Chapter 35 Use of Analgesics in Exotic Felids
Treatment of pain in domestic and nondomestic cats has been a challenge for the clinician. Many cat species are stoic and show few or subtle external signs of pain. Additionally, the adverse effects of nonsteroidal antiinflammatory drugs (NSAIDs) in domestic cats are well documented and have discouraged many practitioners from trying novel NSAIDs in exotic felids.
NONSTEROIDAL ANTIINFLAMMATORY DRUGS
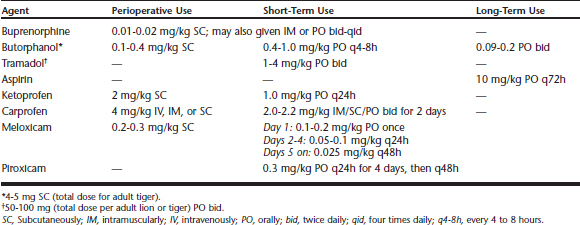
The NSAIDs act both centrally and peripherally. The primary effects are believed to be caused by the ability of NSAIDs to inhibit cyclooxygenase (COX) enzymes in the arachidonic acid metabolism cascade. The COX-1 isoform is regarded as constitutive (continuously expressed) and is responsible for many homeostatic processes, such as maintenance of gastric mucosal integrity, platelet function, and renal autoregulation. Reduced COX-1 activity is believed to be responsible for many of these agents’ adverse effects. The COX-2 isoform is generally thought to be induced in response to noxious stimuli and initiates creation of proinflammatory prostaglandins. Inhibition of the COX-2 isoforms prevents the production of these prostaglandins and the associated pain. This simplification of COX and NSAID physiology is under constant revision but remains the basis for categorizing the activities of NSAIDs. All drug dosages, unless otherwise indicated, are as listed in Table 35-1.
Carprofen is not approved for use in cats in the United States but is approved as a single-dose agent in Europe. It is considered a COX-2 preferential agent and has been studied as a perioperative analgesic for cats. Several authors caution against long-term use of carprofen in cats,11 and there is an anecdotal report of a leopard dying with intestinal ulcers after 2 weeks of carprofen therapy.
Meloxicam is an enolic acid compound with analgesic, antiinflammatory, and antipyretic activities, and it is considered a COX-2 preferential NSAID. It is approved for use in cats in Europe and the United States. The injectable form is approved for single-dose administration, and the oral form is approved for administration up to 5 days. Both the oral and the injectable forms of meloxicam have been used for extended periods in domestic and nondomestic felids with few, if any, adverse effects.
OPIOIDS
Buprenorphine is a partial mu-receptor agonist with potent analgesic properties. It is available in the United States only in the injectable form, but this preparation may also be administered to cats transmucosally, via the oral mucous membranes. Onset of analgesia after oral transmucosal administration is more rapid than when used intramuscularly and is a potential way to administer this drug following recovery from surgery.9 Buprenorpine has relatively few adverse effects when used at recommended dosages and has the distinct advantage of a long duration of activity (>6 hours).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree