Eyelid tumours
Canine
Most commonly benign, eyelid tumours are usually seen in older dogs. Complete excision with narrow margins is curative for sebaceous gland adenoma/Meibomian gland tumour, benign melanoma and squamous papilloma. Papillomas tend to be well circumscribed, superficial with minimal invasion of deeper tissues, and commonly resolve spontaneously. Histiocytomas occur in young to middle-aged dogs, and in young dogs often resolve spontaneously. Roberts et al (1986) reported that 88% of eyelid neoplasms in dogs were sebaceous tarsal gland adenomas, benign melanomas or papillomas, and that malignant tumours (melanoma, adenocarcinoma, basal cell carcinoma, mast cell tumour, squamous cell carcinoma, haemangiosarcoma and myoblastoma) comprised only 8.2%. Other malignant eyelid tumours in dogs include lymphoma (LSA) and fibrosarcoma (FSA).
Feline
Eyelid tumours in cats are less common than in the dog but are more often malignant (Aquino 2007). Most commonly they tend to be squamous cell carcinoma (SCC), mast cell tumour (MCT) or LSA (see relevant chapters). Other tumour types occur rarely.
Treatment
Surgery
For malignant tumours of the eyelid, it is more important to achieve wide clean margins than to achieve perfect eyelid reconstruction. However, the eyelid must be able to function so as to maintain ocular health. Sutures should be buried so as not to irritate the cornea. Enucleation of the eye or exenteration of the orbit may be necessary to allow a curative resection. The usual principles of oncology should be adhered to (e.g. staging prior to surgery, removal of enlarged regional lymph nodes at surgery for staging purposes and assessment of inked surgical margins to ascertain histological margin).
Techniques to reconstruct the eyelid have been reported in both dogs and cats. Lesions involving one-quarter to one-third of the eyelid margin can be reconstructed with a simple V-plasty (wedge excision). Electrosurgery is not recommended but CO2 laser works well (Bussieres et al 2005). A lip-to-lid flap has been used successfully to replace lower eyelid defects in cats (Hunt 2006). Split eyelid flaps can also be used in dogs and cats (Hagard 2005, Lewin 2003). A cross-lid flap method has also been described (Munger & Gourley 1981). Other techniques, such as reverse triangles, H-plasties and rotation flaps, are also reported (Stades 1987), along with use of the third eyelid as part of eyelid reconstruction with transposition flaps. Free hard palate mucosal grafts have also been used with success in dogs to replace eyelid deficits (Zhao & Li 1996).
Hoffman et al (2005) reported six cats (from a database of 3997 feline ocular histopathology submissions) with a histological diagnosis of peripheral nerve sheath tumour arising from the eyelid or conjunctiva. All cats treated with surgical excision, cryoablation or laser ablation had an average of three recurrences of the tumour, whereas two-thirds of those treated with wide excision had no recurrence (Hoffman et al 2005).
SCC of the eyelid is common in cats with repeated exposure to UV light and poorly pigmented skin (similar to SCC of the nasal planum and pinnae in cats, see Chapter 18) (Figure 25.1). Early stages may be confused with chronic conjunctivitis. Surgical resection with adequate margins is often curative as there is a low rate of metastasis (10–15%).
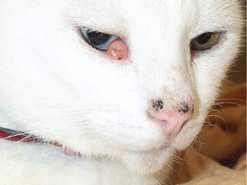 |
| Figure 25.1 (Courtesy R Straw.) |
Radiotherapy
Radiotherapy may be used for tumours with known radiation responsiveness, such as LSA and SCC. However, LSA is often treated with chemotherapy and larger SCCs are better treated with surgery than radiotherapy. Side effects of radiation include erythema, alopecia, leukotrichia, keratoconjunctivitis sicca, keratopathy, uveitis and cataract.
Chemotherapy
Rarely used for eyelid neoplasms in dogs and cats, chemotherapy may be indicated for LSA or MCT of the eyelid (see Chapter 19 and Chapter 22).
Third eyelid and conjunctival tumours
The differential diagnosis in cats and dogs includes SCC, papilloma, haemangioma, haemangiosarcoma (HSA), adenoma, adenocarcinoma, malignant melanoma (MM), MCT, LSA, and FSA. A pseudoneoplastic hypertrophy of the gland of the third eyelid may also occur. Prolapse of the gland of the third eyelid (‘cherry eye’) can usually be differentiated from neoplasia, because neoplasia (adenocarcinoma of the gland of the third eyelid) occurs primarily in older dogs (10–16 years). Fibrous histiocytomas (nodular granulomatous episcleritis) can also occur on the third eyelid.
Canine
Solar radiation appears to be a predisposing factor for the development of SCC (Hargis et al 1978). In humans, conjunctival scrapings are useful for in-house diagnosis (Spinak & Friedman 1977). This may also be the case for animals.
• Papillomas are benign and excisional biopsy is advisable (Bonney et al 1980).
• Adenocarcinomas of the gland of the third eyelid can be invasive. Metastasis (to the regional lymph nodes and orbit) is not a common feature, but is known to occur. They generally appear as a smooth, pink nodule on the bulbar aspect of the third eyelid. They have a good prognosis with early, adequate treatment, but recurrence is expected with incomplete excision. In order to give the best chance of complete excision, the entire third eyelid should be removed, and the eye should be enucleated if involved (Wilcock & Peiffer 1988).
• Malignant melanoma of the conjunctiva in dogs can be aggressive, with local recurrence after excision reported in 6/12 cases and metastasis in 2/12 cases (Collins et al 1993). Local treatment should be aggressive, with wide surgical excision including orbital exenteration if necessary. A combination of wide surgical excision and cryosurgery can be used (Collins et al 1991). The prognosis in dogs is guarded. As in the cat, histological features (mitotic index, cell type, degree of pigmentation) are not good predictors of behavioural malignancy (Collins et al 1993).
• Mast cell tumour: mucocutaneous sites are generally thought to be a poor prognostic location for MCTs (Chapter 19). However, there is little reported information on the behaviour of conjunctival MCTs. One report of a dog with a rapidly progressive grade II conjunctival MCT had excisional biopsy (close margins) and adjuvant treatment with prednisolone and vinblastine, with no evidence of local recurrence or metastasis during a 29-month follow-up period (Barsotti et al 2007). Two other reported canine cases of subconjunctival MCT had a relatively benign course (Johnson et al 1988) (Figure 25.2).
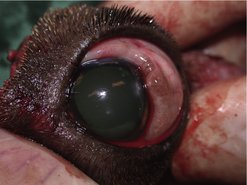 |
| Figure 25.2 |
• Conjunctival LSA in dogs and cats may be misdiagnosed as conjunctivitis, as it can present as a diffusely swollen and firm conjunctiva (usually unilateral) (Figure 25.3). For diffuse conjunctival swelling, an incisional biopsy under topical anaesthesia would be an appropriate diagnostic step (the author (TB) does not use sutures after an incisional biopsy, allowing healing by second intention). A conjunctival LSA in a dog (intermediate-grade, diffuse, large-cell lymphoma with T-cell immunophenotype) was treated with excisional biopsy. No adjuvant chemotherapy was given and there was no recurrence after 12 months.
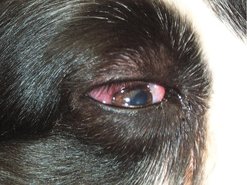 |
| Figure 25.3 |
• Haemangioma and HSA: Pirie et al (2006) evaluated 70 dogs with haemangiomas and 38 dogs with HSA. The average age was 8.6 years. Dogs with increased outdoor activity were predisposed. Most tumours occurred within non-pigmented epithelium along the leading edge of the nictitating membrane (41/108) and temporal bulbar conjunctiva (33/108). Ultraviolet light exposure was likely to be a significant risk factor. Early surgical resection with clean margins was thought to be curative. Local recurrence was more likely with HSA (11/20). In one case report (Liapis & Genovese 2004) of a dog with HSA of the third eyelid, surgical excision with no other treatment showed no recurrence after 9 months.
Feline
• Malignant melanoma: in cats, conjunctival melanoma is usually malignant, with a high rate of early metastasis and a poor prognosis (Patnaik & Mooney 1988). An MM originating from the surface of the nictitating membrane reported in a 10-year-old cat infiltrated the orbital cavity causing exophthalmia, with metastasis to the cerebrum and the lungs (Roels & Ducatelle 1998). However, not all cases with histological features of malignancy have a poor prognosis. One case report (Cook et al 1985) reported complete excision of conjunctival MM without further evidence of local recurrence or metastasis during an 11-month follow-up period.
• Conjunctival LSA: see above comments for dogs. In cats, it is important to check feline leukaemia virus (FeLV) status, as many young cats with LSA are FeLV positive. There is a reported case of a cat with a conjunctival LSA with peripheral lymph node enlargement with Hodgkin’s-like LSA (Holt et al 2006). The conjunctival swelling responded poorly to standard chemotherapeutic protocols for LSA, but responded well to radiation therapy, and had not recurred 3 years after treatment.
• Haemangioma and HSA: feline conjunctival haemangiomas and HSA were evaluated by Pirie & Dubielzig (2006). In all they looked at eight cases seen over a period of 9 years. Third eyelid location was the most common. In seven of eight cases, non-pigmented tissue was affected, and most had a history of high annual UV light exposure. Surgical excision with clean margins appeared to be curative. One case report of HSA of the third eyelid in a cat treated with surgery alone, had no recurrence after 7 months (Multari et al 2002).
Diagnostic work-up and staging
For suspected malignant tumours, fine needle aspirate (FNA) cytology or incisional biopsy is diagnostic. Ultrasound of the eye and periorbital tissues, CT or MRI is indicated if invasion is suspected. Thoracic radiographs (not generally needed for MCT) and abdominal ultrasound may be indicated in specific cases.
If staging is negative, a resection with curative intent is indicated. This includes removal of all the third eyelid for malignant tumours of the third eyelid, and exenteration of the orbit for malignant tumours that have invaded into the orbit. This deficit can be reconstructed with a caudal auricular axial pattern flap (Stiles et al 2003). Another paper has described wound reconstruction after radical orbital exenteration and tumour resection in six cases (Koch et al 1994). Excisional biopsy can be an appropriate first-line treatment for suspected benign disease (especially Meibomian gland adenomas).
Tumours of the external globe (cornea and sclera)
Melanoma of the limbus (the corneoscleral junction) is mostly benign in dogs (predisposition in German Shepherds) and cats, although malignancy and metastasis in cats is reported (Betton et al 1999, Day & Lucke 1995, Giuliano et al 1999). Solar radiation is thought to be a risk factor (Hargis et al 1978), because most occur at the superior limbal region. Surgical excision and cryosurgery is usually curative. However, limbal melanomas in older dogs may grow very slowly, and may be better observed than removed, although in younger dogs and in all cats they should be removed early. In older dogs, treatment is undertaken if there is rapid growth or invasion. Surgical excision and cryosurgery is usually curative.
Nd:YAG laser has been used in which 3 of 15 cats and dogs treated had recurrence (Sullivan et al 1996) (slightly higher than for combined excision and cryosurgery). Incomplete excision may result in slow local recurrence, but not metastasis (Wilcock et al 1986). Melanoma of the conjunctiva or third eyelid behaves more aggressively (see above) and must be differentiated from limbal melanoma. Intraocular tumours may extend into the limbus to mimic limbal melanoma, and examination of the iridocorneal angle (gonioscopy) is needed to rule this out.
Papilloma, histiocytoma, SCC, haemangioma, HSA (Figure 25.4), FSA, epithelioma and adenocarcinoma are uncommon tumours of the external globe.
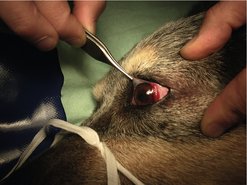 |
| Figure 25.4 (Courtesy R Straw.) |
A lump on the surface of the globe could also be immune-mediated nodular granulomatous episclerokeratitis (NGE) or necrotizing scleritis/keratitis, corneal/conjunctival cysts, foreign bodies, or globe perforation and iris prolapse. SCC of eyelid and conjunctiva can invade the cornea and sclera. Ocular LSA in dogs can invade the cornea, often causing haemorrhage. Infiltrating corneal lesions in dogs and cats may also resemble fibrous histiocytomas, which arise from the limbus. They show continuous growth, a benign appearance and a tendency to recur following excision keratoplasty, Collie dogs may be predisposed (Smith et al 1976).
Diagnostic work-up and staging
A high-frequency ultrasound probe allows tissue examination similar to low-power histology, but tissue penetration is limited to 5–10 mm, ideal for distinguishing between various anterior segment entities (e.g. anterior uveal tumours, iridociliary cysts and iris bombé) (Bentley et al 2003). Other investigations include:
Treatment
Surgery
Surgical options include superficial keratectomy/sclerectomy, full thickness resection of cornea/sclera, and enucleation.
Cryosurgery may be useful for limbal SCC and limbal/epibulbar melanoma. Care should be taken not to damage intraocular tissues.
The use of laser for corneal surgery has been described (Gilmour 2003).
Transplantation of frozen canine amniotic membrane has been used successfully to reconstruct the cornea in dogs and cats (Barros et al 2005).
Porcine small intestinal mucosa grafts covered by a conjunctival flap can be used to repair full thickness corneal wounds in dogs and cats (Bussieres et al 2004).
Intraocular tumours
Animals with intraocular tumours (primary or secondary) may present with glaucoma, uveitis, ocular haemorrhage and retinal detachment, which may disguise the underlying tumour.
Melanoma in dogs
Melanoma is the most common primary intraocular tumour in dogs and cats. It arises from iris or ciliary body (anterior uveal tract), and is rarely choroidal. About 80% of intraocular melanocytomas and 95% of intraocular melanomas were found to arise in the anterior uveal tract. In dogs, intraocular melanoma tends to be benign, with a low rate of metastasis (5%), although they can still be locally invasive (Giuliano et al 1999). Mitotic index is suggested to be the best criterion for histological identification of ocular melanomas with high metastatic potential. Cell type or pattern of growth within the globe was not predictive of biological behaviour (Wilcock & Peiffer 1986).
In a series of 224 cases, tumour extension, tumour size and mitotic index were not found to be reliable predictors of survival. Dogs with MM had only a slightly decreased survival time compared to control dogs and dogs with melanocytomas. At the time of enucleation, most tumours invaded the sclera, but did not show extrascleral extension (Giuliano et al 1999). A worse prognosis was given in a study of 16 dogs with uveal melanomas, where 3 had metastasis within 3 months of enucleation. Benign melanomas tend to be darker and more heavily pigmented than MM (Wilcock & Peiffer 1986). Melanoma of the iris in dogs often appears as a discrete, brown nodule on the surface; however, it can be diffuse and can look like an iris cyst or benign iris melanosis.
Choroidal melanomas are rare (only 4% of canine uveal melanomas). Some remain static for many years; others invade the retina, sclera, optic nerve and orbit. One reported case of a 7-year-old Golden Retriever with a malignant choroid melanoma treated with orbital exenteration had no recurrence or metastasis on thoracic radiographs, and was clinically healthy for 23 months after surgery (Miwa et al 2005). Another Golden Retriever (3 years old) with a choroidal melanoma, with retinal detachment and exophthalmos, was treated with eye enucleation. Histology was benign, but 21 months later gross systemic metastases (lung and liver) were diagnosed as histologically identical to the primary choroidal melanoma (Hyman et al 2002).
Melanoma in cats
In cats, anterior uveal melanomas are the most common primary intraocular tumour (Figure 25.5). Feline iris melanoma tends to present as a diffuse change rather than a discrete mass. A biopsy or aspirate may be helpful to differentiate benign iris melanosis, chronic uveitis and malignant melanoma in cats; however, obtaining a diagnostic sample may be difficult.
 |
| Figure 25.5 (Courtesy R Straw.) |
Malignant uveal melanoma in cats is very infiltrative and has a high rate of metastasis (Patnaik & Mooney 1988), frequently to liver and lungs. Iridial hyperpigmentation takes months to progress, and metastasis is late (1–3 years after eventual enucleation). Cats with melanomas that are larger and more invasive into the iris, ciliary body and scleral venous plexus (and with a higher mitotic index) are more likely to suffer metastasis. Benign anterior iridial melanoma may undergo late malignant transformation in a small number of cats to form larger, diffuse, malignant iris melanoma. In this subset of cats, early enucleation is important to avoid premature death from metastasis. Even with malignant transformation, cats with tumours confined histologically to the iris had good survival times, whereas cats with extensive ocular involvement or secondary glaucoma had shorter survival times (Kalishman et al 1998).
In humans with intraocular melanoma, the tissues around the eye are completely frozen to prevent flow of fluid and blood to or from the tumour, prior to enucleation. This is because manipulation of the tumour at enucleation increases intraocular pressure, significantly decreasing longevity by increasing metastatic potential. People are also advised not to rub or squeeze their eyelids (Fraunfelder et al 1977).
Primary ciliary body epithelial tumours
Examples of such tumours are adenocarcinoma and adenoma. They usually arise from the ciliary body or iris as a single mass extending from behind the iris into the pupil, and have been reported to infiltrate into the drainage angle and iris.
Carcinomas are the second most common primary intraocular tumour in dogs, rare in cats. They are locally invasive but usually have a low metastatic rate. Transillumination is used to diagnose the extent of the lesion and to differentiate benign ciliary body cysts (especially in the Golden Retriever) from ciliary body tumours.
Intraocular sarcoma
Canine
Histiocytic sarcoma (HS) may occur as an intraocular mass, especially in Rottweilers and Retrievers. In 15 of 26 dogs with ocular HS there were no concurrent systemic clinical signs at presentation, despite known systemic behaviour. Ocular HS has a poor prognosis, and may be differentiated from melanoma (usually a good prognosis) with immunohistochemistry (Naranjo et al 2007).
Feline
Sarcoma is the second most common intraocular tumour in cats, locally aggressive and highly metastatic. Previous ocular trauma which has occurred on an average of 5 years prior to sarcoma development is thought to be the cause (Dubielzig et al 1990). However, not all cases reported have a known previous history of trauma. Ocular trauma that triggers phthisis bulbi results in non-visual eyes that are at risk for late sarcoma development and should be enucleated (Ziess et al 2003). Feline leukaemia virus/feline sarcoma virus (FeLV/FeSV) does not seem to play a role in feline ocular sarcoma development (Cullen et al 1998). Enucleation prior to invasion of the optic nerve or sclera improves the prognosis (Dubielzig et al 1990). If complete removal is not possible due to tumour infiltration (even with orbital exenteration), recurrence is then expected and death will be from locally invasive tumour growth (into optic nerve, brain, periorbital tissues) within a few months of enucleation. The role of radiotherapy has not been evaluated in these patients. Metastasis is also reported.
Other primary tumours in dogs and cats are extremely rare. Blue-eyed dogs are at risk of developing spindle cell sarcoma in the anterior uvea.
Secondary/metastatic tumours
These are uncommon except for LSA (Figure 25.6). In one study, 37% of dogs with lymphoma presented with ocular involvement (second most common presenting sign after lymphadenopathy) (Krohne et al 1994). Survival times in dogs with intraocular LSA are 30–40% shorter than dogs without ocular involvement when treated with chemotherapy (Krohne et al 1994). Animals with multiple myeloma may present with ocular signs. Meningiomas can extend from the brain via the optic nerve.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


