Chapter 13 BREEDING MANAGEMENT OF THE PROBLEM MARE
MARE REPRODUCTIVE EFFICIENCY
The following are some of the parameters used to measure reproductive efficiency:
Chapter 26 has a detailed description of the goals for reproductive efficiency for which breeders and veterinarians should strive and the major factors that affect these parameters.
The clinician should be aware of how to investigate the problem breeding mare. Box 13-1 outlines a suggested approach for the examination of a problem breeding mare.
Box 13-1 Outline of a Protocol for the Clinical Examination of a Problem Breeding Mare
Many causes of subfertility can act either alone or in combination with one another. Subfertility causes can be broadly categorized into infectious or noninfectious factors, with the latter being further divided into anatomic abnormalities and functional aberrations. This format, as shown in Box 13-2, is used to discuss the various causes of subfertility and infertility in this chapter.
ANATOMIC ABNORMALITIES OF THE FEMALE REPRODUCTIVE TRACT
Defective Vulva
In the normal mare, the vulva provides the first effective barrier to protect the uterus from ascending infection. The vulvar lips are full and firm and meet evenly in the midline, and 80% or more of the vulvar opening is below the brim of the pelvis. If the vulvar seal is incompetent, aspiration of air and contamination into the vagina can occur. The initial vaginitis may lead to cervicitis and acute endometritis, resulting in subfertility. Caslick1 first pointed out the importance of this condition in relation to genital infection, which is very prevalent in breeds such as the Thoroughbreds or Saddlebreds and is almost unknown in others such as Shires and ponies. Defective vulvar conformation can be congenital, which is very rare, or acquired, which is due to (1) vulvar stretching following repeated foalings, (2) injury to perineal tissue, or (3) poor body condition (old, thin mares).
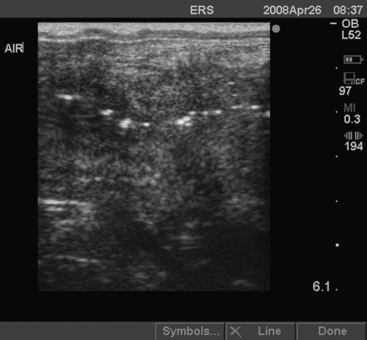
Older, pluriparous mares are more commonly affected with pneumovagina (aspiration of air into the vagina, also called wind sucking). However, pneumovagina can also develop in young mares that are in work and have little body fat or poor vulvar conformation. In some mares, pneumovagina may occur only during estrus when the perineal tissues are more relaxed. Some mares make an obvious noise while walking, but the diagnosis may be more difficult in other mares. The presence of hyperemia and a frothy exudate in the anterior vagina on examination with a speculum is pathognomonic. Rectal palpation of a ballooned vagina or uterus from which air can be expelled confirms the diagnosis. Real-time ultrasound examination of the uterus may reveal the presence of air as hyperechogenic (white) foci sometimes seen as a line at the opposed luminal surfaces (Fig. 13-1). Cytologic and histologic examination of the endometrium may demonstrate significant numbers of neutrophils indicative of an endometritis. Rarely, eosinophils are also found in association with pneumovagina.
Treatment should be directed at correcting the cause of pneumovagina and concurrently treating the resulting acute endometritis. The former can be done surgically by performing a Caslick’s procedure, although in some cases increasing the physical condition and fat status of the mare may be sufficient. When the angle of the vulvar surface relative to the vertical is the primary defect, the Caslick’s procedure is ineffective, and perineal resection should be used to achieve a satisfactory vulvar conformation.2 Many mares are subjected to a Caslick’s procedure unnecessarily; the operation should be reserved for mares with a true vulvar defect rather than be performed merely because the mare has failed to become pregnant.
Uterine Cysts
Uterine cysts are the most common type of uterine lesion identified in the mare. They can be diagnosed easily with the use of ultrasonography and it has been shown that the incidence is much greater than originally suspected based on rectal palpation alone. One detailed study found an overall prevalence of uterine cysts of 26.8%, with mares older than 11 years being 4.2 times more likely to have cysts.3
The relationship between subfertility and uterine cysts is not clear. Some authors suggest that uterine cysts can reduce pregnancy rates.4 However, a large field study concluded that there was no evidence to suggest that uterine cysts adversely affect the establishment or maintenance of pregnancy.3 Their effect on fertility could be by restricting early conceptus mobility and later in pregnancy by interfering with nutrient absorption by contact between the cyst wall and yolk sac or allantois. However, it is difficult to substantiate their primary role because they are a common sign of uterine disease in general, including senility and previous endometritis. In my experience, only mares with multiple large lymphatic cysts have a reduced pregnancy rate.
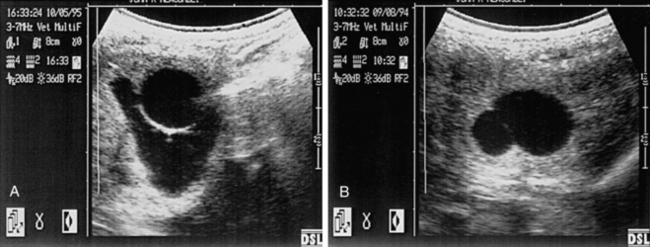
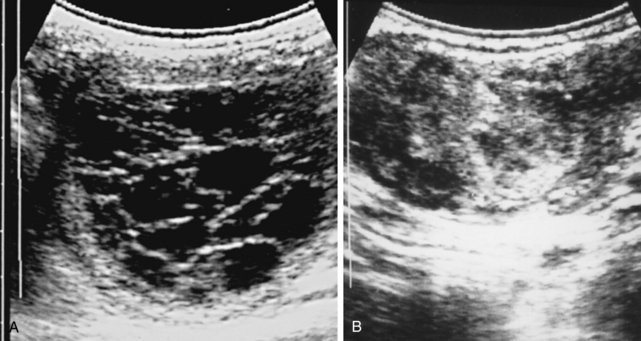
Cysts can be confused with an early conceptus and give rise to false early pregnancy diagnosis or the incorrect diagnosis of twin pregnancies during ultrasound scanning. Differentiation is based on previous cyst mapping, but also the early mobility of the conceptus, the presence of specular reflections, the spheric appearance of the conceptus, and growth rate. Although some cysts can be easily confused with an early pregnancy, there is often an opportunity to scan the mare before or immediately after breeding. In the illustrated case, it is impossible to be certain whether there are twin unilateral vesicles of approximately 20 and 16 days, a single pregnancy and a 19-mm cyst, a cyst with adjacent free fluid, or a 19-mm conceptus with adjacent free fluid. In these cases, it is vital to study the ultrasonographic image carefully. A close look at the wall between the two structures in Fig. 13-2, A, shows a relatively thick and hyperechogenic wall compared with the twin vesicle shown in Fig. 13-2, B. This would confirm that at least one structure is a cyst. Reassessment of the irregular structure confirms that the fluid is contained and does not extend up or down the horn, as would be found with free fluid. The appearance of an embryo proper at around 22 days and a heartbeat at around 25 days of pregnancy provides a definitive diagnosis. Thorough identification of cysts at the beginning of the breeding season minimizes the chance of false pregnancy diagnosis.
Partial Dilation of the Uterus
The discrete collection of fluid in permanent ventral dilations at the base of one or rarely both horns of the uterus, which can be palpated per rectum, was first reported by Knudsen.5 Ventral uterine enlargements have subsequently been discussed by Kenney and Ganjam,6 who suggested that they originate by one of four mechanisms: mucosal atrophy, myometrial atony, lymphatic lacunae, or endometrial cysts. Their precise relationship to subfertility is not clear, but mares that fail to eliminate the fluid and debris that accumulate in these sacculations after mating are susceptible to the establishment of chronic endometritis; treatment for mares with defective uterine clearance is discussed later in this chapter.
Uterine Adhesions
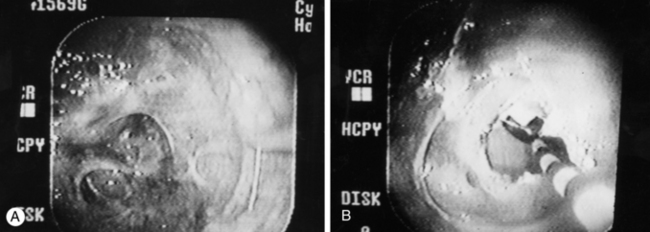
Uterine adhesions are most frequently suspected when mares have had a long-standing uterine infection and cervical tags are detected when performing uterine procedures such as artificial insemination or uterine cultures. The extent of the adhesions in the uterus is diagnosed by endoscopic examination of the uterus.7 Multiple adhesions adversely affect fertility by causing fluid accumulation or by affecting the mobility of the conceptus. Severe adhesions can completely obstruct one or both of the uterine horns. It is possible to remove the obstruction endoscopically by either cauterization or laser techniques, starting at the thin membranous parts of the obstruction (Fig. 13-3, A). It is important not to “burn” too deeply into the uterine wall because more severe damage to the uterine wall might occur. After removing the obstruction, the uterus should be flushed to remove any debris and the mare given PGF2α. In addition to an assessment of an endometrial biopsy, the prognosis for future breeding also depends on the severity of the obstruction and to what extent the obstruction could be removed. Severe adhesions can be too thick to safely treat with cautery or laser techniques (Fig. 13-3, B), and the mare needs to be retired from breeding or placed in an in vitro fertilization (IVF) program.
Uterine Foreign Bodies
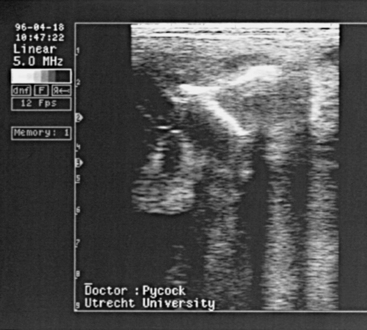
Uterine foreign bodies, which may act as a nidus for the establishment of chronic endometritis or pyometra, have been documented but are uncommon.8 In one case, seen by the author, the mare had a history of failure to conceive and short interestrus intervals following a dystocia. Ultrasound examination revealed hyperechogenic fetal bone (part of the scapula) (Fig. 13-4), which was removed manually via the cervix. The mare conceived after appropriate intrauterine lavage and antibiotic treatment to correct the endometritis. Other foreign bodies that have been reported include straws following insemination with frozen semen and the tips of uterine swabs.
Oviductal Abnormalities
Oviductal abnormalities are rarely reported in the mare, and although evidence of salpingitis can be detected in problem-breeding mares, the incidence is not different than that noted in normal, fertile mares.9 However, a recent a study by Fiala et al.10 suggested that salpingitis is more frequent in aged mares than previously thought. They studied oviducts from Brazilian mares collected at an abattoir and determined that bilateral salpingitis was present in 43.7% whereas unilateral salpingitis was present in 28.1%. Fiala et al. also determined that slight, moderate, and severe inflammation of the oviducts was 74.3%, 17.6%, and 8.0%, respectively, and they speculated that this could be a cause of subfertility in mares.
The presence of collagenous masses within the oviduct that might occlude its lumen has been documented.11 Dye tests are used in cattle to test the patency of the oviducts, but this is difficult in the mare because of the tightness of the uterotubal junction. Case studies have shown that blocked oviducts in the mare are rare. In a study by Allen et al.,12 15 mares 10-21 years of age with unexplained infertility were treated with 0.5 ml of gel containing 0.2 mg of PGE2α. The solution was dripped onto the oviducts through a laparoscope, and 14 of 15 mares were pregnant in that or the next breeding season. The authors concluded that older mares suffering repeated conception failure over several breeding seasons in the absence of identifiable pathology may benefit from PGE2α gel treatment. These authors also concluded that oviductal blockage does occur sporadically within any population of aging brood mares, and this results from a moveable accumulation of intraluminal debris rather than any permanent physical obstruction.
Periovarian Cysts
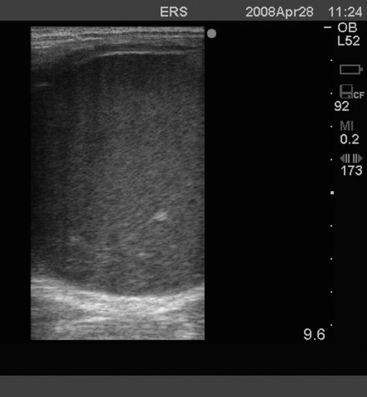
Cysts lying within the ovarian stroma near the ovulation fossa of the ovary arise from the surface epithelium and are often seen in older mares during examination of the ovary. They are known as retention, inclusion, or fossa cysts and generally have no adverse effect on fertility (Fig.13-5, A).
Fossa cysts, normally only a few millimeters in diameter, are not usually seen as large as in Fig. 13-5, B. Care must be taken not to confuse them with a normal ovary with several small follicles (see Fig. 13-5, C) or conceptus. Careful examination allows accurate identification of their position; if they are particularly large and/or numerous, as can occur in older mares, they could impede the release of the oocyte from the ovulation fossa. Small nodules located within the loose connective tissue covering of the ovary known as adrenocortical nodules have also been identified.
FUNCTIONAL SUBFERTILITY
Pregnancy and Pseudopregnancy
Pseudopregnancy is a term used to describe a syndrome in which non-pregnant mares that have been bred do not return to estrus. It occurs if there is EED after 15 days of gestation with persistence of the corpus luteum resulting in a prolonged luteal phase. The cervix remains tightly closed and the uterus is tense and tubular. It is differentiated from pregnancy by the absence of a conceptus on ultrasound examination. If early fetal death occurs after endometrial cup formation at 36 days, mares either become anestrus or show estrus with irregular follicular patterns. In the latter case, follicular luteinization without ovulation is thought to occur and, therefore, the estrus is not fertile. This failure of ovulation lasts until the endometrial cups regress spontaneously at 90–150 days. There is currently no practical way of destroying endometrial cups prematurely. It has also been reported recently that endometrial cups can be retained after abortion, fetal death, or parturition. These mares will have irregular follicular activity and a high incidence of follicular luteinization. It has recently been reported that in a small number of mares, endometrial cups may persist for many months following pregnancy loss.14 This may explain the failure of some mares to show regular behavioral estrus during the breeding season.
Transitional “Spring” Estrus
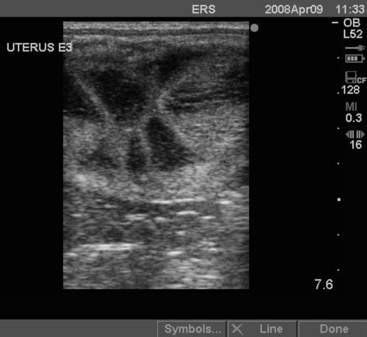
Pressure to breed mares early in the year before the onset of their natural breeding season can cause problems for the veterinarian. Because of the considerable variation in the duration of estrus during the transitional period, efficient breeding of the mare can be difficult. During the transitional period, the behavior is variable, ranging from total rejection of the stallion, to interest but resistance to his mounting, to normal acceptance. These behavioral signs can be consistent or inconsistent. Practitioners traditionally recommend that the interval between matings should not exceed 3 days if a normal fertile stallion is being used; however, the optimal interval between breeding and ovulation is not accurately defined. Few critical studies exist on survival time of sperm in the oviduct of the mare. No significant difference between day of insemination in pregnancy rate for mares inseminated from 1–6 days before ovulation has been reported.15 It is important not to begin breeding too early or the mare will be mated too many times. A recent study found that intervals between mating and ovulation of >2 days significantly lowered pregnancy rates.16 The optimum interval may depend on susceptibility of the mare to delayed uterine clearance (DUC) (see later). In mares with DUC, a longer interval from mating to ovulation >48 hours resulted in higher pregnancy rates.17 The appearance of uterine edema (Fig. 13-6) is an indication that the follicles present gain steroidogenic competence, leading to an increase in circulating estrogen concentrations, which cause the release of LH from the pituitary as a result of a positive feedback mechanism. Estrogen, in the absence of progesterone, is responsible for the appearance of uterine edema, so this may be why the detection of uterine edema is clinically important in signaling the emergence of the mare from the transitional period into the ovulatory period.
Visualization of uterine edema is done by thorough ultrasonic examination and rectal palpation, which reveals transitional follicles reaching preovulatory size (>35 mm). A detailed description of uterine edema patterns is discussed in Chapter 12. Visual identification of a corpus luteum with progesterone levels >4 ng/ml confirms that the first ovulation has occurred and, hence, normal ovarian cyclic activity.
OVULATORY DYSFUNCTION
Anovulatory Hemorrhagic Follicles
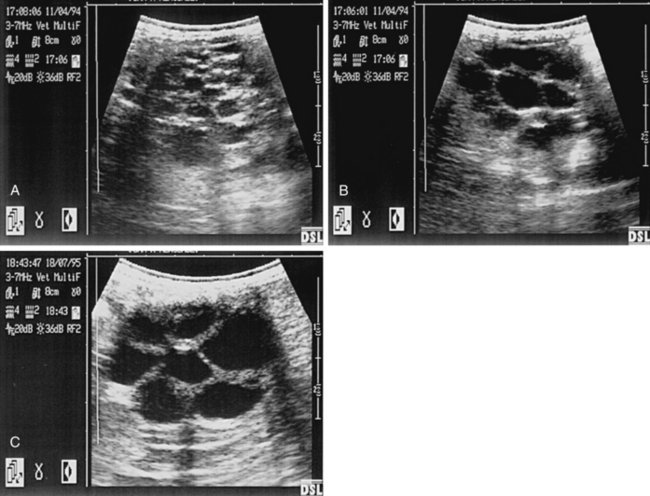
A form of ovulatory failure has been described in the mare in which the preovulatory follicle grows to an unusually large size, fails to rupture and ovulate, typically fills with blood and fibrin (Fig. 13-7), and then gradually regresses. These structures can be as large as 8–10 cm and occasionally much larger and may develop an outer wall of luteal tissue. These structures if undisturbed can persist for a variable period ranging from 3 to 7 weeks, but prostaglandin administration will accelerate their regression when luteal tissue is present. The condition is known as hemorrhagic anovulatory follicle syndrome (AHF) and is initially recognized, using transrectal ultrasound, by the presence of scattered free-floating echogenic spots within the follicular antrum (Fig. 13-8). As the blood coagulates and the fibrin deposits, the ultrasonic appearance varies from honeycomb, or net-like, to a uniformly echogenic mass (Fig. 13-9). The palpation features of these structures are smooth, with varying degrees of firmness. This can be confusing in that they may feel like preovulatory follicles or corpora hemorrhagica, or they may become very large. The most obvious difference in their appearance is when they are examined ultrasonographically. The structures may have a similar appearance to that of a granulosa thecal cell tumor (GTCT): the anechoic areas are separated by trabeculae and are similar to those of a multicystic GTCT. The diagnosis of a hemorrhagic follicle may be made on the basis of clinical signs, namely maintenance of cyclicity, a normal contralateral ovary, the presence of an ovulation fossa, and speed of enlargement and regression of the ovary with time.
Hemorrhagic follicles may be difficult to predict before the appearance of the first signs. The cause of these hemorrhagic follicles is not known but is likely to be possible hormonal pituitary insufficiency to induce ovulation. Similar structures are seen under continued equine chorionic gonadotropin (eCG) stimulation during days 40–50 of pregnancy. However, in a study by Gastal et al.,18 the only abnormality found in AHFs compared with normal ovulatory follicles was an elevated estrogen level and an increase in vascularity of the follicular wall of the AHF.
Anovulatory follicles were reported by McCue and Squires19 to occur in approximately 8.2% of estrous cycles. These authors also reported an increased incidence associated with age and make a distinction between the majority of anovulatory follicles that become luteinized (86%) and those that remain as follicular structures (14%), with the latter ones not responding to prostaglandin.
Anovulatory Follicles in Aged Mares
Even though there is no documented menopause in mares, an age-related ovulation failure has been documented.20 Some aged mares, particularly those >20 years of age, fail to ovulate, despite showing estrous behavior, and hence anovulation may not be detected. On ultrasound examination, their ovaries resemble those of seasonally anovulatory mares with a few small (<10 mm) follicles. Endometrial biopsy shows evidence of gland atrophy. Treatments for these conditions, although not consistent, include eFSH or GnRH therapy. On the other hand, it is important to recognize that some mares ovulate smaller follicles (20–25 mm) and ovulation might go undetected. In these instances progesterone detectable in blood and uterine and cervical tone are present and endometrial biopsy shows evidence of glandular branching.
Ovarian Neoplasia
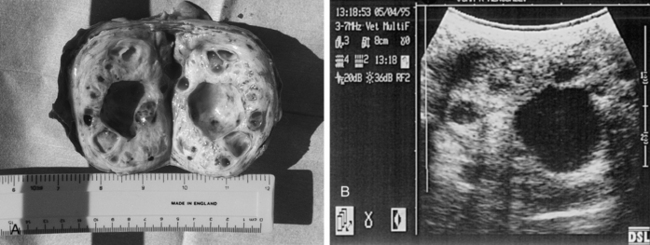
Although many types of tumors have been described, ovarian neoplasia is uncommon in the mare, with GTCTs being by far the most common.21 GTCTs arise from the sex cord stromal tissue within the ovary and may be hormonally active, producing variable amounts of steroids that cause behavioral changes and alteration in normal cyclic activity. Mares can exhibit nymphomania, anestrus, or aggressiveness with signs of virilism (clitoral enlargement, stallion-like conformation). There appears to be no breed predisposition for GTCTs, and there is a wide range of age distribution. The tumors are often large before they are diagnosed. The presence of one grossly enlarged ovary (>10 cm diameter) (Fig. 13-10, A) with the opposite ovary being small and firm on palpation with no visible follicles of >1 cm (resembling an ovary of a mare in deep anestrus) is indicative but not diagnostic of a GTCT. In mares with GTCTs, behavioral changes alone can be misleading because many affected mares do not show virilism or any other behavioral changes, and tumors other than GTCTs can also result in elevated plasma testosterone values. Owners sometimes express the opinion that their mare is “awkward” when in estrus, and they request veterinary treatment. Frequently such mares are required to perform to a high level (e.g., advanced dressage). If examination during a reported period of abnormal behavior reveals marked follicular development, it is tempting to diagnose cystic ovaries as the cause of the behavioral changes. On other occasions, the mare may even be in diestrus when examined. In any case, owner pressure to perform an ovariectomy on suspicion of a GTCT should be resisted, at least until the mare has been monitored throughout several cycles to determine whether her behavioral problems are related to estrus.
When the behavioral problems are thought to be truly linked to estrus, daily supplementation with progesterone or a synthetic progestagen should prevent the mare from showing estrous behavior. Although rare in an unbred mare, there is a possibility of an increased risk for endometritis in a mare on long-term progesterone supplementation, and she should be monitored. In addition, the problems may well recur following cessation of treatment. Abnormal behavior related to estrus can be solved with pregnancy. Potential disadvantages of getting the mare pregnant are that she cannot be shown or compete in the later stages of pregnancy and the problems may recur after birth. Veterinarians dealing with mares exhibiting abnormal behavior should also be aware that refractory visceral pain can be induced in mares with severe pneumovagina and that a simple Caslick operation can sometimes solve the problem.22
The presence of a large ovary is not necessarily indicative of a tumor, and removal of an ovary should be done only after thorough client education and discussion and consent. It is important to accurately diagnose the reason for the enlarged ovary. For example, in one report, 39% (11 of 28) of surgically excised enlarged ovaries did not warrant removal.23 Cases of GTCT may be found at routine examination of mares, maybe even after foaling, and rarely have these mares shown any behavioral changes. Larger tumors that have been present for some time are more likely to cause erratic behavior and colic signs.
It is important not to presume that a large ovary in a mare with behavioral problems is caused by a GTCT. A peripheral blood sample for a GCTC panel (testosterone, estrogen, progesterone, and inhibin levels) is useful. Increased concentrations of testosterone occur in approximately 50% of cases, estradiol concentrations may be raised, and progesterone concentrations are usually low (<1 ng/ml) because these mares are mostly not cycling. Identification of elevated concentrations of the hormone inhibin is more reliable than testosterone in confirming the presence of a GTCT. In a single blood sample of a mare with a granulosa cell tumor, inhibin is elevated in approximately 90% of cases.24,25
The secretion of high amounts of inhibin by the neoplastic granulosa cells inhibits FSH secretion and is thought to be the reason for atrophy of the contralateral ovary—a sign that is present in well over 90% of the mares with a GTCT.12 A GTCT often appears ultrasonographically as a large (7–40 cm), spheric mass, with a multicystic or “honeycomb” appearance (Fig. 13-10, B). However, there is no typical appearance of a GTCT on ultrasound. Some are uniformly dense and others have a single, large, fluid-filled cyst or even several large cysts. The echogenicity of the cyst wall differentiates it from persistent, large anovulatory follicles. However, the ultrasonic appearance of some GTCTs can be similar to that of luteinized, unruptured (“hemorrhagic“) follicles. Histopathologic examination is the only method of obtaining a definitive diagnosis.
Unilateral ovariectomy is the only satisfactory treatment for GTCTs, because the prospect of breeding from the mare is extremely poor unless the neoplastic ovary is removed. It is important not to be too hasty in removing the ovary, and a mare should always be scheduled for a second examination some weeks later. In the case of a tumor, the ultrasonic appearance would change little in the short term and would certainly appear similar if reexamined several weeks later. The reproductive prognosis of the mare is generally good, depending on the state of inhibition of the other ovary and provided that no uterine tissue had to be removed. Most mares return to normal cyclic ovarian activity, although this often takes as long as one breeding season, especially in cases of severe suppression of the remaining ovary. Most GTCTs are unilateral, although a bilateral case has been reported. Metastasis of the tumor is rare but does occur.21
Multiple Ovulation
Double ovulations occur during 8%–35% of estrous cycles, the frequency depending on the breed and type of the mare with Thoroughbreds and Warmbloods having the highest rate and ponies the lowest rate. Accurate detection of such ovulations is important because twinning is highly undesirable. Rectal palpation alone can be misleading in detecting a double ovulation, particularly when the two follicles are on the same ovary. The use of ultrasound examination of the ovaries, which should routinely be performed in conjunction with a thorough rectal examination, usually allows detection of twin follicles and double ovulations. Sometimes, the ovulatory area can appear indistinct for the first 24 hours; in these cases, the mare should be checked again 2 days later, when it can be seen more easily whether there is more than one corpus luteum (Fig. 13-11).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree