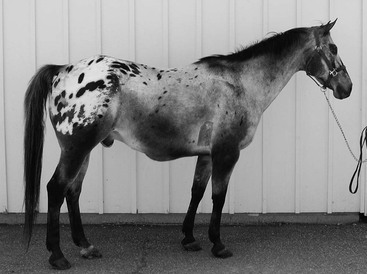
Virginia B. Reef, Consulting Editor Joann Slack Sheila M. McGuirk Edema is an abnormal accumulation of extracellular fluid in the interstitial spaces of the tissues or in body cavities that can be generalized or localized. If the fluid accumulation occurs in the pleural cavity, it is referred to as pleural effusion or hydrothorax; if the fluid accumulation is in the abdominal cavity, it is referred to as peritoneal effusion or ascites. Fluid accumulates more easily in those parts of the body where the connective tissue structure is relatively loose. The accumulated fluid tends to gravitate to the dependent areas of the body. In the cow, generalized edema is detected externally by swelling of the submandibular tissue, the brisket, the ventral abdomen, and occasionally the limbs (Fig. 6-1). External manifestation of generalized edema in the horse is frequently in the pectoral region between the frontlimbs, along the ventral abdomen, in the prepuce in stallions and geldings (Fig. 6-2), in the limbs, and sometimes in the head. Stocking up, or limb edema restricted to the lower limbs, is commonly detected in stabled horses with no underlying disease. Large amounts of fluid may accumulate before clinical signs become evident. External evidence of pulmonary edema (i.e., a frothy, possibly blood-tinged fluid in the nares or expectorated) is rarely detected in large animals (Fig. 6-3). There are numerous causes of edema, including congestive heart failure (CHF) (Boxes 6-1 and 6-2). Edema is a late sign of CHF; other subtle signs of failure may be present before edema appears. Edema is caused by an alteration in the equilibrium between capillary permeability and the forces that govern fluid movements at the capillary level. These forces are as follows: Activation of complement and liberation of cytotoxic agents such as oxygen radicals, leukotrienes, hydrogen peroxide, platelet-activating factor, and lysosomal enzymes contribute to the endothelial and epithelial damage, causing permeability edema. Subsequent increase in colloid osmotic pressure causes fluid accumulation in the interstitial space. The most common causes of increased capillary permeability are trauma, infection, endotoxemia, and hypersensitivity (allergic) vasculitis. Topical administration of counterirritants can also cause local increase in capillary permeability. Equine purpura hemorrhagica, the most common vasculitic disease in horses, may in its mildest form have symptoms of mucosal petechiae and plaques of edema or, in severe cases, serum exudation from and necrosis of skin surfaces. Increased hydrostatic pressure can cause either localized or generalized edema. In horses and ruminants the most common causes of increased hydrostatic pressure are CHF, venous thrombosis, liver disease causing obstruction of the portal venous system, lymphadenopathy, a cranial mediastinal mass, compression bandages, limb immobilization, and topical administration of counterirritants. CHF occurs when there is concomitant pulmonary and systemic vascular congestion. The compensatory salt and water retention increases ventricular diastolic, venous, and capillary pressures, which can result in the formation of generalized edema. Arteriolar vasodilation, caused by release of tissue mediators of inflammation or increased venous pressure resulting from obstruction to venous outflow, can also elevate capillary hydrostatic pressure and result in edema formation. When the plasma protein concentration decreases from normal to values less than 5 g/dL or albumin concentration is less than 1.5 g/dL, generalized edema may occur. Hypoproteinemia can result from (1) decreased production of plasma proteins with starvation, liver disease, or severe heart failure, or (2) augmented loss of plasma proteins resulting from kidney disease, protein-losing enteropathies (Johne’s disease, chronic inflammatory bowel disease), peritonitis, or pleuritis. Hemodilution as a result of overzealous administration of fluids or decreased elimination of fluid can cause edema. Failure to excrete adequate water to maintain fluid balance can result from decreased glomerular filtration as a result of kidney disease or heart failure. Increased tissue colloid osmotic pressure is rarely a cause of edema in horses and ruminants. Interstitial fluid has a lower plasma protein concentration than plasma. When capillary permeability is increased or when abnormal protein-like material is present in the interstitial space, edema can develop by this mechanism. The latter may occur with infection or after administration of topical counterirritants. Lymphedema occurs when lymphatics are absent or obstructed. Congenital absence of lymphatics is extremely rare. Obstruction to lymphatic drainage can be caused by tumor, local inflammation (lymphangitis or lymphadenitis), or elevated central venous pressure as in heart failure. 3. Obtain whole blood for the following: a. Complete blood count (CBC) (includes fibrinogen and plasma protein concentration) b. Selenium concentrations if cardiomyopathy is suspected 4. Test serum or plasma for the following: a. Total protein and albumin concentrations; measure or calculate globulin fraction b. Tests for kidney function as follows: serum urea nitrogen (SUN) and creatinine concentrations, serum electrolyte (Ca, P, Na, K, Cl) concentrations, fractional excretion of electrolytes (see Chapter 34 for method), complete urinalysis; serum concentrations of Na may be decreased, and SUN may also be elevated in chronic heart failure h. Vitamin E levels if cardiomyopathy suspected 6. Analyze fluid to rule out peritonitis, pleuritis, and pericarditis; if physical examination and CBC findings are compatible, examine: Cardiac arrhythmias are abnormalities in the normal heart rate, rhythm, or conduction pattern. Arrhythmias result from abnormalities of impulse generation or impulse conduction or a combination of both. In the normal heart the impulse is generated in the sinus node because it has the highest rate of spontaneous depolarization. Atrial contraction is followed shortly by ventricular contraction. There is variability in reported normal ranges for heart rate in the large adult animal species, but there is general acceptance of the following ranges: Arrhythmias are more common in horses than in other domestic animal species. Up to 25% of horses that have no other signs of heart disease have cardiac arrhythmias during routine examination or electrocardiography.1 During continuous 24-hour electrocardiography, 44% of normal horses had second-degree atrioventricular (AV) block, 10% had sinus arrhythmia, 3% had sinoatrial (SA) block, 27% had occasional supraventricular extrasystoles, and 15% had occasional ventricular arrhythmias.2 Cardiac arrhythmias may be present in 40% of horses that have other signs of cardiac disease.1 Unlike other species, the horse has arrhythmias at rest that are considered benign or functional. Benign, physiologic, or functional arrhythmias are usually bradyarrhythmias and are thought to be the result of increased vagal tone. These arrhythmias disappear at high heart rates (exercise or excitement) or with the administration of atropine (0.02 to 0.05 mg/kg subcutaneously [SC] or intramuscularly [IM]) or glycopyrrolate (0.003 to 0.006 mg/kg SC or IM). Some examples of benign or functional arrhythmias are as follows: Other arrhythmias are usually considered to be pathologic, even if there are no other overt signs of cardiac disease. Some examples of pathologic arrhythmias are: • Frequent atrial and ventricular premature depolarizations • Supraventricular or ventricular tachycardia • Advanced second-degree or third-degree (complete) AV block The most effective method of identifying the specific arrhythmia is by performing an electrocardiogram (ECG). Arrhythmias that are transient or intermittent may not be detected with resting electrocardiography. Radiotelemetry or continuous 24-hour ECG recordings are useful to characterize the type, frequency, and severity of arrhythmias. Exercising electrocardiography may identify arrhythmias that are absent or clinically insignificant at rest but that may impair performance. In general, cattle do not have benign arrhythmias like horses, but they are frequently found to have sinus bradycardia and sinus arrhythmia associated with lack of feed intake. These arrhythmias were previously thought to be abnormal and associated with vagal indigestion but have been shown to occur in normal cattle held off feed for 12 to 48 hours.3 Cattle with gastrointestinal disease seem to have increased susceptibility to cardiac arrhythmias, especially atrial premature depolarizations and fibrillation. Although the reason for the susceptibility is not established, abnormal electrolyte concentrations, acid-base disturbances, and aberrations in autonomic nervous system balance have been proposed.4,5 Sinus arrhythmia in goats is considered to be a benign arrhythmia and is present in many normal animals. Normal camelids also frequently have sinus arrhythmia. Arrhythmias result from abnormalities of impulse generation or impulse conduction or a combination of both. A variety of mechanisms can cause abnormal impulse generation or conduction (Boxes 6-3 and 6-4). Abnormal impulse generation occurs because of localized changes in ionic currents that flow across the membranes of single cells or groups of cells. Abnormal impulse generation can be seen as automaticity (normal and abnormal) or triggered activity. Automaticity, the ability to initiate action potentials spontaneously, is a property of cells in the sinus node, some parts of the atria, the AV junction, and the His-Purkinje system. Cardiac disease can be responsible for the development of automaticity in cells that normally do not have this property. Normal automaticity develops when the membrane potential slowly falls (i.e., becomes less negative) during diastole. When the membrane reaches its threshold potential, an impulse is initiated. The most common clinical arrhythmias that are thought to be caused by the automaticity mechanism are sinus tachycardia and sinus bradycardia, which are the result of alterations in autonomic nervous system tone. Enhanced automaticity in another area of the heart that is capable of automaticity (spontaneous depolarization) may be responsible for atrial or ventricular premature beats. It is not clear what clinical arrhythmias are caused by triggered activity. Under certain circumstances, conduction abnormalities allow a propagating impulse, which has already excited the heart, to persist and reexcite the atria or ventricles after the end of the refractory period. This can occur in an ordered or random fashion. Random reentry occurs over reentrant pathways that continuously change in size and location with time, whereas ordered reentry occurs over a relatively fixed reentrant pathway. Impulse propagation may be slow enough that reentrant circuits can be established in small areas of myocardium. In large animals the size of the myocardial circuit is large enough that relatively mild alterations in impulse propagation may make reentry feasible and may account for the relatively greater frequency of arrhythmias in these species. Although it is not possible to precisely define the mechanism of clinical arrhythmias, it is believed that atrial and ventricular fibrillation may be caused by random reentry. Under clinical conditions, cardiac arrhythmias may be associated with disturbances in electrolyte concentrations, especially potassium and calcium; in acid-base balance; and in autonomic nervous system balance. These conditions can precipitate cellular changes conducive to the development of arrhythmias by any of the previously mentioned mechanisms. It is important to distinguish between abnormal arrhythmias that are primary and those that are secondary. Most abnormal arrhythmias of horses and cattle are tachyarrhythmias. Primary arrhythmias are caused by pathologic conditions of the heart (myocarditis, valvular disease, conduction system abnormalities, and pericarditis). Secondary arrhythmias develop in the absence of heart disease and can be caused by excitement, fever, sepsis, hypoxemia, metabolic or electrolyte imbalances, gastrointestinal disturbances, anesthesia, ionophores, other drugs, or toxemia. The treatment and prognosis for the two types of arrhythmias can be very different, and examination and laboratory tests are used to assist in making the distinction. 2. Perform a physical examination to determine whether there is primary cardiac disease. Record the animal’s vital signs. Determine whether this is a bradyarrhythmia or tachyarrhythmia. Careful auscultation should note which heart sounds are present and should characterize the arrhythmia. There may be irregularities in the basic rhythm, added sounds, or long pauses; classification of heart rate by regularity of rhythm can distinguish one arrhythmia from another (see Chapter 30). Note whether a pulse deficit is present by simultaneous auscultation and palpation of the peripheral arterial pulse; note the strength of the peripheral arterial pulse; observe the jugular vein for pulsations and distention; examine peripheral veins for distention; and examine mucous membrane color and capillary refill time. Careful auscultation of the lungs with and without a rebreathing bag should be performed. 3. Record an ECG. The base-apex lead can be used to screen for arrhythmias; it is attached using positive, negative, and ground leads as follows: b. Negative lead is attached to the skin of the right jugular furrow two thirds of the distance from the ramus of the mandible to the thoracic inlet; using lead I or lead II, this is the right arm; using lead III, it is the left arm. ECG interpretation is discussed in Chapter 30. 5. Obtain feed for analysis if ionophore (monensin, lasalocid, salinomycin) exposure is suspected. 6. Obtain blood for the following: a. CBC b. Selenium concentrations if cardiomyopathy suspected c. Blood gas determinations and acid-base status 7. Test serum or plasma for the following: a. Electrolyte (Na, K, Cl, Ca, Mg, P) concentrations b. Vitamin E (α-tocopherol) concentration c. Cardiac troponin-I (cTnI)6 if myocardial inflammation, injury, or necrosis is suspected 8. Test urine for the following: a. Electrolyte (Na, K, Cl) concentrations b. There is ventricular tachycardia with a rapid rate (heart rate >120 beats/min) c. There are multifocal ventricular ectopic beats d. A QRS is detected in the preceding T wave (R on T) e. There is advanced second-degree or complete (third-degree) AV block
Alterations in Cardiovascular and Hemolymphatic Systems

Peripheral Edema, Pleural Effusion, and Ascites
Mechanisms of Edema
Approach to Diagnosis of Peripheral Edema, Pleural Effusion, and Ascites
Cardiac Arrhythmias
Mechanisms of Cardiac Arrhythmias
Approach to Diagnosis of Cardiac Arrhythmias
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Alterations in Cardiovascular and Hemolymphatic Systems
Chapter 6