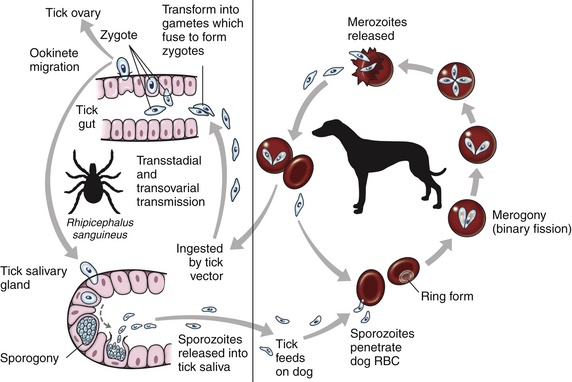
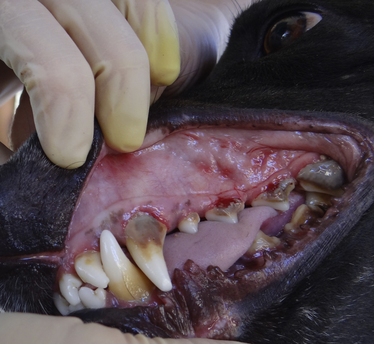
Chapter 75 Babesiosis, caused by infection with organisms from the genus Babesia, is characterized by hemolytic anemia, fever, and splenomegaly. Babesia infections can also be subclinical or cause severe life-threatening illness. Babesia spp. are intraerythrocytic protozoan parasites of the phylum Apicomplexa. Altogether, more than 100 species of Babesia have been described, and with the advent of molecular techniques such as PCR assays, new species and genotypes are identified each year. Historically, species have been named and identified based on the vertebrate host and the size of the parasite (large or small Babesia species). Large Babesia spp. are 3 to 7 µm in length, whereas small Babesia spp. are 1 to 3 µm in length. In most parts of the world, tick vectors are the most important means of transmission of Babesia species. However, for some Babesia species, such as Babesia gibsoni in North America and Europe, direct transmission between dogs, through fighting (and exchange of blood), or congenital transplacental transmission, is believed to be the most common route of transmission when competent tick vectors are absent.3–5 When ticks are involved in transmission, sporozoites are released from the tick salivary glands as the tick feeds and they enter the bloodstream of the vertebrate host (Figure 75-1). They then attach to, and are endocytosed by, erythrocytes. Within erythrocytes, they undergo asexual reproduction (merogony), and the daughter cells infect new erythrocytes. A naïve tick then ingests infected erythrocytes. It is unclear whether transformation from merozoite to gametocyte begins in the vertebrate host or in the tick. In the tick midgut, the sexual phase of reproduction occurs when the gametocytes fuse to form a zygote. The zygote invades epithelial cells of the tick gut. The resultant forms, ookinetes, leave the epithelial cell and invade either the salivary gland or the ovary, where they participate in transstadial and transovarial transmission, respectively. Prior to transmission, another asexual form of reproduction occurs in the salivary gland, sporogony. Babesia canis is the most common large Babesia species and has three distinct subspecies: B. canis vogeli, B. canis canis, and B. canis rossi. It has been proposed that these are in fact three distinct species (B. vogeli, B. canis, and B. rossi).6 B. canis vogeli is transmitted by the brown dog tick (Rhipicephalus sanguineus). Infection is most commonly diagnosed in warm, humid regions of the world, and the disease occurs throughout the year in endemic regions. B. canis vogeli can be found in Africa, Asia, Australia, Europe, and the Americas. In the United States, disease is most often diagnosed in the southern regions. B. canis vogeli reported seroprevalence in dogs has ranged from 3.8% to 59%.7 The prevalence of seroreactivity is higher in adult dogs than in dogs younger than 1 year.8 In an antibody serosurvey of dogs in Florida, 46% of 393 greyhounds were seroreactive.7 The prevalence of seroreactive dogs within these kennels ranged from 17% to 100%; the lower prevalence was noted in kennels with more intensive tick control. None of 50 non-greyhound, individually housed adult pet dogs within the same geographic area were seroreactive, which implicated environment and breed susceptibility as possible risk factors in endemic areas.7 Transplacental transmission of B. canis vogeli infections is strongly suspected but unproven in an experimental setting.9 B. canis canis is found in Europe and Africa. It is transmitted by the ornate cow tick, Dermacentor reticulatus, but R. sanguineus may also be a vector.10 The incidence of infection is highest in the fall and spring. Higher prevalence rates are most commonly found in rural or suburban areas that are adjacent to prairies or woodlands that provide suitable habitat for D. reticulatus.11 B. canis canis infection is most often diagnosed in France. Infections are being reported more frequently from other European countries such as Croatia, Poland, and Germany, possibly due to changes in the distribution of D. reticulatus.12 Outbreaks of disease due to B. canis canis infection in Polish sled dogs share many similarities with those due to B. canis vogeli infection in North American greyhounds.13 B. canis rossi is transmitted by the yellow dog tick, Haemaphysalis elliptica (formerly Haemaphysalis leachi).14 Reports of B. canis rossi infection have been limited to Africa, with the vast majority of reports coming from South Africa, where more than 10% of the dogs evaluated in some veterinary hospitals may be affected.15,16 The incidence of infection is highest during the summer months. Breed predispositions for infection have not been well studied, but the traditional fighting breeds (American pit bull terriers, Staffordshire bull terriers, and bull terriers) are more likely to die when they are diagnosed with severe babesiosis.17 A large Babesia sp. has been isolated from dogs in North America that had been splenectomized or were undergoing chemotherapy for cancer. Infected dogs have been identified in North Carolina, New Jersey, New York, and Texas.18–20 A tick vector has not been identified. A different novel large Babesia sp. was reported in a dog from Great Britain that never traveled outside of that country.21 The dog died as a result of infection. Infections with B. gibsoni occur throughout the world, and the insidious nature of this infection has allowed the inadvertent transport of infected dogs from Asia to other areas. B. gibsoni can be found in Africa, Asia, Australia, North and South America, and Europe. Depending on the availability of suitable vectors, two distinct epidemiologic scenarios exist for B. gibsoni: tick transmission and direct transmission between dogs.22,23 B. gibsoni is transmitted primarily by the tick vectors Haemaphysalis bispinosa and Haemaphysalis longicornis, and possibly by R. sanguineus.24–28 In its original endemic area of Asia, the geographic range of B. gibsoni correlates with that of H. bispinosa, and tick infestation is a risk factor for infection in nonfighting breeds. In the United States where competent tick vectors are not endemic, B. gibsoni infections occur in dogs engaged in fighting activities.23,29 Most dogs with B. gibsoni infection in the United States have been American pit bull terriers.29,30 However, an increased prevalence of infection has even been noted in fighting breeds in areas such as Japan, where Haemaphysalis spp. are endemic. In fighting breeds, transmission has been associated with a fight or bite by an infected dog or having been born to an infected bitch. Perinatal transmission is believed to occur.3–5 B. conradae (previously referred to as “the California isolate” and the “western piroplasm” and historically misidentified as B. gibsoni)31 has been detected primarily in dogs from southern California.31,32 Tick vectors have not been identified. Transmission studies that attempted to prove vector competence for R. sanguineus and Dermacentor variabilis were unsuccessful or inconclusive.33 The author has identified R. sanguineus as well as soft-bodied ticks, Ornithodoros coriaceus, on B. conradae–infected dogs. A high prevalence of B. conradae infection was noted in a kennel of greyhound mixed-breed dogs that were used to hunt coyotes.34 A history of coyote fights was a risk factor for infection. B. conradae DNA was detected in the spleen of 1 of 50 coyotes, so fighting may be one mode of transmission for this Babesia species. A B. microti–like parasite (also referred to as Babesia annae or Theileria annae) has been identified in dogs, the majority of which have lived in, or traveled to, northwestern Spain.35–38 A single American pit bull terrier confiscated from a suspected dog-fighting operation in North America was also infected.39 A high percentage of European and North American foxes are infected with a genetically identical parasite.40,41 A tick vector has not been definitively identified, but in Spain an association between Ixodes hexagonus infestation and B. microti–like infection has been observed.42 Feline babesiosis has not been studied as extensively as canine babesiosis. Several small Babesia species have been identified in cats, which include Babesia felis, Babesia cati, and Babesia leo. Babesia felis is a highly pathogenic Babesia species that infects domestic cats in southern Africa and the Sudan.43 Infection of domestic cats has primarily been identified along the coast of South Africa.44,45 Affected cats are usually younger than 3 years, and there is no recognized breed or sex predilection. Babesia cati is less pathogenic and found primarily in India. Babesia leo has been detected in lions (Panthera leo) in Kruger National Park, as well as a single domestic cat in a molecular survey.46 A small piroplasm was visualized in blood smears from feral cats in Rio de Janeiro, Brazil, but no molecular data were available to confirm the identity of these organisms.47 Large Babesia species also appear to infect cats. DNA sequences, most similar to those of B. canis canis and a B. microti–like parasite, have been amplified from the blood of three cats with retroviral infections from Spain and Portugal, but no organisms were visualized.48 B. canis presentii was identified in two cats in Israel.49 Another B. canis–like parasite has been identified in a naturally infected cat from Poland.50 Feline babesiosis has not been reported in the United States. Clinical Signs and Their Pathogenesis The pathogenicity of Babesia organisms is determined primarily by the species and strain involved. Host factors, such as the age of the host and the immunologic response generated against the parasite or vector tick, are also important. Although most often subclinically infected, dogs infected with “avirulent” species such as B. canis vogeli may have severe clinical disease. Similarly, dogs infected with “virulent” species such as B. canis rossi may be subclinically infected without overt clinical or laboratory findings. However, features that should raise clinical suspicion for babesiosis include fever, thrombocytopenia, hemolytic anemia, and splenomegaly. Dogs often have nonspecific signs such as lethargy, anorexia, and weakness (Box 75-1). Occasionally owners note jaundice, mucosal pallor, or discoloration of the urine caused by bilirubinuria or hemoglobinuria. Dogs infected with B. canis vogeli may have fever of unknown origin only without overt hematologic abnormalities.51 Co-infections with other tick- or bloodborne pathogens may also influence the clinical signs of disease. Parasite antigens are incorporated on the erythrocyte surface and induce host-opsonizing antibodies, which in turn leads to removal of infected erythrocytes by the mononuclear-phagocyte system. Soluble parasite antigens can also adhere to the surface of noninfected erythrocytes and platelets. This may lead to their opsonization by antibodies, with or without complement, and account for hemolytic anemia and thrombocytopenia that is often not correlated with the level of parasitemia. Parasitemia and anemia are more severe in splenectomized dogs, and splenectomy may precipitate disease in dogs with chronic subclinical infections. Mechanisms other than immune-mediated destruction that contribute to erythrocyte damage include increased erythrocyte osmotic fragility, direct injury of erythrocytes by Babesia parasites, accumulation of cyclic nucleotides, and oxidative injury.52–56 Lipid peroxidation increases erythrocyte rigidity and slows the passage of erythrocytes through capillary beds. Soluble parasite proteases activate the kallikrein system and induce fibrinogen-like protein formation. These proteins make erythrocytes more “sticky,” and they sludge in capillary beds, which contributes to anemia and many of the other potential clinical signs. The most severe sludging occurs in the central nervous system (CNS) and muscles. Thrombocytopenia may result from immune-mediated or coagulatory consumption of platelets. Despite severely decreased platelet counts, bleeding is rarely observed in dogs infected with most Babesia strains, and other abnormal coagulation test results are uncommon (with the exceptions of B. conradae and also B. canis rossi). Dogs with B. conradae infection have experienced life-threatening hemorrhages, the pathogenesis of which requires further study.57 Other possible complications include membranoproliferative glomerulonephritis, which may have an immune-mediated pathogenesis.58–60 Renal failure occurs in up to 40% of dogs infected with B. microti–like parasites, and their presence has been associated with increased mortality.35,36,38,61,62 Nonregenerative anemia, azotemia, and proteinuria with high urine protein/creatinine ratios were found in these dogs.35 Virulent B. canis canis and especially B. canis rossi strains induce a profound systemic inflammatory response, which can result in a severe sepsis-like syndrome with multiple organ dysfunction (see Chapter 86). The majority of dogs infected with B. canis rossi have uncomplicated babesiosis and can be treated as outpatients. However, up to 31% of the dogs examined at a university clinic required hospitalization, and 10% of hospitalized dogs did not survive.17 Severe clinical illness in dogs infected with B. canis rossi results from hypotension, acute renal failure (ARF), neurologic complications, disseminated intravascular coagulation (DIC), a hepatopathy, and acute respiratory distress syndrome (ARDS). Tissue hypoxia results from anemia, shock, vascular stasis, excessive endogenous production of carbon monoxide, parasitic damage to hemoglobin, and decreased ability of hemoglobin to off-load oxygen.53,63 Lactic acid generation from tissue hypoxia can result in severe metabolic acidosis.64 “Red biliary syndrome” is a paradoxical phenomenon of severe intravascular hemolysis (manifested as hemoglobinemia and hemoglobinuria) in combination with hemoconcentration (high-reference-range or elevated hematocrit).63 The hemoconcentration is thought to occur when plasma shifts from the vascular to the extravascular compartment as a result of increased capillary permeability, with a resultant decrease in blood volume. Hemoconcentration has been associated with cerebral babesiosis, DIC, ARF, and ARDS. Neurologic complications result from sludging of parasitized erythrocytes in CNS capillary beds, with congestion and macroscopic and microscopic hemorrhages. Severe hypoglycemia can also result in neurologic signs. Other complications of severe babesiosis include pancreatitis, rhabdomyolysis, ocular involvement, upper respiratory signs, cardiac arrhythmias, necrosis of the extremities, and fluid accumulation. Pulmonary, CNS, and renal complications are associated with a higher rate of mortality. Persistent lactate concentrations above 40 mg/dL are a poor prognostic indicator for survival.65 Cats with babesiosis from southern Africa generally show lethargy, anorexia, weakness, an unkempt haircoat, and/or diarrhea.45 Fever and icterus are less common. Anemia can be severe and is the underlying reason for the clinical signs. The disease is chronic, and signs may not be apparent until a later stage of illness. Cats usually adapt to the anemia and may have only mild clinical signs until they are stressed by handling. Complications of the hemolytic anemia have included hepatopathy, pulmonary edema, renal failure, CNS signs, and concurrent infections. Physical examination abnormalities in most dogs with babesiosis consist of fever, mucosal pallor, lethargy, splenomegaly, and bounding pulses. Fever is not consistently identified, because it often waxes and wanes. Tachycardia and tachypnea may be present in severely anemic dogs. Mucosal hemorrhages and/or epistaxis may be present in dogs with B. conradae infection (Figure 75-2), and excessive bleeding from venipuncture sites may be noted. FIGURE 75-2 Greyhound infected with Babesia conradae. Mucosal hemorrhages are apparent. These resolved after treatment with atovaquone and azithromycin. (Courtesy Dr. Jane Sykes, University of California, Davis.) Dogs with severe babesiosis may show CNS signs such as incoordination, pelvic limb paresis, muscle tremors, nystagmus, anisocoria, intermittent loss of consciousness, seizures, stupor, coma, aggression, or vocalization.63 Dogs with red biliary syndrome may have congested mucous membranes or icterus. Other clinical abnormalities in dogs with severe babesiosis include tachypnea, increased lung sounds, and cardiac arrhythmias. Affected cats may be lethargic, pale, tachycardic, and tachypneic, and in some cases, icteric. In dogs with babesiosis, the primary hematologic abnormalities are anemia and thrombocytopenia. Thrombocytopenia is often present, even when anemia is absent. A mild, normocytic, normochromic anemia is generally noted in the first few days after infection, which becomes macrocytic, hypochromic, and regenerative as the disease progresses. Uncommonly with B. canis rossi infections, a relative polycythemia may be noted.63 Leukocyte abnormalities are inconsistently observed but may include leukocytosis (with or without a left shift), leukopenia, neutrophilia, neutropenia, lymphocytosis, and/or eosinophilia.34,66–68 Leukopenia or a low-normal leukocyte count due to relative neutropenia has been frequently observed in dogs from Europe with B. canis infections and dogs from the United States with B. conradae infections.34,68 Autoagglutination, positive direct antiglobulin (Coombs’) tests, and spherocytosis may also be present. In cats, anemia associated with B. felis infection is typically macrocytic, hypochromic, and regenerative.69 Thrombocytopenia is an inconsistent finding. There are no pathognomonic biochemical findings in dogs with babesiosis. Common findings in North American dogs include hyperglobulinemia (which may be present in the absence of other laboratory abnormalities), mildly increased liver enzyme activities, and, less commonly, hyperbilirubinemia. A study of dual infections with B. canis and Ehrlichia canis showed that the prevalence of hyperglobulinemia was higher in dogs with dual infections than in dogs with a single infection caused by either organism.70 Hyperbilirubinemia is a consistent finding during acute disease caused by B. canis canis and B. canis rossi but not by B. gibsoni or B. conradae.34,67 Dogs with severe B. canis rossi infections may have hemoglobinemia, moderately increased liver enzyme activities, increased BUN and serum creatinine concentrations, hypoalbuminemia, hypoglycemia, and metabolic acidosis. Cats infected with B. felis typically have elevated serum ALT activity and total bilirubin concentrations. Serum protein values are usually within reference limits, but hyperglobulinemia can occur. Renal parameters are unaffected.69
Babesiosis
Etiology and Epidemiology
Babesiosis in Dogs
Babesia canis
Unnamed Large Babesia Species
Babesia gibsoni
Babesia conradae
Babesia microti–Like Organism
Feline Babesiosis
Clinical Features
Canine Babesiosis
Severe Babesiosis
Feline Babesiosis
Physical Examination Findings
Diagnosis
Complete Blood Count
Serum Chemistry Profile
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Babesiosis
Only gold members can continue reading. Log In or Register to continue