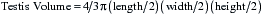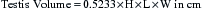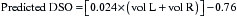Chapter 1 ANATOMY AND PHYSICAL EXAMINATION OF THE STALLION
EXTERNAL GENITALIA
The Scrotum
The scrotum of the stallion is located high in the inguinal region and is slightly pendulous. It forms two distinct pouches that contain, protect, and thermoregulate the testes, epididymides, spermatic cords, and cremaster muscles. The testes are located in the scrotum to maintain testicular temperature at several degrees below core body temperature, a necessity for normal spermatogenesis.1,2 Thermography of scrotal contents of stallions has demonstrated a scrotal skin temperature of 33°C, with testis contents at 30.5° to 32.5°C.1
The wall of the scrotum consists of four layers: (1) skin, (2) tunica dartos, (3) scrotal fascia, and (4) parietal vaginal tunic.3–6 The scrotal skin is thin, generally hairless, and slightly oily, containing numerous sebaceous and sweat glands, which assist in testis thermoregulation.3,7 The tunica dartos layer is adherent to the scrotal skin and consists of muscular and fibroelastic tissue. It lines both scrotal pouches and extends into the median septum, seen externally as the median raphae of the scrotum. The degree of contraction or relaxation of this layer allows alterations in the size, shape, and position of the scrotum in relation to the body wall, thereby aiding testis thermoregulation. The scrotal fascia, a loose connective tissue layer between the tunica dartos and parietal vaginal tunic, allows the testes and associated parietal tunic layer to move freely within the scrotum.5 The innermost layer of the scrotum, the parietal vaginal tunic, is an evagination of the parietal peritoneum through the inguinal rings, which forms during testicular descent. This layer forms a sac that lines the scrotum and is closely apposed to the visceral vaginal tunic, the outer layer of the testis. The vaginal cavity is the space between the parietal and visceral layers of the vaginal tunic. It normally contains a very small amount of viscous fluid to allow some free movement of the testis within. The vaginal cavity is a potential space within which considerable fluid may accumulate as a result of a variety of causes.
The scrotum of the normal stallion should appear slightly pendulous, globular, and generally symmetric (Fig. 1-1). Normal variations may be observed in the positioning of the testes if one is relatively anterior to or ventral to the other. The skin should have no evidence of trauma, scarring, or skin lesions. Palpation of the scrotum of a normal stallion reveals a thin and pliable covering, which slides loosely and easily over the testicles and epididymides within.
The Testes
Testicular Descent
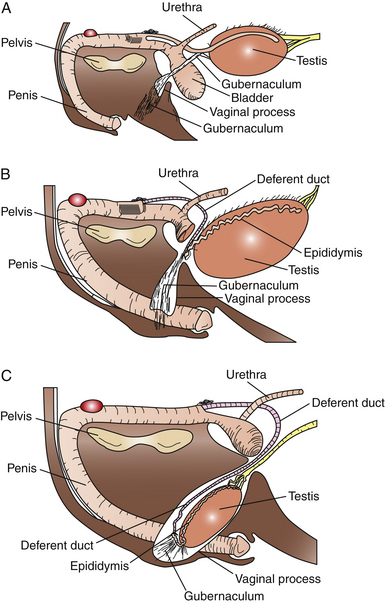
The testicles normally descend into a scrotal position between the last 30 days of gestation and the first 10 days postpartum (Fig. 1-2).7,8–11 In some colts, the testes may descend into the inguinal region and remain there for some time before fully descending into the scrotum. The hormonal factors involved in testicular descent in the stallion are poorly understood. Research in the rat has demonstrated the involvement of androgens and luteinizing hormone (LH) in the process. The timing of testicular descent coincides with significant rises in endogenous gonadotropins. It appears that an intact hypothalamic-pituitary axis, adequate LH levels, and several physical factors must be present for normal testicular descent to occur. In midgestation, the abdominal fetal testis hypertrophies significantly, resulting in the developing gonad resting close to the vaginal ring. The developing gubernaculum and abdominal pressure hold the testes in place until late gestation, despite considerable decrease in the size of the gonads later in gestation. The caudal ligament, which attaches the epididymis to the caudal pole of the testis, initially lengthens faster than the rest of the gubernaculum, resulting in the epididymis being drawn into the vaginal ring and inguinal canal. Dilation of the vaginal ring and inguinal canal, combined with abdominal pressure and traction from the gubernaculum, eventually draws the testes into the ring as well.
Failure of the testes to descend into a normal scrotal position is termed cryptorchidism. Unilateral cryptorchidism is far more common than bilateral cryptorchidism in horses. Only 14% of cryptorchid stallions had a bilateral condition.10 At birth, the testes are often located within the inguinal ring. Further contraction of the gubernaculum results in the testes being drawn into the scrotum. In a newborn colt, palpation of the prominent gubernaculum and/or epididymis within the scrotum may be mistaken for a testis. Diagnosis of cryptorchidism is attained by manual palpation of scrotal contents. Rectal palpation and careful inguinal palpation may assist in identification of an abdominally or inguinally retained testis. Ultrasonography has also been recommended as a diagnostic tool for this.12 In horses with bilaterally retained testicles or apparent geldings with stallion-like behavior, hormonal profiles may be useful in diagnosis of a retained testis.13,14 Baseline testosterone (T) levels have been suggested as a method to diagnose retained testicular tissue in an apparent gelding.13,15 However, low wintertime T values in normal stallions, a relatively high percentage of nondiagnostic values, and false-negative values are problems with the test. The use of a single measurement of plasma total estrogens or conjugated estrogens (ES), without human chorionic gonadotropin (hCG) stimulation, also appears to be reliable in the diagnosis of cryptorchidism in colts older than 3 years of age.13 A stimulation test using hCG reduces the number of nondiagnostic test results obtained with both ES and T measurement. To perform the hCG stimulation test, a baseline blood sample is collected immediately prior to injection of 5000–10,000 IU of hCG intravenously. Follow-up blood samples for T or ES are obtained 60 and 120 minutes later. A fivefold or greater increase in T indicates that a retained testicle is present. One study demonstrated that the increase in T after hCG stimulation peaked 2–3 days after injection,14 so it may be advisable to take additional samples at 24 and 48 hours following injection. One study demonstrated that T, but not ES, values increased following the hCG stimulation test. Since the testis of colts younger than 3 years of age produces significantly less ES, false-negative results may occur when ES is measured.13
Hormonal therapy to induce testis descent in a cryptorchid stallion is controversial, and no studies exist on its efficacy. LH therapy is commonly used to try to stimulate testis descent in boys with inguinally retained testis. In rats, hCG treatment was able to reverse the blocking effect of estradiol treatment on testicular descent, suggesting a role for LH in the process. One recent study evaluated the ability of the testis of prepubertal colts to respond to hCG hormone injections.16 Colts of 180–200 days age were given 2500 IU hCG intramuscularly twice weekly for 4 weeks. Treatment resulted in significant increases in testosterone values by 48 hours post-injection. The testis volume in treated colts was greater at the end of therapy (16.4 cm3 vs. 22.5 cm3) whereas untreated colts showed no significant change in testis volume. This study suggests a possible mechanism by which LH therapy for cryptorchidism might be efficacious. However, controlled studies and clinical trials involving hCG or LH therapy for cryptorchidism are lacking. In stallions, twice-weekly treatment for 4 weeks with 2500 IU to 10,000 IU of hCG has been suggested. The author has used this treatment on a low number of cases with unilateral inguinally retained testis, without apparent efficacy. The timing of therapy is generally too late, given the very early closure of the inguinal rings in colts.
Normal Testes
The testes of a normal stallion are palpable as two oval structures of nearly equal size lying horizontally within the scrotal pouches, with the tail of the epididymis directed caudally (Fig. 1-3). Normal orientation of the testis is ascertained by palpation of the tail of the epididymis and the ligament of the tail of the epididymis (or caudal ligament of the epididymis). This structure is a remnant of the gubernaculum, the fetal ligament that is believed to play a role in guiding the testis into the scrotum. The ligament is palpable as a 5- to 19-mm fibrous nodule, attaching the tail of the epididymis to the caudal pole of the testis.5 It is particularly large in newborn colts and, upon palpation, may be mistaken for a testis within the scrotum. On occasion, examination of a normal stallion may identify rotation of one or both testes, up to 180 degrees. This finding was seen in 3%–4% of “normal” light horse stallions presented for a breeding soundness examination in one study.17 Rotation may be more common in certain breeds; 9 of 23 Welsh pony stallions (39%) examined in another report exhibited testis rotation.18 The authors of the latter study commented that the high incidence appeared to be related to specific family lines. Testis rotation is believed to occur in about 15% of Paso Fino stallions (Samper, personal communication, 1997).
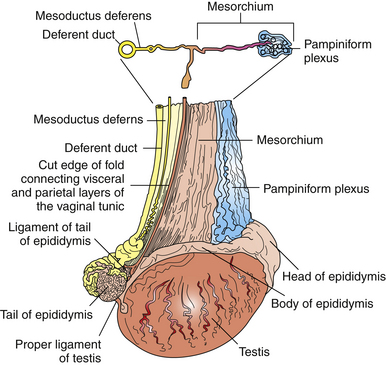
Figure 1-3 Lateral view of the right testis of a stallion.
(From Dyce KM, Sack WO, Wensing CJG: Textbook of Veterinary Anatomy. Philadelphia: WB Saunders, 1987.)
Within the scrotum, the testis is encapsulated by the tunica albuginea, a layer of tough collagenous tissue and smooth muscle, which is fused externally to the visceral layer of the vaginal tunic. The tunica albuginea sends supportive trabeculae into the testicular parenchyma, dividing the testis into lobules.5–7 Its muscular content is thought to play a role in intratesticular sperm transport and determination of testicular tone.2 Unlike the bull and other species, the testis of the stallion does not contain an axially oriented mediastinum or rete testis.3,5,6 The mediastinum testis is a partial separation within the testis consisting of fibrous tissue continuous with the tunica albuginea. In the stallion, it is located at the cranial pole of the testis, where the excurrent ducts leaving the testis cross the tunica albuginea and enter the head of the epididymis.6,7 This results in a mediastinum testis that is less prominent grossly on cut section and difficult to identify ultrasonographically. The microanatomy of the testis is described in Chapter 2.
Testicular tone is described as the degree of turgidity of the testicle; the testicle should be firm to turgid but resilient on palpation. Deviation from the normal toward a softer or firmer testis may be associated with degenerative, neoplastic, or traumatic conditions of the testis. Testicular degeneration is an acquired condition in which damage to the germinal epithelium results in eventual atrophy of the epithelium and, grossly, of the testis.7 In general, early in the course of testicular degeneration, the affected testis is softer than normal and may be enlarged. As the disease progresses, the degenerating testicle becomes small and firm as testicular parenchyma is replaced with fibrous tissue. Wrinkling of the scrotum and tunic may be evident.7 Testicular conditions may affect only one testis; therefore, comparison of the size and consistency of the two testes of any individual stallion is imperative. Changes in testicular tone or consistency are likely best determined by sequential examinations of the stallion over time, allowing the clinician to monitor the severity and rate of change as the disease progresses. Regular physical examinations of breeding stallions are an important part of routine stallion management and may allow early detection of problems that may significantly impact fertility.
Testicular Size and Volume
Each testis of a postpubertal stallion weighs between 150 and 300 grams9 and measures approximately 50–80 mm wide, 60–70 mm high, and 80–140 mm long.6 Testicular size varies among stallions depending on breed, season, age, and reproductive status.1,19–21 Table 1-1 gives the recommended minimum testicular measurements for light horse stallions. Table 1-2 gives the recommended minimum testicular measurements for miniature stallions. Testis parenchymal weight correlates highly with daily sperm production (DSP)17,20,22–24 and therefore is a useful predictor of a stallion’s breeding potential. However, because parenchymal weight cannot be measured in the live stallion, estimates must be used. In the bull, scrotal circumference is an extremely important measure used during reproductive evaluation and has been shown to be correlated with daily sperm output (DSO).25 Measurement of scrotal circumference is difficult in the stallion because the testes are held close to the body wall. However, total scrotal width (TSW) can be measured using calipers. TSW correlates well with testis parenchymal weight (r2 = 0.83) and DSP (r2 = 0.75).20,24 Caliper measurements should be used judiciously based on their inherent potential sources of error, including caliper sensitivity, operator technique, and testis location within the scrotum.20,22,24 Taking the average of several measurements helps increase repeatability and accuracy. An additional concern of the value of the TSW measure is the reliability of a single linear measurement, such as width, in estimating the true size of the testicles (three-dimensional structures).22 Even though TSW is not likely the optimal measurement for use in estimating testis size, it is easily and quickly obtained with minimal, inexpensive equipment. Table 1-3 gives a guideline of the minimum acceptable TSW for light horse stallions by age. Table 1-4 gives the calculated TSW measurements from a study of 216 miniature stallions.
Table 1-1 Mean Testicular Measurements of 43 Light Horse Stallions
| MEASURE MEAN | RECOMMENDED MINIMUM (SD) |
|---|---|
| Left width (mm) | 57.8 (5.2) |
| Left length (mm) | 103.1 (8.2) |
| Right width (mm) | 55.8 (5.8) |
| Right length (mm) | 107.5 (8.0) |
Adapted from Thompson DL, Pickett BW. Squires EL, et al. Testicular measurements and reproductive characteristics in stallions. J Reprod Fertil 27(Suppl):13-17, 1979.
Table 1-2 Testicular Measurements of Miniature Stallions by Stallion Size (n = 216)
| TESTIS MEASUREMENT | RECOMMENDED MINIMUM IN MM (+/- SD) SMALL/MEDIUM (72–96 CM) | RECOMMENDED MINIMUM IN MM (+/- SD) LARGE (97–104 CM) |
|---|---|---|
| Left width | 39.0 (0.05) | 43.13 (0.07) |
| Left length | 63.74 (0.08) | 70.30 (0.10) |
| Right width | 38.3 (0.05) | 43.00 (0.07) |
| Right length | 62.64 (0.08) | 69.20 (0.10) |
Adapted from Paccamonti DL, Buiten AV, Parlevliet JM, et al: Reproductive parameters of miniature stallions. Theriogenology 51:1343-1349, 1999.
Table 1-4 Calculated Total Scrotal Width Measurements for Miniature Stallions by Stallion Size (n = 216)∗
| MINIATURE HORSE SIZE (HEIGHT IN CM) | TOTAL SCROTAL WIDTH MEAN (MM) |
|---|---|
| Small/Medium (72–96 cm) | 72.5 |
| Large (97–104 cm) | 79.5 |
∗ TSW was calculated using the equation TSW = 1.74 + 0.696 (LW + RW).
Adapted from Paccamonti DL, Buiten AV, Parlevliet JM, et al: Reproductive parameters of miniature stallions. Theriogenology 51:1343-1349, 1999.
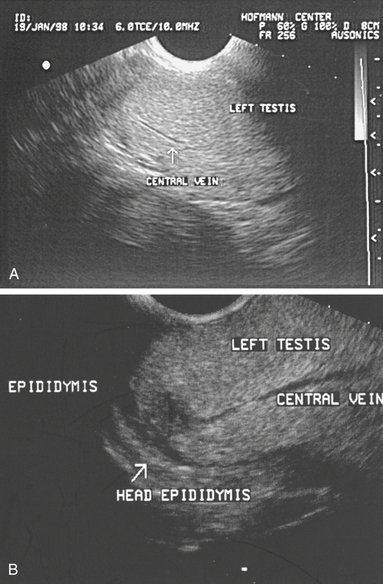
The testes should also be measured individually using calipers for length, width, and height in an effort to estimate their size.1,7,22,26 Individual testicular measurements correlate well with parenchymal weight, with r2 values ranging from 0.57 to 0.82.21,24 Correlation estimates for individual testicular measurements and DSO range from 0.52 to 0.76.20,24 Testis measurements can be assessed by either caliper (Fig. 1-4) or ultrasonographic (Fig. 1-5) measurement.22,27 Ultrasonographic measurements may be more accurate in that they are less likely to be affected by sources of error, although proper placement of the probe across the testis to ensure that a cross-sectional image is obtained is critical. Deviation toward an oblique image dramatically affects measurements obtained with this method. Testicular volume, rather than dimensions, has been suggested to more accurately predict DSO.22 Because a testis approximates the shape of an ellipsoid, the following formula is used to convert length, width, and height measurements into testicular volume22:
Love28 also recommends using this volume to predict the expected DSO of the stallion, using the following formula:
Testicular measurements are an important part of the physical examination of any breeding stallion. Because testicular size is likely as heritable in stallions as it is in bulls,17,22,29 stallions with scrotal width measurements that are less than the aforementioned guidelines should not be recommended for breeding purposes. However, for stallions selected for breeding based on performance or conformation characteristics rather than on fertility characteristics, it may be more useful to use testis measurements as a management tool. Veterinarians may caution owners that a stallion with small testicles will have lower sperm production and may not perform well with a large book of mares. However, by creating realistic owner expectations, modifying stallion management such as frequency of breeding, and/or increasing the frequency of examination of the mares so that they are bred only once close to ovulation, the veterinarian or manager can optimize the fertility of such a stallion.
Epididymides and Excurrent Duct System
Each epididymis is a highly convoluted but unbranched duct approximately 70 meters long that has a grossly distinct head, body, and tail.5–7 The head of the epididymis is a flattened structure that lies dorsomedially along the cranial border of the testis and is closely attached to it (see Fig. 1-3
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree