Chapter 4 Cardiac disease and pathology
CARDIAC PATHOLOGICAL EXAMINATION
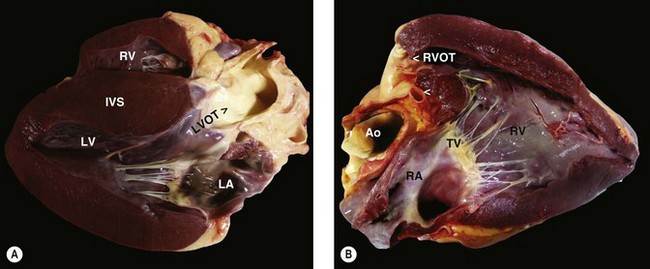
Most pathological techniques to examine the heart are designed to examine each chamber in isolation, usually opening the chamber along its border with the intraventricular septum and displaying its internal surface for examination and determination of weight.1 While this technique does have several advantages it does not enable the clinician to visualize the heart in the manner to which it has been examined by echocardiography. Therefore, a more clinically orientated approach to gross pathological examination of the heart is recommended.2 This approach is lesion orientated and therefore should commence by examining the side of the heart which is most affected by disease.
The pericardium should be examined for evidence of pathology, and the pericardial fluid volume should be determined and a sample obtained for cytological and/or bacteriological examination. The left side of the heart is examined by placing metal rods (blunt ended and at least 30 cm in length) through a small hole made in the left atrium and advanced through the mitral valve and rested at the cardiac apex; it is advisable not to place the rod through one of the pulmonary veins as this will not create the correct anatomical view. A second metal rod should be placed through the aortic valve and placed at an angle so that it reaches the same point on the cardiac apex. A large flat knife, such as a 12-inch disposable dissection knife, should then be used to cut alongside these metal guides to create a sagittal section and open the ventricle to mimic a left parasternal view of the left ventricle and left ventricular outflow tract (Fig. 4.1). The right ventricle should be examined in a similar manner by placing a metal rod down through the pulmonary artery and another through the right atrium into the right ventricular apex through the tricuspid valve. Care should be taken to avoid the moderator band within the ventricular lumen (trabeculae septomarginalis). This will mimic the cranially angled right parasternal view of the right ventricle and right ventricular outflow tract.
PRIMARY MYOCARDIAL PATHOLOGIES( VMD)
VMD)
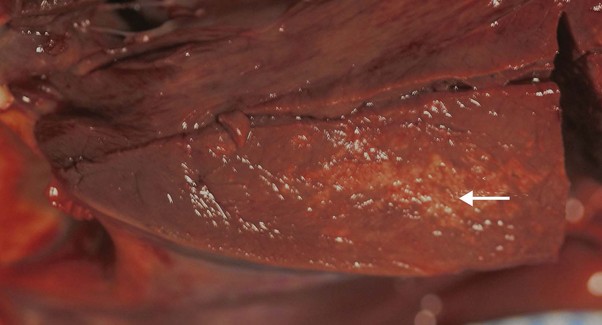
Primary myocardial disease occurs rarely in the horse; dilated cardiomyopathies are occasionally reported and are described in Chapter 19. Myocardial fibrosis has been associated with ventricular tachycardia, presumably due to disruption of the cardiac conduction system of the ventricle, but can also be detected as an incidental finding.3 The gross appearance of these changes are of discolouration within the myocardium, but are rarely specific to a particular disease process (Fig. 4.2).
In most cases a local ischemic event was assumed to be the primary disease process whereas a primary inflammatory aetiology has also been implicated.4 However, as fibrosis is an end-stage process the primary disease process can be difficult to confirm. In a group of horses microemboli from parasitic lesions were suspected based on statistical associations.3 In human patients and animal models, fibrosis is associated with myocardial remodelling and this process is described below in more detail. Myocarditis is often a clinical diagnosis based on the presence of ventricular dysrhythmias with no apparent underlying cause, although viral and bacterial causes, including Streptococcus equi var equi, have been implicated;1 however, in many cases the primary initiating cause is unknown. (![]() ET)
ET)
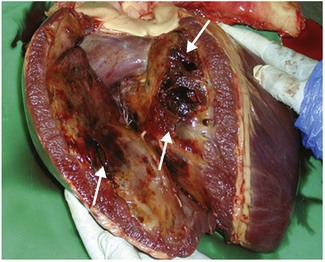
Other primary myocardial pathologies are rare, although do occur sporadically (Fig. 4.3) and usually present clinically with cardiac dysrhythmias or signs of low cardiac output (see Chapter 19). (![]() CN)
CN)
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree