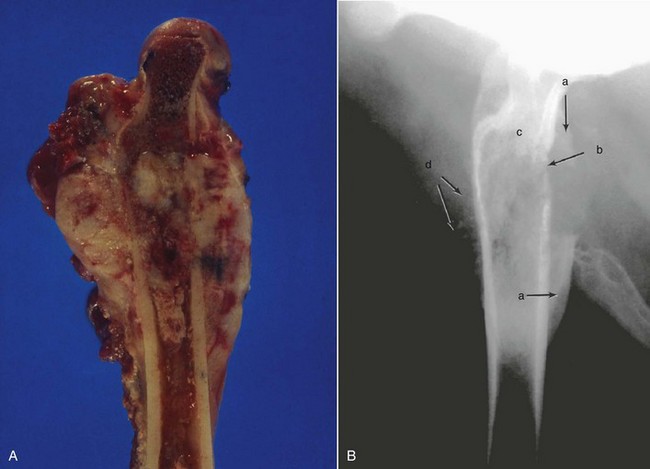
24 Osteosarcoma (OS) is the most common primary bone tumor in dogs, accounting for up to 85% of malignancies originating in the skeleton.1–5 OS is estimated to occur in more than 10,000 dogs each year in the United States; however, this is probably an underestimation, since not all cases are confirmed or recorded.6,7 The demographics of canine OS have been well reported.1–5,8–19 It is largely a disease of middle-aged to older dogs, with a median age of 7 years. There is a large range in age of onset, with a reported case in a 6-month-old pup20 and a small early peak in age frequency at 18 to 24 months.13 Primary rib OS tends to occur in younger adult dogs with a mean age of 4.5 to 5.4 years.21,22 OS is classically a cancer of large and giant breeds. In a review of 1462 cases of canine OS, dogs weighing more than 40 kg accounted for 29% of all cases and only 5% of their tumors occurred in the axial skeleton. Only 5% of OSs occur in dogs weighing less than 15 kg, but 59% of their tumors originated in the axial skeleton. Increasing weight and, more specifically, height appear to be the most predictive factors for the disease in the dog.23 In the United States, the breeds most at risk for OS are Saint Bernard, Great Dane, Irish setter, Doberman pinscher, Rottweiler, German shepherd, and golden retriever; however, size seems to be a more important predisposing factor than breed.* A hereditary basis for the formation of OS has been suspected, based primarily on the (large) breed prevalence of the disease, as well as the subjective assessment of increased incidence in some related families. Males are reported to be slightly more frequently affected than females (1.1 to 1.5 : 1),† with the exception of the Saint Bernard, Rottweiler, and Great Dane and for dogs with primary OS of the axial skeleton (except rib and spine) in which affected females outnumber males.2,22 However, in 1775 cases of canine OS of all sites treated at Colorado State University between 1978 and 2005, the male-to-female ratio was 1 : 1 (unpublished data, Colorado State University Animal Cancer Center [CSU-ACC] OS Database). Intact males and females were reported to have an increased risk for OS23; however, in the Rottweiler breed, male and female dogs that underwent gonadectomy before 1 year of age had an approximate one in four lifetime risk for bone sarcoma and were significantly more likely to develop bone sarcoma than dogs that were sexually intact.24 There was a highly significant inverse dose-response relationship between duration of lifetime gonadal exposure and incidence rate of bone sarcoma independent of adult height or body weight. Approximately 75% of OSs occur in the appendicular skeleton, with the remainder occurring in the axial skeleton.2,22 The metaphyseal region of long bones is the most common primary site, with front limbs affected twice as often as rear limbs, and the distal radius and proximal humerus are the two most common locations.11 It is extremely rare for OSs to be located in bones adjacent to the elbow, although there is one report of 12 cases located at the proximal radius or distal humerus.25 There was no prognostic difference in these cases as compared to more common appendicular sites. In the rear limbs, tumors are fairly evenly distributed between the distal femur, distal tibia, and proximal tibia, with the proximal femur a slightly less common site.2 Primary OS distal to the antebrachiocarpal and tarsocrural joints is relatively rare in dogs.26 In 116 cases of canine primary OS in the axial skeleton, it was reported that 27% were located in the mandible, 22% in the maxilla, 15% in the spine, 14% in the cranium, 10% in ribs, 9% in the nasal cavity or paranasal sinuses, and 6% in the pelvis.22 Single reports of OS development in the os penis27 and the patella28 exist for the dog. Clinically documentable multicentric OS at the time of initial diagnosis occurs in less than 10% of all cases.29 OS of extraskeletal sites is rare, but primary OS has been reported in mammary tissue, subcutaneous tissue, spleen, bowel, liver, kidney, testicle, vagina, eye, gastric ligament, synovium, meninges, and adrenal gland.30–35 The etiology of canine OS is generally unknown. Some have speculated a viral cause because OS can occur in litter mates and may be experimentally induced by injecting OS cells into canine fetii.36 However, an etiologic virus has not been isolated. A simplistic theory based on circumstantial evidence is that because OS tends to occur in major weight-bearing bones adjacent to late-closing physes and heavy dogs are predisposed, multiple minor trauma and subsequent injury to sensitive cells in the physeal region may occur. This may initiate the disease by inducing mitogenic signals, increasing the probability for the development of a mutant lineage. One in vitro study comparing the incidence of microdamage in cadaver radii of small- and large-breed dogs found no difference between the groups.37 OS has been associated with metallic implants used for fracture repair, with chronic osteomyelitis, and with fractures in which no internal repair was used.38–42 OS has also been reported at the site of a bone allograft used for fracture repair 5 years previous.43 Exposure to ionizing radiation can induce OS.35,44–52 In humans exposed to plutonium, 29% and 71% of the OSs were in the appendicular and axial skeleton, respectively, with the spine having the most tumors (36%). An almost identical distribution of plutonium-induced OS was reported for dogs injected with 239Pu as young adults in experimental studies. This distribution of OS is quite different from the distributions of naturally occurring OS for both species and appears to be related to bone volume and turnover. Similar findings were seen for dogs injected with 226R (radium).52 A distribution favoring bone marrow volume was seen for dogs exposed to strontium-90.51 OS is a rare, late complication of radiation therapy (RT) in humans and dogs.44,46,48–50 Three of 87 (3.4%) spontaneous tumor-bearing dogs treated for soft tissue sarcomas developed OS within the field of radiation.44 In another experimental study, 21% of dogs undergoing intraoperative RT (>25 Gy) followed by external-beam RT to the vertebral column developed OS following treatment.48 Secondary OS developed between 1.7 and 5 years after radiation in that study, and the authors speculated that high dose of radiation per fraction or total dose may predispose to this serious late effect of irradiation. OS was reported in 3% of 57 dogs irradiated for acanthomatous epulis of the oral cavity in another study.46 Postirradiation OS in humans comprised approximately 2% to 4% of all OSs reviewed in two large series.53,54 OSs have been concurrently seen in dogs with bone infarcts, but it is not clear whether there is any causal relationship.55–59 Bone infarcts are uncommon, are of unknown etiology, and may be identified as incidental findings by radiography. Bone infarcts are probably not associated with tumor emboli and appear to be more common in smaller breeds. A single report exists of OS occurrence secondary to a bone infarct caused by total hip arthroplasty.60 A case of OS was also reported to be associated with osteochondritis dissecans in the humoral head,61 and another report documents malignant transformation of an aneurysmal bone cyst.62 Ample evidence exists implicating the involvement of genetic and heritable factors for the development of OS in dogs. Currently, the most thoroughly described gene mutation that contributes to OS formation and/or progression in dogs is p53.63–70 Initial studies performed in immortalized canine OS cell lines demonstrated that the functionality of the p53 gene was defective based on the incapacity of p53 to regulate appropriately the transcriptional expression of downstream target genes including p21 and mdm2 following genotoxic insult.65 Furthermore, p53 mRNA and protein were overexpressed in 60% of cell lines and correlated with the presence of missense point mutations within the DNA-binding domain.65 Corroborating in vitro cell line studies, mutations in p53 have also been demonstrated in dogs with spontaneously-arising OS. Several studies using either single-strand conformational polymorphism, polymerase chain reaction, or southern blotting, followed by nucleotide sequence analysis have identified missense mutations involving exons 4 to 8 of p53 in 24% to 47% of all spontaneously arising OS samples.63,67,69,70 In addition to exons 4 to 8, the entire gene sequence of p53 has also been assessed by polymerase chain reaction (PCR) and single-strand conformational polymorphism from 59 spontaneously arising appendicular and axial OS samples.64 In 41% of tumors, p53 mutations were identified, with the majority of abnormalities being point mutations (74%), which resulted in an amino acid substitution, with a lesser percentage of mutations (26%) being deletions.64 Finally, through the implementation of targeted microarray-based comparative genomic hybridization analysis of 38 canine OS cases, similar recurrent cytogenetic aberrations classically present in human OS samples were also identified in OS specimens collected from dogs, including loss of heterozygosity (LOH) of the p53 gene in 18% of tumors.71 Substantiation for the presence of p53 mutations in sporadic canine OS has also been documented by immunohistochemical studies because a hallmark of many p53 mutations is enhanced protein stability of this normally labile protein, enabling detection of protein with methodologies such as immunohistochemistry (IHC).72 In one study evaluating p53 protein expressions in 106 osteogenic tumors, a greater percentage of appendicular (84%) OSs overexpressed p53 protein in comparison with OSs arising from the axial skeleton (56%) and other non-OS bone tumors (20%).68 Finally, loss of p53 gene function in 167 osseous tumors has been characterized by p53 nuclear staining frequency and intensity expressed as a p53 index. Of 103 OS samples, 67% stained positively for p53 protein, and the p53 index was significantly greater in OS derived from the appendicular (n = 84) versus axial (n = 38) skeleton.66 Interestingly, the p53 index of appendicular OS derived from Rottweilers was significantly higher than in Great Danes or other commonly affected breeds, supporting the notion that p53 gene mutations may be associated with breed susceptibilities to OS development.24 Another tumor suppressor gene likely to be permissive for OS development is the RB gene. Based on investigations using five tumorigenic immortalized canine OS cell lines, the RB gene signaling pathway was found to be dysregulated with the persistence of hyperphosphorylated RB protein in the absence of mitogen stimulation. Despite apparent aberrant RB gene signaling, reduction in RB protein was identified in only one of five cell lines.65 Corroborating these in vitro findings, the evaluation of 21 spontaneously arising OSs failed to identify gross RB gene alterations by Southern blotting, and protein expressions of RB were identified in all OS samples evaluated.67 Despite normal protein expression of RB in canine OS samples, the observed translational normalcy does not exclude the possibility for allelic deletion of the RB gene because prior studies in human OS samples have demonstrated that LOH at the RB gene locus does not absolutely correlate with inactivation of RB at the protein level.73 Substantiating the possibility that RB gene may have allelic deletion in spontaneously arising canine OS, analysis of 38 OS samples with comparative genomic hybridization techniques identified copy number loss in 11/38 cases (29%), resulting in a correlative reduction or absence of RB protein expression in 62% of OS samples tested.71 Based on these recent findings, it is probable that aberrations in the RB gene indeed participate in sporadic OS formation and/or progression in dogs. In addition to p53 and RB gene abnormalities, the phosphatase and tensin homolog (PTEN) tumor suppressor gene is suspected to participate in the genetic pathogenesis of canine OS. Original in vitro studies conducted with canine OS cell lines demonstrated that the majority of cell lines (60%) harbored mutations in PTEN, resulting in the absence of gene transcription and protein translation. Corroborating the cell line findings, expression of PTEN was either absent (n = 6) or variable (n = 4) in 15 spontaneously arising OS samples.74 Further support for the loss of PTEN gene in canine OS pathogenesis has been the identification of specific recurrent chromosome copy number aberrations (CNAs) through targeted microarray-based comparative genomic hybridization studies.71,75 In one study utilizing 38 OS samples derived from Rottweilers and golden retrievers, deletion of a chromosomal region (CFA 26q25), which encompasses the PTEN tumor suppressor gene locus, occurred in 42% of OS samples.71 In a subsequent study analyzing 123 OS samples predominantly derived from Rottweilers, greyhounds, golden retrievers, and great Pyrenees, high-resolution comparative genomic hybridization studies similarly identified high recurrent copy number loss encompassing the PTEN gene in 30% of samples.75 In addition to the PTEN gene, other genes identified by high-resolution comparative genome hybridization studies potentially involved in the genetic pathogenesis of OS included overexpression of RHOC and RUNX2 and underexpression of TUSC3.75 A growing body of evidence in dogs supports breed-associated inheritance of OS, especially in Scottish deerhounds, Rottweilers, greyhounds, Great Danes, Saint Bernards, and Irish wolfhounds.24,30,76–79 Many domestic dog breeds have narrow genetic diversity as a consequence of selective breeding practices; this has provided the opportunity to more clearly elucidate the heritability of OS in dogs. For Scottish deerhounds in particular, the reported incidence of OS formation is 15%,76,77 and the narrow heritability in this breed was 0.69, indicating that 69% of the cause for OS development in Scottish deerhounds is due to heritable trait, likely a Mendelian major gene with dominant expression.77 Further studies in Scottish deerhounds using a whole genome linkage approach have mapped a novel locus (OSA1) for OS formation in this breed to CFA34 and provide the opportunity to pinpoint specific candidate genes directly involved in OS etiology for this specific dog breed. Because of the heterogeneous and chaotic nature of OS, it has been difficult to definitively ascribe specific molecular derangements responsible for the etiopathogenesis of OS.80 Nonetheless, substantive progress has been achieved through experimental, preclinical, and comparative investigations to identify dysregulated intracellular signaling and cell survival pathways likely to participate in the pathogenesis of OS. Significantly, the identification and tumorigenic consequences of several putative pathways have been recently characterized not only in immortalized OS cell lines but also in spontaneously arising OS samples. The MET proto-oncogene encodes a tyrosine kinase receptor that on ligation with hepatocyte growth factor (HGF) mediates multiple cellular functions, including cell scattering, motility, and proliferation. Given its biologic activities, excessive or dysregulated MET signaling in canine OS has been demonstrated to promote tumorigenic phenotypes in cell lines.81–83 Furthermore, in a small pilot study with spontaneously arising OS samples, the expression of MET proto-oncogene was identified in the majority (5/7, 71%) of tumor specimens by northern blot analysis.84 Additionally, a novel germline mutation that results in constitutive receptor phosphorylation and aberrant MET signaling has been identified primarily in the Rottweiler breed.85 In a larger study of 59 primary OS samples, mRNA expressions for MET and HGF were detected by real-time PCR (RT-PCR) in all specimens and suggested the existence of a putative MET/HGF autocrine or paracrine feedback loop.86 In a subset of OS samples, proteolytically activated HGF was identified in homogenized tumor lysates (n = 6) by western blot, and MET protein was expressed by 100% of tumors analyzed (n = 16) by IHC. Increased expression of MET mRNA correlated with regional lymph node metastases, supporting the participation of MET signaling for metastases and cell invasion. The cellular effects of growth hormone (GH) are mediated through the hepatic production of insulin-like growth factor-1 (IGF-1). In osteoblasts, IGF-1 induces cell mitogenesis and protection from apoptosis, as well as promoting angiogenesis. Derived from experimental and preclinical investigations, aberrant or excessive IGF-1 signaling likely participates in OS pathogenesis. In three canine OS cell lines, the expression and functionality of the IGF-1/IGF-1 receptor (IGF-1R) signaling cascade has been reported.87 In all cell lines, IGF-1R expression was confirmed by northern blot analysis and radioligand binding studies; however, despite uniform receptor expression, IGF-1–mediated promotion of anchorage independent growth and invasion appeared to be cell-line specific.87 Furthermore, to validate IGF-1/IGF-1R as a molecular target, dogs treated with definitive surgery and systemic chemotherapy received a long-acting somatostatin analog (OncoLAR) in an adjuvant setting to attenuate the protumorigenic effects of IGF-1. Despite significant reductions in circulating IGF-1 concentrations, no clinical benefit was detected in dogs treated with OncoLAR compared to placebo.88 The proto-oncogene erbB-2 encodes human epidermal growth factor receptor-2 (HER-2), which is a tyrosine kinase receptor capable of promoting cell transformation and growth. In both dogs and humans, the overexpression of HER2 protein as a result of gene amplification is a negative prognostic factor in mammary carcinoma; however, less clarity exists for the role of HER2 overexpression in OS.89,90 To better characterize HER2 expressions in canine OS, one study evaluated HER2 mRNA transcript and protein expressions in seven cell lines and 10 OS tumor specimens.91 Based on RT-PCR, the majority of OS cell lines (6/7) and 40% of primary OS tumor samples overexpressed HER2. Similarly, the overexpression of HER2 protein was confirmed by IHC. Although not adequately powered, the results of the study suggested the possibility of HER2 overexpression as a negative prognostic factor for survival. The mammalian target of rapamycin (mTOR) is an evolutionary conserved protein kinase downstream of Akt, which acts as a central hub for the integration of cellular signals induced by growth factors, nutrients, energy, and stress for the purposes of regulating cell cycle progression and growth. As such, aberrant signaling through the mTOR pathway contributes to growth, survival, and chemotherapy resistance in multiple tumor types. To characterize the functionality of the mTOR pathway in canine OS, one study using three canine OS cell lines investigated the expressions of mTOR and p70S6K, a downstream effector protein of mTOR.92 Study results indicated that the mTOR pathway was active in canine OS cells, and phosphorylation of mTOR and p70S6K could be inhibited by rapamycin, resulting in apoptosis and reduced growth of OS cells in vitro. In a complementary phase I dose-escalation study to assess the feasibility of mTOR inhibition as a treatment strategy, the pharmacokinetics and pharmacodynamics of rapamycin were investigated in dogs with primary OS.93 Results from the phase I trial demonstrated that biologically effective concentrations of rapamycin were safely obtainable in dogs and that modulation of mTOR target proteins by rapamycin was achievable within the bone tumor microenvironment. The tropomyosin-related kinase (Trk) proto-oncogenes encode for high-affinity receptor tyrosine kinases (RTKs), including TrkA, B, and C. Specifically in osteoblasts, TrkA receptor binds nerve growth factor (NGF), resulting in cellular mitogenesis and the inhibition of apoptosis. To characterize the possible involvement of TrkA signaling in OS pathogenesis, one study investigated the expression of TrkA and the functional consequences of TrkA receptor ligation in OS cell lines and tumor samples.94 All canine OS cell lines (n = 2) and the majority of primary OS tumors (10/15) and pulmonary metastatic lesions (9/12) expressed TrkA receptors. Additionally, although TrkA signaling did not induce OS cell mitogenesis, blockade of basal TrkA signaling with either a small molecule inhibitor or blocking antibody increased serum starvation-induced apoptosis. The inhibition studies suggest that TrkA signaling participates in OS cell survival and may serve as a novel druggable target. Telomeres are highly conserved nucleoprotein complexes at the ends of linear chromosomes that safeguard against harmful recombination events. The maintenance of telomere length and thus prevention of cellular senescence is through the activity of telomerase, a ribonucleoprotein complex that catalyzes the addition of telomeric sequences to the 3′ ends of chromosomes. Telomerase endows cells with infinite replicative capacity, and given its protumorigenic potential, telomerase has been investigated in canine OS. In an exploratory study, telomerase activity was identified in 100% of OS cell lines (5/5) and in the majority of primary tumors (5/6, 83%).95 In a larger corroborative study using 67 OS tumors, telomerase activity was identified in 73% of OS samples and supported the hypothesis that the majority of OSs possess the enzymatic machinery required for infinite replicative capacity.96 In addition to various growth and survival pathways that potentially contribute to OS pathogenesis, the ability of OS cells to interact with their immediate microenvironment found in bone and lung tissues likely influences OS progression and metastases. Tissue invasion and focal osteolysis are hallmark characteristics of OS, and local disease progression is promoted by several OS-associated proteins, including matrix metalloproteinases (MMPs), receptor activator of NF-κB ligand (RANKL), and lysosomal cathepsin K.97–100 Similar to the ability of OS cells to invade local tissues, specific proteins have been identified to participate in the progression of canine OS metastases, including ezrin,101 a cytoskeletal linker protein, as well as CXCR4,102 a chemokine receptor. OS is a malignant mesenchymal tumor of primitive bone cells. These cells produce an extracellular matrix of osteoid, and the presence of tumor osteoid is the basis for the histologic diagnosis, differentiating OS from other sarcomas of bone. The histologic pattern may vary between tumors or even within the same tumor. Small biopsy samples of an OS may lead to misdiagnoses such as chondrosarcoma, fibrosarcoma, hemangiosarcoma, or simply reactive bone. These histologic diagnoses from small biopsies must be interpreted with caution. It is important to obtain a histologic analysis of the entire tumor following definitive excision to confirm the diagnosis. There are many histologic subclassifications of OS based on the type and amount of matrix and characteristics of the cells: osteoblastic, chondroblastic, fibroblastic, poorly differentiated, and telangiectatic OS (a vascular subtype). Alkaline phosphatase staining on histopathologic and aspiration cytology specimens has been shown to aid in differentiating OS pathologically from other connective tissue tumors.103–105 In dogs, it has not been well established that there is a difference in the biologic behavior of the different histologic subclassifications; however, histologic grade, based on microscopic features, may be predictive for systemic behavior (metastasis).106 Newer techniques (see previous section) designed to recognize molecular or genetic alterations are being evaluated to determine their potential use in predicting behavior of OS.107 The degree of aneuploidy, as measured by flow cytometry, of primary and metastatic tumors is potentially indicative of biologic behavior.80 OS has very aggressive local effects and causes lysis, production of bone, or both processes, which can occur concurrently (Figure 24-1). The local disease is usually attended by soft tissue swelling. Pathologic fracture of the affected bone can occur. Pathologic fracture at presentation in humans and dogs with OS does not necessarily preclude limb salvage and does not carry a worse prognosis than patients without fracture at presentation.108–111 OS rarely will cross a joint surface. This confinement to the bone may be secondary to collagenase inhibitors limiting progression through synovium.112,113 Metastasis is very common and arises early in the course of the disease, although usually subclinically. Although less than 15% of dogs have radiographically detectable pulmonary or osseous metastasis at presentation, approximately 90% will die within 1 year (median survival time [MST]: 19 weeks) with metastatic disease, usually to the lungs, when amputation is the only treatment.2,15 Metastasis via the hematogenous route is most common; however, on rare occasions, extension to regional lymph nodes may occur.114 Although the lung is the most commonly reported site for metastasis, tumor spread to bones or other soft tissue sites occurs with some frequency.115 An increase in the incidence of bone metastasis following systemic chemotherapy has also been documented in humans and is suspected in dogs.116,117 Possible explanations for this change include a change in the behavior of this cancer independent of treatment; selective killing of metastatic cancer by chemotherapy in certain sites such as the lung, which allows metastasis in other sites to become clinically relevant; lung resection and chemotherapy have improved survival, and bone sites become clinically relevant; more sensitive detection methods, which allow previously undetectable metastases to be seen; or more complete and detailed necropsies compared to those performed previously, which identify asymptomatic metastatic sites. The concept of concomitant tumor resistance has been described in which animals harboring large primary tumors are resistant to the growth of smaller metastatic tumors by systemic angiogenic suppression.118 Development and growth of metastatic lesions can be rapid after the removal of the primary tumor and consequent increase in angiogenic activity in animal OS models.119 Suspected locoregional synchronous regional bone metastases (skip metastases) have also been reported.120 Skip metastases in human patients are a rare (1% to 6%) presentation and have a poor prognosis.121 Whole body magnetic resonance imaging (MRI) or positron emission tomography/computed tomography (PET/CT) imaging can aid in detection of occult skip metastases. Some differences in metastatic behavior have been observed based on the anatomic location of the primary OS site. For example, mandibular OS and, to a degree, other calvarium locations may have a less aggressive metastatic behavior, although contradictory evidence exists (see the later section on Therapy and Prognosis).122,123 There is a report of four cases of histologically confirmed OS that subsequently underwent spontaneous regression without tumor-specific treatment.124 This phenomenon, while extremely rare, has also been reported in humans. Dogs with OS of appendicular sites generally present with a lameness and swelling at the primary site. There may be a history of mild trauma just prior to the onset of lameness. This history can often lead to misdiagnosis as another orthopedic or soft tissue injury. The pain is likely due to microfractures or disruption of the periosteum induced by osteolysis of cortical bone with tumor extension from the medullary canal. The lameness worsens and a moderately firm-to-soft, variably painful swelling arises at the primary site. Dogs may present with acute, severe lameness associated with pathologic fractures, although pathologic fractures account for less than 3% of all fractures seen.59 Large- and giant-breed dogs that present with lameness or localized swelling at metaphyseal sites should be evaluated with OS as a likely diagnosis. Dogs rarely have respiratory signs as the first clinical evidence of pulmonary metastasis; rather, their first signs are usually vague. With radiographically detectable pulmonary metastasis, dogs may remain asymptomatic for many months, but most dogs develop decreased appetites and nonspecific signs such as malaise within 1 month. Hypertrophic osteopathy may develop in dogs with pulmonary metastasis (see Chapter 5). Alterations in energy expenditure, protein synthesis, urinary nitrogen loss, and carbohydrate flux have been documented in dogs with OS, similar to results documented in humans with neoplasia. Changes were documented in resting energy expenditure and protein and carbohydrate metabolism in dogs with OS. These changes were evident even in dogs that did not have clinical signs of cachexia.125 Systemic, metabolic derangements reported for dogs with OS include lower chromium and zinc levels, lower iron and iron-binding capacity, and increased ferritin levels as compared to normal dogs.126 Hypercalcemia is extremely rare. The impact of these changes on patient treatment, response, or outcome is unknown. Initial evaluation of the primary site involves interpretation of good quality radiographs taken in lateral and craniocaudal projections. Special views may be necessary for lesions occurring in sites other than in the appendicular skeleton. The overall radiographic abnormality of bone varies from mostly bone lysis to almost entirely osteoblastic or osteogenic changes (see Figure 24-1, B). There is an entire spectrum of changes between these two extremes, and the appearance of OS can be quite variable. There are some features, however, that are commonly seen. Cortical lysis is a feature of OS and may be severe enough to leave obvious areas of discontinuity of the cortex leading to pathologic fracture. There is often soft tissue extension with an obvious soft tissue swelling, and new bone (tumor or reactive bone) may form in these areas in a palisading pattern perpendicular or radiating from the axis of the cortex (i.e., “sunburst”). As tumor invades the cortex, the periosteum is elevated and new bone is laid down by the cambium layer providing a triangular-appearing deposition of dense new bone on the cortex at the periphery of the lesion. This periosteal new bone has been called “Codman’s triangle,” but this is not pathognomonic for OS. OS does not directly cross articular cartilage, and primary lesions usually remain monostotic. The tumors may extend into periarticular soft tissues, however, and adjacent bones are at risk because of extension through adjacent soft tissue structures. Other radiographic changes that can attend OS are loss of the fine trabecular pattern in the metaphysis, a vague transition zone at the periphery of the medullary extent of the lesion (rather than a sharp sclerotic margin), or areas of fine punctate lysis. Any one or combinations of these changes may be seen, depending on the size, histologic subtype, location, and duration of the lesion. The radiographic appearance of OS is similar to osteomyelitis, specifically of fungal etiology.127 In cases in which the travel or clinical history might support the possibility of osteomyelitis, a biopsy with submission for histology and culture may be warranted. A diagnosis of primary malignant bone tumor may be suggested by signalment, history, physical examination, and radiographic findings. Cytology has not been thought to be definitive for diagnosis; however, it may support the tentative diagnosis, and enough confidence in the diagnosis, combined with clinical features and radiographic appearance, may exist to facilitate a discussion of treatment options. Consistent cytologic criteria of OS has recently been described, and with repeated evaluations, dependent on experience, cytopathologists may be more definitive in making a diagnosis from cytology alone.128,129 Alkaline phosphatase staining of cytologic samples has been shown to differentiate OS from other vimentin-positive tumors.103 However, in most cases, a definitive diagnosis lies in procurement and interpretation of tissue for histopathology. With new treatments such as limb sparing (see subsequent sections), knowledge of the specific tumor type may avoid overextension or inappropriate treatment of bone tumors thought to be OS (e.g., chondrosarcoma, myeloma, or lymphoma). It is crucial to the success of a limb-sparing surgery that the biopsy procedure is planned and performed carefully with close attention to asepsis, hemostasis, and wound closure.130 The skin incision for the biopsy must be small and placed so that it can be completely excised with the tumor at limb sparing without compromising the procedure. Transverse or large incisions must be avoided. It has been recommended that the surgeon who is to perform the definitive surgical procedure (especially if limb sparing) should be the person to perform the preoperative bone biopsy.131 Bone biopsy may be performed as an open incisional, closed needle (Figure 24-2), or trephine biopsy. The advantage of an open technique is that a large sample of tissue is procured, which presumably improves the likelihood of establishing an accurate histologic diagnosis. Unfortunately, this advantage may be outweighed by the disadvantages of an involved operative procedure and risk of postsurgical complications such as hematoma formation, wound breakdown, infection, local seeding of tumor, and pathologic fracture.132,133 Although biopsy with a trephine yields a diagnostic accuracy rate of 93.8%, there is the increased risk of creating pathologic fracture when compared with using a smaller gauge needle.134 This underscores some of the advantages of a closed biopsy using a Jamshidi bone marrow biopsy needle (Jamshidi bone marrow needle, American Pharmaseal Co, Valencia, CA; bone marrow biopsy needle, Sherwood Medical Co, St. Louis, MO) or similar type of needle. Jamshidi needle biopsy has an accuracy rate of 91.9% for detecting tumor versus other disorders and an 82.3% accuracy rate for diagnosis of specific tumor subtype.135 Accuracy of diagnoses from needle core samples can depend on the pathologist’s experience and comfort level with examining small samples. Histology reports indicating the presence of reactive bone should not rule out the presence of a primary bone tumor or other pathology, especially if the radiographic changes suggest tumor. In some cases, it can be very difficult to get the diagnosis by preoperative biopsy (i.e., repeated biopsy attempts yield “reactive bone”) and yet the pathologist has no trouble identifying tumor when the entire specimen is available for histopathologic analysis (e.g., postamputation), This is likely a result of the heterogeneity of the tumor tissue itself and the large amount of reactive bone present within the tumor. Figure 24-2 A, The Jamshidi bone biopsy needle: cannula and screw-on cap (a), tapered point (b), pointed stylet to advance cannula through soft tissues (c), and probe to expel specimen from cannula (d). B, With the stylet locked in place, the cannula is advanced through the soft tissue until bone is reached. The inset is a close-up view showing stylet against bone cortex. C, The stylet is removed, and the bone cortex penetrated with the cannula. The cannula is withdrawn, and the procedure repeated with redirection of the instrument to obtain multiple core samples. D, The probe is then inserted retrograde into the tip of the cannula to expel the specimen through the base (inset). Rights were not granted to include this figure in electronic media. Please refer to the printed book. (Reprinted with permission from Powers BE, LaRue SM, Withrow SJ, et al: Jamshidi needle biopsy for diagnosis of bone lesions in small animals, J Am Vet Med Assoc 193:206–207, 1988.) J Am Vet Med Assoc The biopsy site is selected carefully. Radiographs (two views) are reviewed, and the center of the lesion chosen for biopsy. Biopsy at the lesion periphery will often result in sampling the reactive bone surrounding the tumor growth without a resulting diagnosis.135 The skin incision is made so the biopsy tract and any potentially seeded tumor cells can be completely removed at the time of definitive surgery. Care is used to avoid major nerves, vessels, and joint spaces. A 4-inch, 8- or 11-gauge needle is used. With the dog anesthetized, prepared, and draped for surgery, a small stab incision (2 to 3 mm) is made in the skin with a #11 scalpel blade. The bone needle cannula, with the stylet locked in place, is pushed through the soft tissue to the bone cortex. The stylet is removed, and the cannula is advanced through the bone cortex into the medullary cavity using a gentle twisting motion and firm pressure. The opposite cortex is not penetrated. The needle is removed, and the specimen is gently pushed out of the base of the cannula by inserting the probe into the cannula tip. One or two more samples can be obtained by redirecting the needle through the same skin incision so that samples of the transition zone may also be obtained. Ideal specimens should be 1 or 2 cm in length and not fragmented. Biopsy is repeated until solid tissue cores are obtained. Material for culture and cytology may be taken from the samples prior to fixation in 10% neutral buffered formalin. Diagnostic accuracy is improved when samples are evaluated by a pathologist thoroughly familiar with bone cancer. Fluoroscopy or advanced imaging (CT) can assist in obtaining needle-core biopsy samples of suspected bone lesions, especially for axial sites.136 Examination for evidence of apparent spread of the disease is important. Regional lymph nodes, although rarely involved, should be palpated, and fine needle cytology should be performed on any enlarged node.114 Sites of bone metastasis may be detected by a careful orthopedic examination with palpation of long bones and the accessible axial skeleton. Organomegaly may be detected by abdominal palpation. Usually, pulmonary metastases are undetectable by clinical examination, but careful thoracic auscultation is important to detect intercurrent cardiopulmonary disorders. High-detail thoracic radiographs should be taken during inspiration with the patient awake. Although some controversy exists,137 it is still considered important by most oncologists to include three views: a ventrodorsal or a dorsoventral view and both right and left lateral views. OS pulmonary metastases are generally soft tissue dense and cannot be detected radiographically until the nodules are 6 to 8 mm in diameter. It is uncommon to detect pulmonary metastatic disease at the time of diagnosis (<10% of dogs). Advanced imaging (e.g., CT, MRI, PET/CT) may play a role in patient staging and is used to evaluate for pulmonary metastases and for evaluation of tumor vascularity, soft tissue and medullary involvement, metabolic or functional activity, and response to treatment.138,139 Currently, published treatment recommendations and prognoses are based on the results of plain radiographs. As advanced imaging becomes more commonplace for staging dogs with OS, comparisons to previous protocols will be subject to stage-migration and lead-time bias because earlier diagnosis will result.140 Bone survey radiography has been useful in detecting dogs with second skeletal sites of OS.29 Bone surveys include lateral radiographs of all bones in the body and a ventrodorsal projection of the pelvis using standard radiographic technique appropriate for the region radiographed. In one study, 171 dogs with primary bone tumors underwent radiographic bone surveys and thoracic radiography; at presentation, there was a higher yield in finding other sites of OS with radiographic bone survey (6.4%, 11 of 171 dogs) than with thoracic radiographs (4%, 7 of 171 dogs).141 There are conflicting reports on the usefulness of nuclear scintigraphy (bone scan) (Figure 24-3) for clinical staging of dogs with OS.142–146 Bone scintigraphy was used in one study to identify suspected second bone sites of OS in 14 of 25 dogs with appendicular primaries.143 Seven of these lesions were biopsied and confirmed to be OS. Another study of 70 dogs with appendicular primary bone tumors resulted in only one scintigraphically detectable occult bone lesion.142 In a third report, of 23 dogs with suspected skeletal neoplasia that were evaluated with scintigraphy and radiography, 4 dogs had second skeletal sites suspected to be neoplastic.146 The suspicious site in one of these dogs was found on histologic evaluation to be normal bone. Another study found secondary sites considered highly suspect of bony metastasis in 7.8% of 399 cases; however, most suspected lesions were not subjected to histologic conformation.144 Nuclear bone scan can be a useful tool for the detection and localization of bone metastasis in dogs presenting for vague lameness or signs such as back pain, and, although very sensitive, it is not specific for identifying sites of skeletal tumor. Any region of osteoblastic activity will be identified by this technique, including osteoarthritis and infection. Follow-up with high-detail radiographs of sites found suspicious on scintigraphy will generally help rule out nonneoplastic disease; however, definitive biopsy may be necessary. A surgical staging system for sarcomas of the skeleton has been devised for humans.147 This system is based on the histologic grade (G), the anatomic setting of the primary tumor (T), and regional or distant metastasis (M). There are three stages: stage I, the low-grade (G1) lesions without metastasis; stage II, the high-grade (G2) lesions without metastasis; and stage III, the lesion with regional or distant metastasis regardless of histologic grade. The stages are subdivided by the anatomic setting: A is intracompartmental (T1) and B is extracompartmental (T2). According to this system, most dogs with OS present with stage IIB disease. Scintigraphy can be used to evaluate the degree of bone involvement from a primary bone tumor.148 In one study, scintigraphy overestimated the length of OS disease in limb-sparing patients by 30%, allowing for adequate margin prediction, preoperatively.149 CT may be useful to plan surgery, especially for tumors located in the axial skeleton; however, one study reported that plain radiographs were as accurate as advanced imaging (CT, MRI) in predicting true length of tumor involvement.150 In contrast, MRI was more accurate than plain radiographs or CT in predicting length of tumor involvement for appendicular canine OS in another study.151 This could prove invaluable for limb-sparing patients, and further evaluation is warranted. Anatomic Location and Signalment In a multiinstitutional study of 162 dogs with appendicular OS treated with amputation alone, dogs younger than 5 years of age had shorter survival than older dogs.15 Additional studies have related large tumor size13,152,153 and humerus location154,155 to poor outcome. Large tumor size has been reported to be a negative prognostic factor for humans with OS.156 For OS originating from flat bones, small dog size and completeness of excision were positive prognostic indicators.157 Although there are differences in disease distribution and prevalence, documentation of improved survival for small dogs with OS is lacking.158 A negative prognosis can also be predicted by a higher tumor grade and mitotic index, based on the results of one study.106 The biologic behavior for nonappendicular sites of OS appears to be similar (aggressive) with the exception of the mandible and possibly the rest of the calvarium.122,123,159–162 OS affecting the head (mandible, maxilla, and skull) is locally aggressive but has a lower metastatic rate (37%) than appendicular OS.162 Reported median disease-free intervals (DFIs) and survival times for skull OS are 191 days and 204 days, respectively. Dogs with OS of the mandible treated with mandibulectomy alone had a 1-year survival rate of 71% in one study.122 In contrast, maxillary OSs have demonstrated a median survival of 5 months following maxillectomy.159,160 A study evaluating response to treatment for orbital OS reported long-term survival following complete surgical excision.163 Similar behavior is seen for OSs of flat bones in humans.164 Median survival for rib OS lesions is reported to be 3 months for dogs treated with rib resection alone and 8 months for dogs treated with resection and adjuvant chemotherapy.165–168 OS of the canine scapula has been reported to have a poor prognosis when treated with subtotal scapulectomy surgery and chemotherapy.157,169,170 DFI and MST in dogs diagnosed with scapula OS was 210 days (range 118 to 245 days) and 246 days (range 177 to 651 days), respectively.171 Use of adjunctive chemotherapy was prognostic for both increased DFI and survival. Limb function after subtotal scapulectomy is good to excellent.171 Survival of dogs with OS distal to the antebrachiocarpal or tarsocrural joints was somewhat longer (median: 466 days) than survival of dogs with OSs of more common appendicular sites; however, OS in these sites is aggressive, with a high potential for metastasis.26 Vertebral OS is uncommon; however, reported cases indicate aggressive local and systemic behavior.22,172 In 15 dogs treated with a combination of surgery, radiation, and chemotherapy, the median survival was 4 months.172 The biologic behavior of OS in other nonappendicular bone sites (e.g., pelvis) has not been thoroughly evaluated. Extraskeletal OS is rare and most commonly affects visceral sites (gastrointestinal tract, spleen, liver, kidney, urinary bladder), skin or subcutaneous tissue, or mammary glands. Extraskeletal (soft tissue) OS sites also appear to have aggressive systemic behavior with a high metastatic rate. In one report, extraskeletal OS treated with surgery alone had a median survival of only 1 month, and a median survival of 5 months was obtained for cases treated with surgery and adjuvant chemotherapy.31 In a larger study, soft tissue OSs were separated from mammary gland OSs; median survival of nonmammary gland soft tissue lesions was 1 month and mammary gland lesions 3 months following primarily surgical resection alone.32 The major cause of death was local recurrence (92%) in the soft tissue OS cases and pulmonary metastasis (62.5%) in the mammary gland OS cases. Dogs presented with stage III disease (measurable metastases) have a very poor prognosis.115 MST of 90 dogs with stage III disease at presentation was 76 days, with a range of 0 to 1583 days. No significant differences in survival times on the basis of age, sex, breed, or primary site were observed. Dogs with bone metastases (132 days) had a longer survival time than dogs with lung (59 days) or lung and other soft tissue (19 days) metastases. Dogs with lymph node metastasis had short survivals with a median of only 57 to 59 days, compared to 318 days for dogs without nodal spread.114,115 Dogs with stage III disease treated palliatively with RT and chemotherapy had a significantly longer survival time (130 days) than dogs in all other treatment groups. Elevated alkaline phosphatase (AP) has been clearly associated with a poorer prognosis for dogs with appendicular OS in several studies.114,154,173–175 A preoperative elevation of either total (serum) or the bone isoenzyme of AP (>110 U/L or 23 U/L, respectively) is associated with a shorter DFI and survival (Figure 24-4). Likewise, dogs that have elevated preoperative values that do not return to normal within 40 days following surgical removal of the primary lesion also develop earlier metastasis. One study substantiated the predictive nature of elevated preoperative AP levels; however, no association was found for elevated postoperative serum levels.173 Figure 24-4 A, Disease-free interval (DFI) outcome of dogs treated for OS comparing bone alkaline phosphatase levels above and below 23 U/L preoperatively. B, Survival outcome of dogs treated for OS comparing serum alkaline phosphatase levels above and below 110 U/L preoperatively. BALP, Bone alkaline phosphatase; TALP, total alkaline phosphatase. Rights were not granted to include this figure in electronic media. Please refer to the printed book. (Reprinted with permission from Erhart N, Dernell WS, Hoffmann WE, et al: Prognostic importance of alkaline phosphatase activity in serum from dogs with appendicular OS: 75 cases (1990-1996), J Am Vet Med Assoc 213:1003, 1998.) J Am Vet Med Assoc
Tumors of the Skeletal System
Osteosarcoma in Dogs
Etiology
Physical Factors
Genetic Factors
Molecular Factors
Pathology and Natural Behavior
History and Clinical Signs
Systemic Alterations
Diagnostic Techniques and Work-Up
Tissue Biopsy
Staging and Patient Assessment
Systemic Staging
Surgical Staging
Known or Suggested Prognostic Factors
Serum Alkaline Phosphatase

![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree