Norin Chai More than 6200 species of anurans have been currently recorded,1 and these live on all continents except Antarctica. Although the larvae are aquatic, anurans have successfully expanded their habitats into numerous and markedly different ecologic types, in the Arctic Circle, in deserts, in tropical rain forests, and practically everywhere in between. Actually, 54 families are proposed; however, anuran taxonomy is still a matter of dispute. Table 1-1 lists some relevant families. The goliath frog (Conraua goliath), the largest anuran, is able to grow up to 33 cm and weigh up to 3 kg. The smallest known frog is Paedophryne amauensis (Microhylidae); with its 7.7 mm length, it is also the world’s smallest known vertebrate. In captivity, average life spans are typically 4 to 15 years. The goliath frog may live up to 21 years in captivity. TABLE 1-1 Selected Families of Anurans In a strict sense, the term “toads” represent frogs belonging to the family Bufonidae. In a larger sense, “toad” is used for any terrestrial frog having “warty”—dry skin and parotid glands—voluminous glandular masses behind the eyes. Other frogs have smooth, moist skin without warts and (most of the time) lack parotid glands. The terms “frog” and “toad” are not clear. For instance, the European Fire-bellied Toad (Bombina bombina) is a warty, semi-aquatic “toad” with no parotids behind the eyes. Anurans are the best represented in zoos, compared with other amphibians. Some, such as Xenopus laevis and Silurana tropicalis, have been model species for research for many years. With the Amphibian Crisis, publicized by the EAZA in 2008 with the “Year of Amphibians,” wild anurans are now the focus of major global ecologic concerns, including pollution, climate changes, habitat destruction, and nonnative species translocation. Campaigns all around the world (for example, the Amphibian ARK) of awareness and information on amphibians, have led to a huge amount of information available online on the husbandry of many anuran species. Consequently, veterinarians are consulted more frequently for information on health and disease. All adult anurans are without a tail (the “tail” of tailed frogs [Ascaphus sp.] is, in fact, an extension of the male cloacae, used as a copulatory organ). Highly specialized in the hopping mode of locomotion, their long hind legs have given rise to their alternative name salientias (jumpers). However, considerable specialization exists in this regard. Some arboreal frogs may move by quadrapedal walking or climbing. Burrowing frogs dig head first with hind legs adapted for excavation. Eyes are voluminous; vision plays a great role in nutritional behavior. Prey movement triggers the feeding response. A nictitating membrane is present. Posterior to each eye, the circular tympanic membrane represents the ear externally. A large tongue is attached anteriorly and is folded back into the oral cavity such that its distal, bifid end lies posteriorly. The tongue is extended to catch insects. A single row of small teeth lies around the margin of the upper jaw. The coelomic cavity is not divided. The intestinal tract is relatively short and follows the normal vertebrate plan. The liver serves as an important erythropoietic center and plays an important role in immune function, the synthesis of nitrogenous compounds, antioxidation reactions, and the metabolism of various endogenous and exogenous substances. The gall bladder is intimately associated with the liver, with a bile duct connecting it to the duodenum. In some species, it joins the pancreatic duct before it enters the intestinal tract. The cloaca is present posteriorly. However, due to the absence of a tail, it appears to be located somewhat dorsally. Anurans are ectothermic and environmental temperature may really modulate their life history, influencing body temperature, evaporative water loss, digestion, and oxygen uptake, as well as the velocity of muscle contraction, locomotion, and vocalization. Anurans will compensate daily thermal fluctuation by modifying their behavior and metabolic changes, for instance, by oriented aerobic depression of several organs.9 Therefore, it is important to keep the animals within the preferred optimal temperature zone (POTZ). Some species (mostly temperate) hibernate and estivate. Anurans that hibernate in colder climatic conditions accumulate more energy before winter and even after emerging and before breeding. Fats are the preferred substrates of aerobic metabolism if oxygen is not limiting, and are the main source of at least 80% of the energy used during hibernation.9 The skin not only has a protective and sensory capacities but also plays critical roles in thermoregulation, fluid balance, respiration, transport of essential ions, respiration, and sex recognition. The cutaneous gland (in the dermis) secretions may be irritating, toxic, and even potentially lethal, like the steroid alkaloid toxins of the poison frogs (Dendrobates and Phyllobates). One of the natural defenses of the skin is production of antimicrobial peptides in granular glands.14 Discharge of the granular glands is initiated by the stimulation of sympathetic nerves. Antimicrobial peptides produced in the skin are an important defense against skin pathogens and may affect survival of populations. The skin has low resistance for water evaporation, and most anurans are vulnerable to rapid water loss. In terrestrial species, mucous or waxy substances are produced by a variety of glands to reduce evaporative water loss. All anurans may absorb water through the ventral pelvic skin and also reabsorb water in the kidney and from the urinary bladder. Amphibian lymph consists of all the components of blood, with the exception of erythrocytes. In anurans, the lymphatic system is highly developed and has a major role in fluid exchange and blood volume regulation. It is composed of pulsatile lymph hearts (that beat independently of the heart), an elaborate series of lymph vessels, and subcutaneous lymph sacs. Lymph flow is unidirectional; one-way valves are present between the sacs. Lymph heart failure should be in the differentials for subcutaneous and coelomic cavitary accumulations of fluid. In anurans, the primary nitrogenous waste may be ammonia, urea, or uric acid. Aquatic species excrete a higher concentration of ammonia, whereas many terrestrial anurans have evolved metabolic adaptations to excrete urea and even uric acid. Dehydrated animals will decrease their glomerular filtration rate, thereby accumulating ammonia in body tissues, which may lead to azotemia. Anurans seem to be quite resistant to high plasma urea levels. Urea is less toxic than ammonia and may be stored in body tissues until water may be replenished. However, the limit after which toxic effects appear is not clear. Still, urea will be excreted rapidly on rehydration. Larval stages maintain gills for respiration, whereas adults primarily respire via the lungs and the buccopharyngeal cavity. In addition, all anurans show some degree of cutaneous oxygen respiration.12 The heart has three chambers, with two atria and one ventricle. All anurans show a complete interatrial septum, limiting the mixing of oxygenated and unoxygenated blood that still occurs in the single ventricle. The heart is seen contracting on the midline just caudal to the animal’s shoulders. A renal portal venous blood system exists. Housing requirements for anurans will definitively depend on their specific needs and their natural habitat. However, several key points should always be monitored. Providing an appropriate temperature gradient and a mosaic of thermal zones allows these animals to self-regulate their body temperature (heat lamps may desiccate the animals and should be avoided). Unlike reptiles, most sick amphibians recuperate better in a cooler rather than a warmer environment. Anurans that are kept above or below their POTZ may show signs of inappetence, weight loss, agitation changes in skin color, immunosuppression, and bacterial overgrowth. Monitoring hygrometry prevents evaporative water loss. Tadpoles and aquatic species need dechlorinated water. Water quality parameters (ammonia, pH, and chlorine) should be routinely evaluated with home aquarium test kits. Poor water circulations, overcrowding, or both commonly lead to water quality problems. Waste material and uneaten foods should be removed. Dilute chlorine bleach is a simple and good general disinfectant. Review of husbandry and zoological records is part of the diagnostic process. Most diseases come from a lack of understanding of specific management requirements. In general, natural feeding is opportunistic. Although most tadpoles are herbivorous or omnivorous, all adult anurans are carnivorous, consuming a wide variety of live invertebrates and also mice, rat pups, fish, or any small vertebrates for the large ones. Terrestrial anurans only target moving prey. Many aquatic amphibians are more likely to target food by scent and may consume inert food. Most anurans are voracious feeders and tend to eat anything that fits into their mouth. Gastric overload and impaction, as well as ingestion of non-food items are fairly common. Frequency of feeding depends on the primary energy and nutritional requirements of the species, their seasonal activity, and breeding cycle. For energetic species such as Dendrobates sp., insects should remain in the enclosure between feedings so that the animals are fed ad libitum. In this case, having an insect farm is essential. In more sedentary species, which are prone to obesity, feeding rates should be adjusted accordingly. For instance, an adult Ceratophrys sp. is fed every 10 to 15 days (with various insects, neonatal or suckling mice, and dead adult mice [to prevent bites]). Digestion, assimilation, and metabolic rates generally increase with increased temperature, and feeding increases to a peak and then declines as temperatures become too high. Amphibians cannot synthesize carotinoids, including vitamin A.23 Nutritional disorders caused by unbalanced vitamin A supplementation have been observed.15,23 Analysis of longstanding husbandry practices showed that ultraviolet B (UVB) exposure and dietary calcium-to-phosphorus ratio were deficient compared with wild conditions—likely causing chronic underlying metabolic bone disease.2,21 The key point for feeding anurans in captivity is to provide few invertebrates of different species and of different sizes. In our zoo, we have our own insect and rodent farms: grasshoppers, crickets, locusts, mealworms, and mice. In our opinion, it would be hazardous to supplement the diet of the frogs and toads directly. It is better to prevent single-item food sources and give a balanced diet to the prey. A huge amount on feeding information of many species may be found in the hobbyist and professional literature.23 Most anurans are docile; however, large ones may bite. Once a Ceratophrys sp. has bitten, it never opens the mouth (to prevent the prey from escaping!). Smooth nets, plastic bags, or gloves may be used for moving animals. Wrapping with wet paper towel is a good technique to restrain an animal for a quick examination or for medication administration. Wearing moistened, powder-free gloves prevents the transfer of microorganisms from the hands of the clinician and also provides protection against secreted toxins. In all cases, manual restraint is used for short and nonpainful procedures. For a longer clinical examination, it is better to place the animal in a transparent container only used for this purpose. General anesthesia may be required for biopsies, blood sampling, and surgical procedures such as gastrotomy and laparoscopic or exploratory surgery. Surgery and other invasive procedures are often perceived as painful. Thus, anurans should always be given analgesic drugs. Analgesia potentiates the effects of anesthetic drugs and reduces recovery time.10 We have been using meloxicam empirically for several years at a dosage of 0.2 milligrams per kilogram (mg/kg). A recent study recommends systemic administration of meloxicam at a dosage of 0.1 mg/kg once daily.17 In general, anurans do not require fasting prior to anesthesia. Their larynx remains tightly closed even under general anesthesia, and the chance of aspiration is very low. However, it is better not to feed large frogs and toads 24 to 48 hours before anesthesia. The righting reflex is used as a primary indicator to determine the stage of anesthesia. Loss of this reflex suggests a light stage of anesthesia. A surgical plane is indicated by the loss of the withdrawal reflex. Anesthetized amphibians usually become apneic; abdominal and gular respirations may cease. Heart rate is a useful tool for anesthesia monitoring. Putting the frog in dorsal recumbency may help perform direct visualization. In our opinion, electrocardiography (ECG) leads are traumatic, and the use of alcohol may be deleterious. The drug of choice for sedation or anesthesia is tricaine methanesulfonate (MS-222), which has also demonstrated analgesic potential (Figure 1-1). Table 1-2 presents only those protocols that have been used and evaluated by us. More protocols may be found elsewhere.6 Aquatic animals should have their head out of water during recovery. TABLE 1-2 Protocols for Anesthesia and Analgesia in Anurans g/L, Gram per liter; IM, intramuscular; mg/kg, milligram per kilogram; mL/g, milliliter per gram; mL/L, milliliter per liter. Amphibians are generally good candidates for surgery. They are quite resistant to blood loss. Biopsies and skin surgery follow the same techniques used in other vertebrates. For biopsies, only a small surface of the skin may be taken, as it is not very extensible. When the surgery is too extensive (neoplasia or abscess), we may perform chemical cauterization with metacresolsulfonic acid and formaldehyde 36% (Lotagen TM, Schering-Plough Animal Health). Lotagen has an astringent action on healthy mucous membrane and promotes granulation and epithelialization. We frequently use these compounds with good results. Coelomic exploratory and gastrointestinal surgeries are common procedures. Presurgical preparations include hydration of the animal in a shallow water bath and prophylactic antimicrobial therapy (either by bath or injection). Preparation of the skin is accomplished by gently tapping the skin with cotton-tipped applicators and povidone-iodine solution diluted with sterile saline (
Anurans
Biology
Family
Number of Species, Representative Species
Geographic Location and Comments
Bombinatoridae
10 species
Bombina frogs (Bombina sp.)
Eurasia
Specialized glands in their skin secrete a toxin, which may cause irritation; often display the unken reflex when disturbed
Pipidae
33 species
Clawed frogs (Xenopus sp., Silurana sp.)
Surinam toads (Pipa sp.)
South America (genus Pipa) and sub-Saharan Africa (four other genera)
Tongueless frogs, lack vocal cords, exclusively aquatic, and often found in animal facilities
Hemiphractidae
100 species
Marsupial frogs (Gastrotheca sp., Flectonotus sp.)
Central and South Americas
Marsupial female frogs possess a dorsal pouch, where fertilized eggs are kept; Gastrotheca riobambae is well represented in zoos
Bufonidae
571 species
True toads (Bufo sp.)
Common toad (Bufo bufo)
Widespread on every continent except Australia and Antarctica
Terrestrial, toothless, dry warty skin, a pair of parotid glands; all males have the Bidder organ (potentially active ovary)
Dendrobatidae
177 species
Poison dart frogs (Dendrobates sp., Phyllobates sp.)
Nicaragua to the Amazon Basin of Bolivia and to southeastern Brazil
Small, very colorful frogs, famous for their toxic skin production; very popular with hobbyists and zoos; Phyllobates terribilis secrete one of the most dangerous venoms in the world
Hylidae
926 species
Known as “tree frogs” (Hyla sp., Litoria sp., Phyllomedusa sp., Agalychnis sp.), even though some hylids are terrestrial or semi-aquatic
America, Eurasia to Australo-Papuan region; extreme northern Africa
Most are arboreal and have forward-facing eyes and have adhesive pads at the extremity of each finger; monkey frogs (Phyllomedusa bicolor) have parotid glands and were the origin of the prototypical antimicrobial peptide family, the dermaseptins; white tree frog (Litoria caerulea) is a very popular pet frog
Ranidae
“True frogs”
355 species
Common frog (Rana sp.)
Worldwide except Antarctica
Ranids species are commonly pet frogs; many wild populations are subjects of research work; also used in research facilities
Leptodactylidae
189 species
Argentine horned frog (Ceratophrys ornata), Smokey Jungle frog (Leptodactylus pentadactylus)
Southern United States, Mexico, northern Antilles, south to Brazil
Argentine horned frog, also called “Pacman frogs,” and Smokey Jungle Frog are very popular pets
Microhylidae
519 species
Tomato frogs (Dyscophus sp.)
North and South America, sub-Saharan Africa, India to northern Australia
Very colorful species of frogs; popular pets; Madagascar tomato frog (D. antongilii) endangered as a result of deforestation and overcollecting for the pet trade
Anatomy and Physiology
Special Housing Requirements
Feeding
Restraint, Anesthesia, and Analgesia
Drug
Dosage and Route
Comments
Tricaine methanesulfonate
(MS-222)
Tadpoles and aquatic frogs
0.25 to 0.5 g/L (Bath)
Adult frogs and toads
1 g/L (Bath)
(see Figure 1-1)
Buffer 2 g of MS-222 with 40 mL of Na2HPO4 (0.5 mL/L). Induction times are variable. After induction, place the frog into a shallow amount of nonanesthetic water or on a wet towel. Recovery is generally achieved 30 to 90 minutes after the anesthetic is removed.
Isoflurane
5% in oxygen (inhalation or bubbling in bath) (see Figure 1-1)
Gentle stimulation encourages continued respiration. Effective but slow induction.
2 to 3 mL/L (Bath)
Isoflurane is sprayed directly into the water.
0.01 to 0.06 mL/g (topical)
Apply directly OR dilute 3 mL of liquid isoflurane in 1.5 mL of water, and 3.5 mL of K-Y Jelly. Shake until a uniform gel. This gel is applied at dosages of 0.025 to 0.035 mL/g.
Lidocaine / Prilocaine
0.5 mL /150 g (topical)
Short induction for 30 minutes of surgical plane.
Medetomidin (M) / Ketamin (K) / Meloxicam (Mel) / Butorphanol (But)
M 0.5 mg/kg
+ K 50 mg/kg
+ Mel 0.2 mg/kg
+ But 25 mg/kg (IM)
Effective protocol in Xenopus laevis for heart surgery (n = 12, unpublished data).
Reversed with atipamezole hydrochloride at equal volume to medetomidine IM.
Butorphanol
25 mg/kg (IM)
Unpublished data.
Meloxicam
0.1 to 0.2 mg/kg (IM or ICe)
Surgery
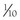 ). Incisions should be made with one bold, clean stroke. When performing a laparotomy, the surgeon must take care of macroscopic glands, lymph hearts, and blood vessels, especially the midventral vein. The abdominal membrane is punctured and dissected smoothly. Everting-type suture patterns, with simple interrupted sutures using nonabsorbable material, are recommended for skin closure (Figure 1-2). Surgical tissue glues may also be used in conjunction with sutures for skin closure. Insufflation is needed in laparoscopy to improve the visibility of all organs. Management of cloacal prolapse sometimes requires surgery (Table 1-3). Veterinarians are sometime asked to surgically withdraw eggs for research purposes (Figure 1-3). Postoperative infections are rare in healthy animals.24
). Incisions should be made with one bold, clean stroke. When performing a laparotomy, the surgeon must take care of macroscopic glands, lymph hearts, and blood vessels, especially the midventral vein. The abdominal membrane is punctured and dissected smoothly. Everting-type suture patterns, with simple interrupted sutures using nonabsorbable material, are recommended for skin closure (Figure 1-2). Surgical tissue glues may also be used in conjunction with sutures for skin closure. Insufflation is needed in laparoscopy to improve the visibility of all organs. Management of cloacal prolapse sometimes requires surgery (Table 1-3). Veterinarians are sometime asked to surgically withdraw eggs for research purposes (Figure 1-3). Postoperative infections are rare in healthy animals.24
Anurans
Chapter 1