CHAPTER | 15 Avian and Exotic Animal Dermatology
Parasitic
Acariasis
Features
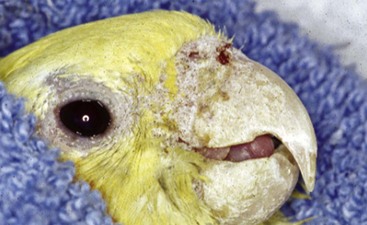
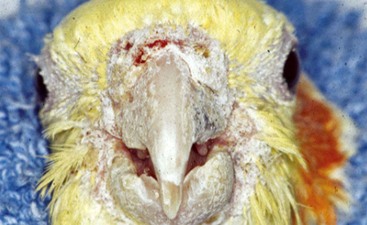
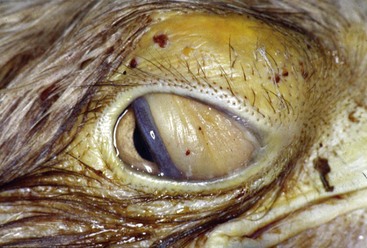
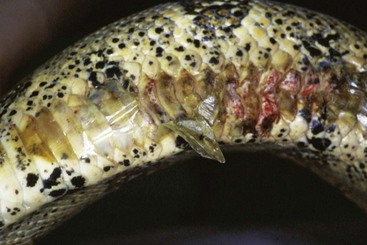
Common mites of avian and exotic animal pet species are generally species specific (Table 15-1). Fur mites generally cause dandruff and patchy alopecia with little associated pruritus. The rabbit fur mite (Cheyletiella parasitovorax) is considered zoonotic, causing mild hyperemia and pruritus in humans. The ear mite, Otodectes cynotis, is commonly found in ferrets and can spread between cats and ferrets; therefore it is important to treat all susceptible pets in the household. The presence of black earwax in ferrets does not necessarily denote ear mite infestation. The rabbit ear mite, Psoroptes cuniculi, can cause such an accumulation of dry crusts that an upright ear can be weighted down into the lop-ear position. Demodex spp. are commonly demonstrated in hamsters greater than 2 years of age with concomitant Cushing’s disease. Hamsters are generally exposed to the mites when young but do not exhibit clinical signs until immunocompromised. Rabbits and guinea pigs with intense pruritus should be checked for their respective mites because this would be the most common differential. Birds, typically budgerigars and canaries, infected with Knemidokoptes generally show pitting of the beak and facial skin (budgerigars), or pitting of the skin of the legs (canaries). Snake mites (Oophyionysus spp.) can be found anywhere on the snake’s body, but typically they can be found around the eye or between the cleft of skin under the chin. Severe infestations in snakes can cause significant anemia. African hedgehogs kept as pets in the United States can present with Chorioptes spp. mites, resulting in quill loss and skin crusting and flaking.
TABLE 15-1 Common Mites of Avian and Exotic Animal Pets
| Mite | Characteristics |
|---|---|
| Fur Mites | |
| Mouse | |
| Myobia musculi | Found over head, neck, and shoulder area |
| Radfordia affinis | |
| Myocoptes musculinus | Found over entire body |
| Rat | |
| Radfordia ensifera | |
| Rabbit | |
| Cheyletiella parasitovorax | “Walking dandruff mite,” zoonotic |
| Listrophus gibbus | |
| Ear Mites | |
| Rat | |
| Notoedres muris | |
| Rabbit | |
| Psoroptes cuniculi | Dry crusts in ear canal and pinna |
| Ferret | |
| Otodectes cynotis | Dark, waxy discharge in ears |
| Hamster | |
| Notoedres notedres | Found over ears, nose, feet, and anus |
| Skin | |
| Hamster | Common secondary to Cushing’s disease |
| Demodex criceti | Found in epidermis |
| Demodex aurati | Found deep (hair follicles, sebaceous glands) |
| Guinea Pig | |
| Trixacarus caviae | Marked purities: neck, shoulder, inguinal |
| Chirodiscoides caviae | Nonpruritic: lumbar, lateral rear limbs |
| Rabbit | |
| Sarcoptes scabiei | Intense pruritus, self-mutilation |
| Budgerigars and Canaries | |
| Knemidokoptes pili | “Scaly leg and face mite,” pitting beak, skin |
| Snake | |
| Ophionyssus natricis | Check crypts around eye and cleft under chin |
| Hedgehog | |
| Chorioptes spp. | Quill loss; skin crusting and flaking |
Treatment and Prognosis
 Do not hang mite treatments, such as those that contain paradichlorobenzene, on the side of the cage, as they may be toxic to birds and they do not rid the bird of mites.
Do not hang mite treatments, such as those that contain paradichlorobenzene, on the side of the cage, as they may be toxic to birds and they do not rid the bird of mites. In rabbits, selamectin has been shown to clear infection of ear mites with one dose. When treating rabbits for ear mites, it is important to not remove the crusts, as they will fall out within 1 week after treatment with very little trauma.
In rabbits, selamectin has been shown to clear infection of ear mites with one dose. When treating rabbits for ear mites, it is important to not remove the crusts, as they will fall out within 1 week after treatment with very little trauma. Because snake mites spend time off the host, any porous material in the enclosure should be discarded, and nonporous surfaces can be sprayed with a well-shaken mixture of ivermectin at 5 mg/L water. The propylene glycol base does not mix well with water.
Because snake mites spend time off the host, any porous material in the enclosure should be discarded, and nonporous surfaces can be sprayed with a well-shaken mixture of ivermectin at 5 mg/L water. The propylene glycol base does not mix well with water.
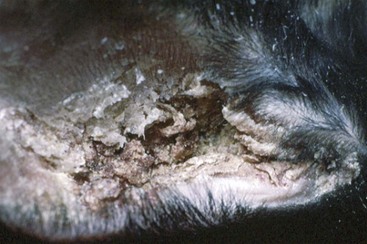
FIGURE 15-3 Acariasis.
Rabbit with scabies due to a Sarcoptes spp. mite. This rabbit had intense pruritus.
Pediculosis
Diagnosis
Microscopy of tape preparation or plucked hair demonstrates the parasite or the ova (nits).
Subcutaneous Parasites
Treatment and Prognosis
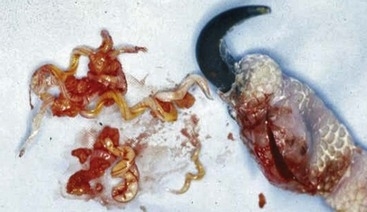
 For rabbits, remove the parasite under general anesthesia by surgically incising the skin in each direction from the breathing hole, taking care not to traumatize the parasite to prevent release of parasite antigen. Clean and débride the wound and leave open to heal by second intention, with daily cleansing.
For rabbits, remove the parasite under general anesthesia by surgically incising the skin in each direction from the breathing hole, taking care not to traumatize the parasite to prevent release of parasite antigen. Clean and débride the wound and leave open to heal by second intention, with daily cleansing.Myiasis, Flea or Tick Infestation
Treatment and Prognosis
 Fleas: In general, the treatment for fleas in ferrets is similar to that for cats. Rabbits on the other hand should never be given fipronil (Frontline) as it causes seizures and/or death in rabbits. Rabbits can be given imidocloprid (Advantage) or pyrethrin products.
Fleas: In general, the treatment for fleas in ferrets is similar to that for cats. Rabbits on the other hand should never be given fipronil (Frontline) as it causes seizures and/or death in rabbits. Rabbits can be given imidocloprid (Advantage) or pyrethrin products. Ticks: Direct removal. Recently imported animals with ticks should have the ticks submitted for identification to be sure they are not foreign. Always save parasite specimens in 70% alcohol, never formalin, as formalin renders physically indentifying structures on the parasite unusable.
Ticks: Direct removal. Recently imported animals with ticks should have the ticks submitted for identification to be sure they are not foreign. Always save parasite specimens in 70% alcohol, never formalin, as formalin renders physically indentifying structures on the parasite unusable.
FIGURE 15-15 Myiasis, Flea or Tick Infestation.
Wild cottontail rabbit infested with unidentified tick species.
Bacterial
Rabbit Syphilis (Treponema cuniculi)
Top Differentials
Differentials include acariasis (see Figure 15-3) and papillomavirus, (see Figure 15-45), which typically involve other areas of the body than the mucocutaneous junctions.
Treatment and Prognosis
Consult Carpenter’s Exotic Animal Formulary for further drug information.
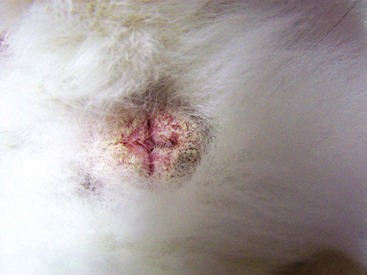
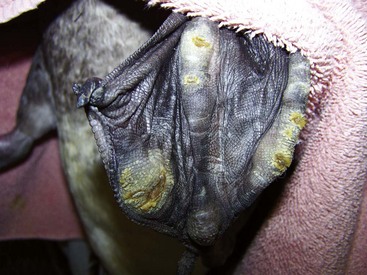
FIGURE 15-17 Rabbit Syphilis.
Perineal region of the same rabbit as in Figure 15-18, with typical mild to moderate case of rabbit syphilis caused by Treponema cuniculi. Lesions are near mucocutaneous junctions.
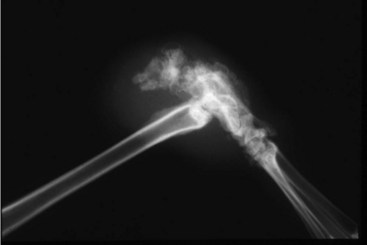
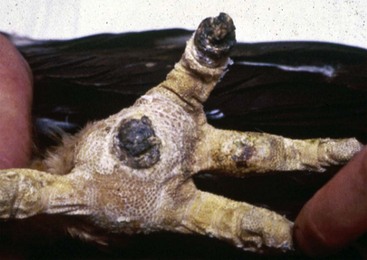
Ulcerative Pododermatitis—rabbit (sore hock), bird (bumblefoot)
Diagnosis
Diagnosis is based on direct visualization. A radiograph can determine if there is osteomyelitis.
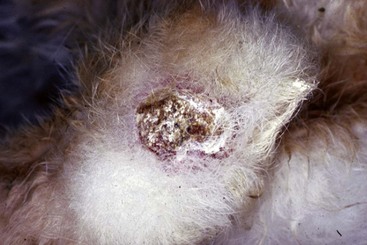
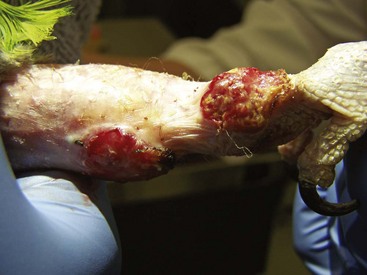
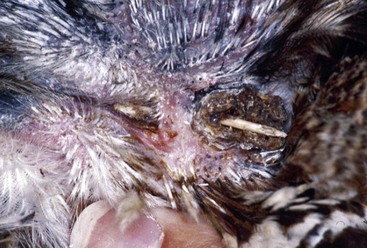
Bacterial Dermatitis—bird, ferret
Treatment and Prognosis
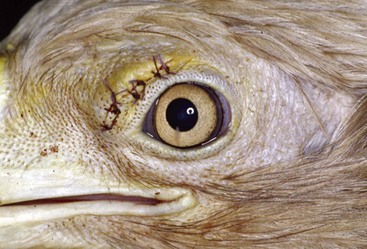
FIGURE 15-29 Bacterial Dermatitis.
Same captive adult bald eagle with a supraorbital SC abscess after surgical removal.
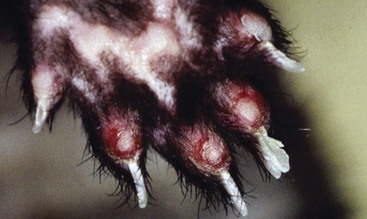
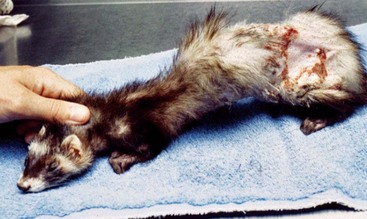
FIGURE 15-31 Bacterial Dermatitis.
Same ferret as in Figure 15-30, with bacterial dermatitis showing plantar surface of foot.

FIGURE 15-33 Bacterial Dermatitis.
Same gecko as in Figure 15-32 showing dysecdysis (abnormal shedding of the entire body). Retained sheds on toes have a constricted blood supply and cause necrosis of the toes. This gecko ended up responding to appropriate antibiotic therapy based on culture and multiple warm water soaks but lost almost all its toes.
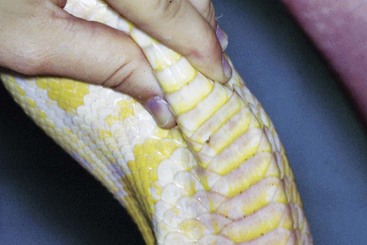
Sepsis in Reptiles versus Shedding
Diagnosis
If the skin blanches with digital pressure, it is hyperemia, and not the pigmentation of the animal.