Chapter 43 West Nile Virus in Raptors
Epidemiology
Birds are the primary amplifying host of WNV and infections have been detected in more than 300 species of native and exotic birds in North America, including raptor species of five families.15 Raptor species, including the American kestrel (Falco sparverius), red-tailed hawk (Buteo jamaicensis), Eastern screech owl (Megascops asio), and great horned owl (Bubo virginianus), have been deemed reservoir competent via laboratory-derived infections.10,15,16 Additionally, raptors have presented with a variety of WNV-associated clinical syndromes at rehabilitation, education, propagation, and zoological centers in the United States and Canada.*
Transmission of WNV in nature is maintained between avian hosts and mosquito vectors (most notably Culex spp.) with occasional transmission to dead-end hosts (e.g., humans and horses). Natural infection of vertebrates occurs primarily through mosquito blood feeding, although additional transmission routes of lesser epidemiologic importance likely occur. For example, laboratory infections of birds resulted after ingestion of infectious prey or liquid as well as contact with infectious conspecifics.10,15 The latter most likely occurs because of virus-laden oral secretions being ingested or virus entering microabrasions in oral mucosa or skin during allopreening, aggressive behavior, or parental feeding of offspring or mates. Nonmosquito arthropods such as swallow bugs (Oeciacus vicarious), louse flies (Icosta americana), and ticks (Carios capensis) have been assessed for WNV vector competence, and laboratory-controlled transmission to birds was demonstrated by the latter. However, whether these nontraditional vectors successfully transmit WNV under natural conditions is unknown.
Within tropical and subtropical climates, WNV transmission may occur year-round, whereas temperate regions experience seasonal transmission (from about June through October in the United States and Canada). However, annual and seasonal transmission rates vary, depending on environmental, host, and vector-related factors. Infection rates in birds as estimated by seroprevalence vary geographically and topographically, as well as taxonomically. WNV has been documented in raptors occupying multiple geographic regions throughout the contiguous United States and southern Canada, including owls, hawks, eagles, falcons, and osprey.†
Pathogenesis, Immunity, Clinical Course, and Outcome
As the natural reservoir host of WNV, most birds are susceptible to infection, which often involves marked virus replication in blood and other tissues followed by a robust humoral immune response. Experimental WNV infection in raptors and other birds has led to a better understanding of timing and intensity of shedding, viremia, tissue tropism, seroconversion, and clinical outcomes. Notably, some if not most WNV-infected raptors experience viremia and shed virus soon after infection, usually prior to onset or in the absence of clinical signs. Shedding of infectious WNV via oral and cloacal secretions typically begins within 1 to 3 days after infection and may continue intermittently through 14 or more day(s) postinoculation (DPI), albeit at low titers after 7 DPI. Viremia is often detectable by 1 DPI and is cleared by 6 to 8 DPI in raptors that survive acute infection (i.e., approximately 10 DPI or later); peak viremia titers of some experimentally inoculated raptors were relatively high (i.e., >107.5 plaque-forming units/mL serum) between 2 and 4 DPI. WNV also infiltrates multiple organ systems soon after infection and, again, is usually cleared within approximately 1 to 2 weeks in survivors. However, WNV has been detected in tissues of clinically normal raptors for up to 27 DPI.10,15,16 For raptors that succumb to WNV relatively early after infection (i.e., within 1 to 2 weeks), viral titers in tissues are relatively high at the time of death.
Although the specific roles of innate and adaptive immunity following WNV infection in birds are not known, evidence in mammals suggests that humoral immunity is critical to survival. In raptors, serum antibodies are detectable by approximately 5 to 7 DPI and antibody titers usually increase fourfold or more within the first month after infection; antibodies persist and protect against subsequent infections for 4 years or longer.15–18
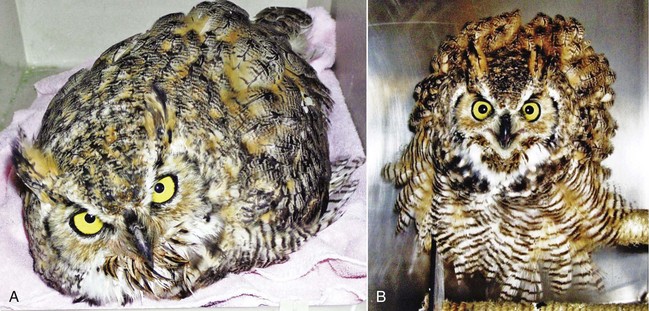
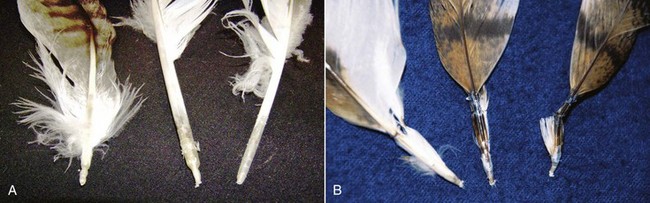
Clinical responses to WNV infection among raptors range from subclinical (likely the most common) to fatal. Although many species are susceptible to WNV neurologic disease, neurologic syndromes appear to be more common in strigiforms, such as the great horned owl, whereas nonspecific syndromes (e.g., emaciation and dehydration) and feather abnormalities (e.g., pinched-off feathers at the calamus, stunted feather growth) may be more common in some falconiforms, such as the American kestrel, red-tailed hawk, and Swainson’s hawk (Buteo swainsoni) and can also occur in owls and eagles9,18,23 (Figs. 43-1 to 43-4). Consistent with the wide array of WNV clinical manifestations among raptors, death may be rapid and unexpected, whereas complete recovery may be prolonged.18,24 The precise timeline between WNV infection and subsequent onset and duration of clinical signs has been difficult to determine in naturally infected birds; death occurred between 4 and 13 DPI in experimentally inoculated birds, including two screech owls at 8 to 9 DPI.10,16
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree