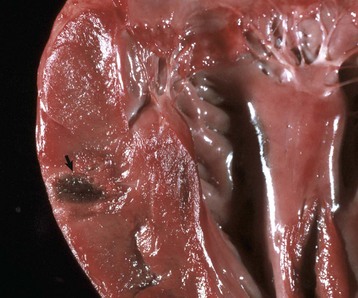
Euthanasia solution is often injected into the left ventricle. In this case, solution was injected into the myocardium and caused a localized area of hemorrhage and discoloration. A mixture of euthanasia solution and blood often form a brownish sludge in the ventricle. (Courtesy College of Veterinary Medicine, University of Illinois.)
Examination of the Cardiovascular System and Lymphatic Vessels at Necropsy and Tissue Sampling for Histopathologic Evaluation
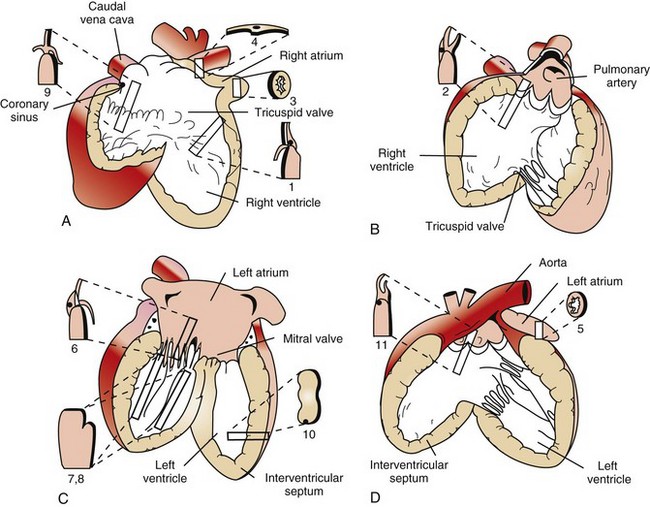
Diagrams A to D illustrate the heart opened. The numbers indicate the area and the shape of the blocks of tissue removed for histopathology. A, Right ventricle and right atrium. B, Right ventricular cavity and pulmonary outflow tract. C, Left ventricle and left atrium. D, Left ventricle and aortic outflow tract. 1, Right ventricular free wall, atrioventricular valve, and atrium. 2, Pulmonic valve, right ventricular outflow tract, and pulmonary artery. 3, Right auricular appendage. 4, Sinoatrial node. 5, Left auricular appendage. 6, Left atrioventricular valve, ventricle, and atrium. 7 and 8, Left ventricular free wall and papillary muscles. 9, Atrioventricular node, right atrioventricular valve, and atrium. 10, Interventricular septum. 11, Aortic valve, left aortic outflow tract, and aorta. (From Bishop SP: Necropsy techniques for the heart and great vessels. In Fox P, Sisson D, Moise N, eds: Textbook of canine and feline cardiology, ed 2, Philadelphia, 1999, Saunders.)
Function
Myocardium
Cardiac Pathophysiology: Myocardial Dysfunction
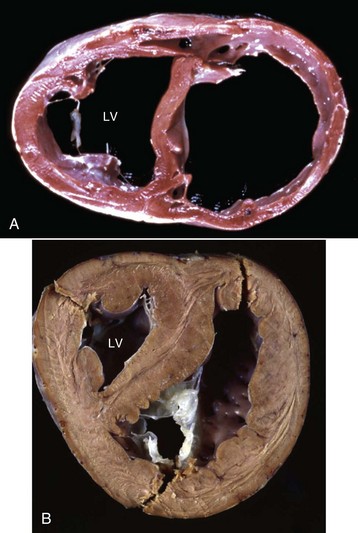
A, Cardiac dilation. Note the thin walls of both dilated ventricles. LV, Left ventricle. B, Cardiac hypertrophy (fixed tissue). Note that the right ventricular and left ventricular (LV) walls are approximately the same thickness, indicating that there is right ventricular hypertrophy. (A courtesy Dr. Y. Niyo, College of Veterinary Medicine, Iowa State University; and Noah’s Arkive, College of Veterinary Medicine, The University of Georgia. B courtesy College of Veterinary Medicine, University of Florida; and Noah’s Arkive, College of Veterinary Medicine, The University of Georgia.)
Syndromes of Cardiac Failure or Decompensation
Types of Heart Failure
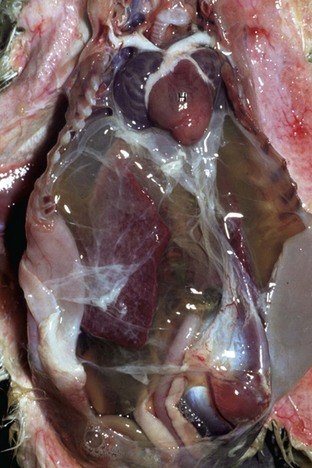
Note prominent accumulations of serous fluid in the abdomen and fibrin deposits over the surface of the liver. The heart (H) is dilated. (Courtesy School of Veterinary Medicine, Purdue University.)
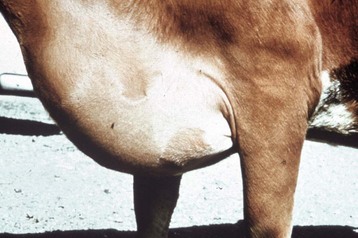
The extensive subcutaneous edema is the result of chronic congestive heart failure. (Courtesy School of Veterinary Medicine, Purdue University.)
Cardiac Conduction System
Endocardium and Heart Valves
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Veterian Key
Fastest Veterinary Medicine Insight Engine
