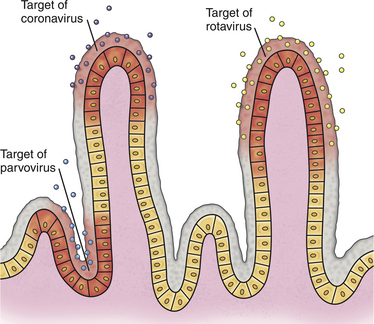
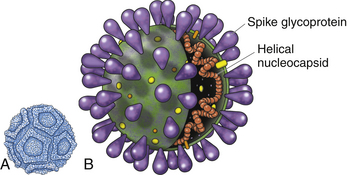
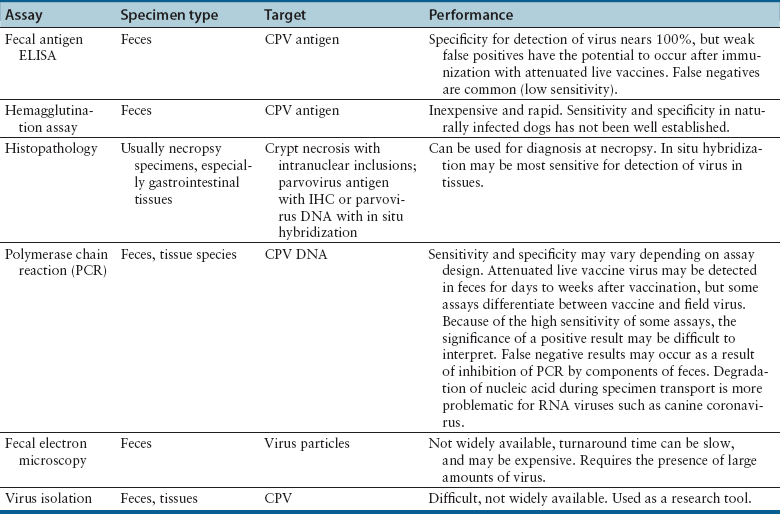
Chapter 14 Canine parvovirus is the most widely recognized cause of transmissible viral diarrhea in dogs and one of the most common infectious diseases of dogs worldwide. It is caused by variants of canine parvovirus-2 (CPV-2), which are members of the genus Parvovirus. CPV-2 emerged in the early to mid-1970s and caused a worldwide pandemic of illness in dogs.2 The spread of the virus worldwide occurred over a remarkable period of about 6 months. CPV-2 may have been derived from feline panleukopenia virus (FPV) or a closely related virus of wild carnivores. It has since mutated to CPV-2a in 1979, CPV-2b in 1984, and, most recently, CPV-2c, which was first detected in Italy in 2000 and has subsequently been found worldwide, with the exception of Australia. Separate lineages have been identified in different geographic locations worldwide.3 The virus uses the transferrin receptor to enter host cells. With the mutation from CPV-2 to CPV-2a, the virus developed the ability to replicate readily in feline cells, and CPV-2a, CPV-2b, and CPV-2c are now responsible for some cases of feline viral enteritis.4,5 Feline parvoviral enteritis is further discussed in Chapter 19. CPV-2 variants must be differentiated from CPV-1, also known as canine minute virus, which belongs to the genus Bocavirus. In general, CPV-1 is thought to have minimal pathogenic potential, but it has been associated with abortion in pregnant dogs; respiratory, cardiac, and gastrointestinal signs in neonatal dogs; severe gastroenteritis in adult dogs; and possibly neurologic signs in dogs.6–8 Parvoviruses are small, non-enveloped single-stranded DNA viruses (Figure 14-1, A) that can survive for long periods (over 1 year) in the environment. As a result, contact with virus that persists in the environment is an important means of transmission. Insects and rodents may also serve as mechanical vectors for the virus. Canine parvovirus requires the presence of mitotically active cells in order to replicate. Young animals (6 weeks to 6 months, and especially those less than 12 weeks of age) are more likely to develop severe illness; however, disease can also occur in unvaccinated or improperly vaccinated adult dogs. In North America, Rottweilers, American pit bull terriers, Doberman pinschers, English springer spaniels, and German shepherd dogs appear to be at increased risk for development of parvoviral enteritis,9,10 but this has not been the case in other geographic locations. A seasonal distribution of disease has been reported in some geographic locations, which may reflect times when dogs access the outdoors and contact virus in the environment. For example, in Saskatoon, Canada, dogs were three times more likely to be admitted with parvoviral enteritis in July, August, and September, compared with the rest of the year.9 In other locations, the seasonal pattern of disease has differed or been nonexistent.11,12 For dogs older than 6 months of age, intact males were twice as likely as females to develop parvoviral enteritis.9 A surveillance study from Australia found a correlation between clusters of parvoviral enteritis and regions of relative socioeconomic disadvantage.12 FIGURE 14-1 A, General structure of canine parvovirus. The virus is non-enveloped, is 25 nm in diameter, and has icosahedral symmetry. B, Structure of a coronavirus, which is an enveloped virus. Canine parvovirus is about one-quarter the size of a coronavirus. Other viral pathogens that have been associated with enteritis and diarrhea in dogs are canine distemper virus (CDV) (see Chapter 15), canine enteric coronavirus (Figure 14-1, B), rotaviruses, astroviruses, adenoviruses, caliciviruses, and novel viruses that include a norovirus, kobuvirus, sapovirus, and possibly also a circovirus.13–17 Canine enteric coronavirus primarily causes mild diarrhea in puppies that are less than 6 weeks of age and may be found in co-infections with other viral causes of gastroenteritis, including CPV-2 variants. Rarely, it has been identified as a more significant cause of diarrhea in young dogs.18 This chapter focuses on canine parvoviral enteritis, which is the most widely recognized and pathogenic viral enteritis of dogs. Transmission of parvovirus and other viral causes of gastroenteritis occurs by the fecal-oral route, after exposure to virus in feces or vomit, or importantly, virus that persists on fomites. Virus is shed for a few days before the onset of clinical signs, and shedding declines considerably after 7 days.19 The severity of clinical signs depends on factors such as virus strain and host immunity, which is affected by stressors such as weaning and overcrowding, maternal antibody, and the presence of concurrent infections such as other enteric viral and parasitic infections. Subclinical infections are probably widespread. The incubation period for canine parvoviral enteritis is 7 to 14 days in the field, but shorter incubation periods (as short as 4 days) have been observed with experimental infections. The virus replicates in oropharyngeal lymphoid tissues, after which viremia occurs. Damage occurs to rapidly dividing cells in the gastrointestinal tract, thymus, lymph nodes, and bone marrow. Affected gastrointestinal tissues include the epithelium of the tongue, oral cavity, esophagus, and intestinal tract, and especially the germinal epithelial cells of the intestinal crypts. Neutropenia results not only from infection of the marrow but also sequestration of neutrophils in damaged gastrointestinal tissue. Malabsorption and increased intestinal permeability result. Secondary bacterial infections of the gastrointestinal tract, which may be followed by bacterial translocation, bacteremia, and endotoxemia, play a key role in the pathogenesis of the disease. Mucosal candidiasis has also been described in puppies with parvoviral enteritis (see Chapter 67). The outcome is clinical signs of fever, lethargy, inappetence, vomiting, diarrhea, rapid dehydration, and abdominal pain. Diarrhea is often liquid, foul-smelling, and may contain streaks of blood or frank blood. Ascarid nematodes may be identified in the vomitus of some dogs. Dogs with canine parvoviral enteritis have evidence of disordered coagulation, with decreased antithrombin activities, prolonged activated partial thromboplastin time, increased thromboelastography maximum amplitude, and increased fibrinogen concentrations. Catheter and organ thrombosis may occur.20 Secondary bacteremia may be associated with multiple organ failure and death. Infection of the dam by CPV-2 variants early in gestation can lead to infertility, resorption, or abortion. Puppies that are infected in utero or up to 2 weeks of age may develop viral myocarditis, which results in signs of sudden death or congestive heart failure.21–23 Damage to the developing myocardium usually occurs up to the first 2 weeks of life, but clinical signs of myocardial damage may be delayed until up to 2 months of age. Cerebellar hypoplasia has been rarely reported in dogs after in utero infection24 but is more common in kittens infected with FPV (see Chapter 19). Generalized infection has been reported in neonatal puppies, with hemorrhage and necrosis within the brain, liver, lungs, kidneys, lymphoid tissues, and gastrointestinal tract.25 Because maternal antibody protects puppies during this period, the incidence of neonatal complications of parvovirus infection and myocarditis has declined dramatically since the virus first emerged, because of widespread vaccination and exposure of adult animals. Neurologic signs in puppies with parvoviral enteritis may result from hypoxia secondary to myocarditis, hypoglycemia, or intracranial thrombosis or hemorrhage. The possibility of co-infection with CDV should also be considered. The DNA of CPV-2 variants has also been detected in the central nervous system,26 and there have been rare reports of leukoencephalomalacia in association with infection.27,28 In contrast to canine CPV-2 variants, the replication of less pathogenic enteric viruses is restricted to the intestinal tract, and the crypt epithelial cells are generally spared (Figure 14-2). Canine enteric coronavirus, for example, infects the mature enterocytes at the tips of the villi. Crypt cell hyperplasia occurs to replace the damaged cells. The villi become shortened or distorted, which leads to malabsorption and diarrhea. Physical examination of puppies with parvoviral enteritis often reveals fever (up to 41°C or 105°F), lethargy, weakness, dehydration, and abdominal tenderness and a fluid-filled intestinal tract on palpation (Figure 14-3). Vomiting or diarrhea may occur during the examination, or there may be evidence of diarrhea or frank blood on the perineum or the rectal thermometer. Occasionally abdominal palpation reveals a tubular mass as a result of intestinal intussusception. Ulcerative glossitis can occur in some puppies. Mucosal pallor, prolonged capillary refill time, or rarely hypothermia may be observed in some dogs.29 Septic shock may be associated with tachycardia or bradycardia, mental obtundation, and poor pulse quality. Uncommonly, neurologic signs such as tremors and seizures are observed. Puppies with myocarditis may be tachypneic and have increased lung sounds as a result of congestive heart failure. Erythema multiforme has been reported in dogs with canine parvoviral enteritis; these dogs had generalized cutaneous and mucosal ulceration as well as swelling of the pinnae and paws.30,31 The most common abnormalities found on the CBC are leukopenia, neutropenia, and lymphopenia (Table 14-1). Toxic neutrophils and monocytopenia may also be present. Only around one third of dogs had leukopenia in one study.29 Leukopenia can develop after the onset of gastrointestinal signs, when puppies are first brought for examination. Some dogs have leukocytosis due to a neutrophilia and monocytosis. Although the presence of leukopenia supports a diagnosis of parvoviral enteritis, other severe gastrointestinal infections such as salmonellosis can also cause leukopenia and diarrhea, so it is not specific for diagnosis of the disease. Thrombocytosis or, less commonly, thrombocytopenia may also occur.29 Some puppies develop anemia as a result of gastrointestinal blood loss, which may be non regenerative or become regenerative. The serum biochemistry panel in dogs with parvoviral enteritis often shows hypoproteinemia, hypoalbuminemia, and hypoglycemia. Mild hyperglycemia has also been reported. Electrolyte abnormalities such as hyponatremia, hypochloremia, and hypokalemia may occur (Table 14-2). Occasionally severe dehydration results in prerenal azotemia. Puppies with bacterial sepsis may develop increased liver enzyme activities and hyperbilirubinemia. Coagulation abnormalities have been reported in a small number of dogs with parvoviral enteritis. When abnormalities occur, findings include prolonged activated partial thromboplastin time, decreased antithrombin activity, increased fibrinogen concentrations, and increased thromboelastography maximum amplitude.20 An increase in D-dimers or fibrin degradation products has not been reported. More research is warranted to understand the range of coagulation abnormalities that can occur in parvoviral enteritis. Diagnostic assays for canine parvoviral enteritis in dogs are listed in Table 14-3. TABLE 14-3 Diagnostic Assays Available for Canine Parvoviral Enteritis in Dogs CPV, Canine parvovirus (refers to CPV-2 variants); IHC, immunohistochemistry. The most widely used assay for diagnosis of canine parvoviral enteritis is an in-house fecal antigen ELISA, which is performed on a rectal swab specimen. Several assays are available, and although they detect all variants of CPV-2 including CPV-2c, their sensitivities and specificities vary.32,33 Sensitivity is particularly problematic, because viral shedding is transient, and antibody present may bind viral antigen so that it is unavailable for reaction with the assay. The sensitivities of three commercially available fecal antigen assays (SNAP Parvo antigen, IDEXX Laboratories GmbH; FASTest Parvo Strip, Scil Animal Care Company GmbH; Witness Parvo Card, Selectavet GmbH) were 50%, 40%, and 60%, respectively, when compared to immunoelectron microscopy, and 18%, 16%, and 26%, respectively, when compared with results of a fecal PCR assay.33 Because some dogs that lack evidence of gastrointestinal signs can be positive using PCR assay, immunoelectron microscopy may be a more appropriate gold standard for disease (although it is not widely available for routine diagnosis). In another study, the sensitivity of a fecal antigen test for detection of CPV-2a, CPV-2b, and CPV-2c, on specimens that contained high CPV DNA loads (>105 copies/mg of feces as determined with real-time PCR) was 80%, 78%, and 77%, respectively.32 When compared with PCR and immunoelectron microscopy of stool, the specificities of the three commercially available antigen assays above were 98%, 98%, and 92%, respectively,33 so false positives were uncommon. It has been suggested that false-positive antigen tests can occur 4 to 8 days after vaccination with attenuated live CPV-2 vaccines.34 In kittens, false-positive test results did occur after vaccination for FPV, but they were more likely to occur with some assays as opposed to others, were generally weak positives, and occurred even after vaccination with inactivated vaccines.35 Similar studies have not been reported for CPV. Canine parvovirus agglutinates erythrocytes, and so the presence of the virus in stool can be detected with a simple hemagglutination test that involves mixing a suspension of feces with porcine erythrocytes. Agglutination of erythrocytes in a microwell plate or on a slide indicates the presence of parvovirus in the feces.36 The sensitivity and specificity of these assays in dogs with and without natural parvoviral infections in the field require further investigation. Several commercial veterinary diagnostic laboratories now offer real-time PCR assays for detection of CPV-2 variants and other enteric viral pathogens (such as canine coronavirus). Assays may detect as few as 1000 copies of viral DNA per milligram of stool. Turnaround times are less than 24 hours in some laboratories. PCR assays are useful when fecal antigen tests are negative but parvoviral enteritis is still suspected as a diagnosis, or when canine enteric coronavirus infection is a potential cause of illness (because PCR panels that assay for parvovirus DNA also often include an assay for canine enteric coronavirus RNA). Unfortunately, although infrequent, positive PCR assay results for CPV can occur in dogs without signs of gastroenteritis or in dogs with chronic diarrhea, and so it may be difficult to ascertain whether a positive parvovirus PCR result indicates that CPV is the cause of a dog’s illness. Attenuated live vaccine virus can also be detected in the feces with PCR assays after vaccination, although assays have been designed that can differentiate between vaccine and wild-type virus.37 It is not yet clear for how long after vaccination false-positive PCR results might occur, and this could vary based on the design of the PCR assay used. Both vaccine and field virus have been detected simultaneously in some dogs using these assays. In the future, quantitation of virus loads in feces using real-time PCR may be helpful for interpretation of the significance of a positive PCR assay result.
Canine Parvovirus Infections and Other Viral Enteritides
Etiology and Epidemiology
Clinical Features
Physical Examination Findings
Diagnosis
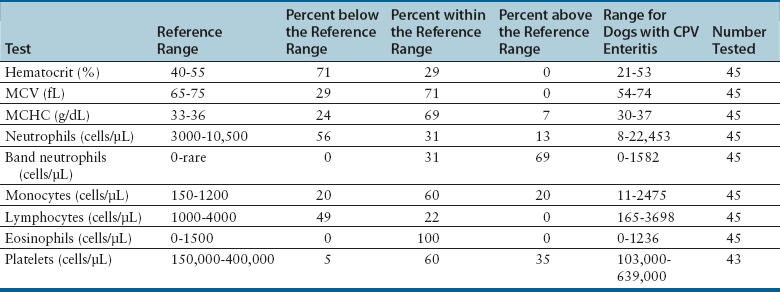
Complete Blood Count
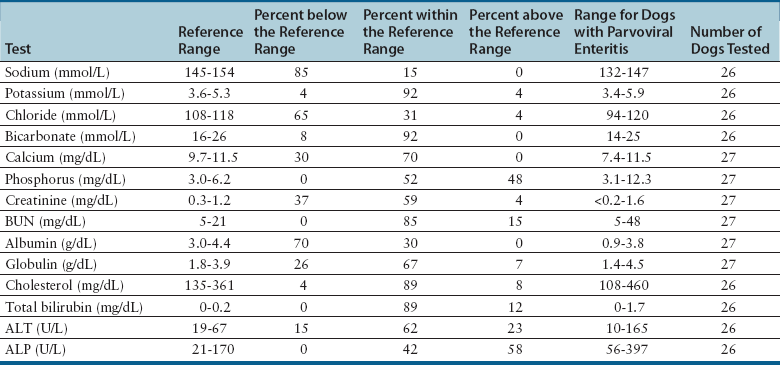
Serum Biochemical Tests
Coagulation Profile
Microbiologic Tests
Fecal Parvovirus Antigen ELISA
Hemagglutination Testing
Molecular Diagnosis Using the Polymerase Chain Reaction
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Canine Parvovirus Infections and Other Viral Enteritides
Only gold members can continue reading. Log In or Register to continue