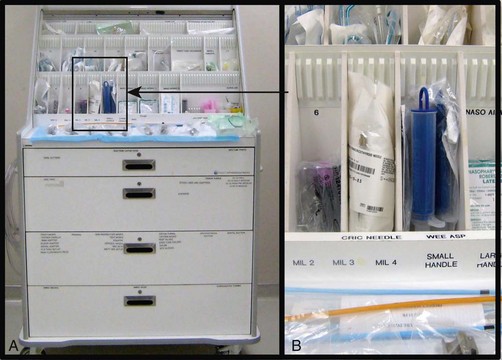
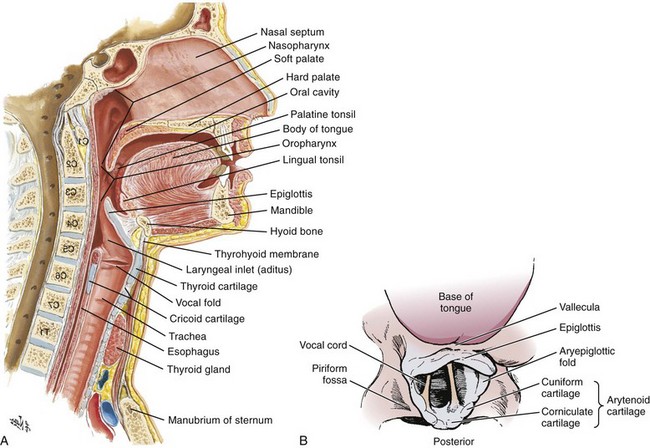
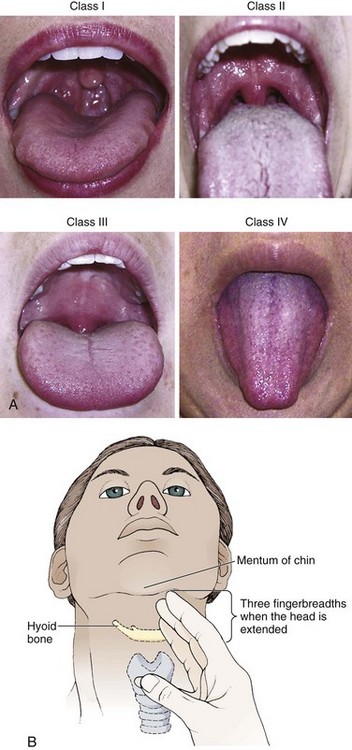
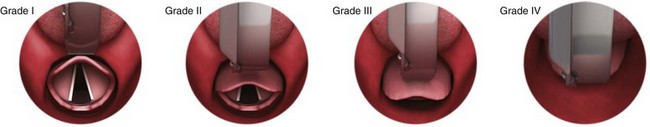
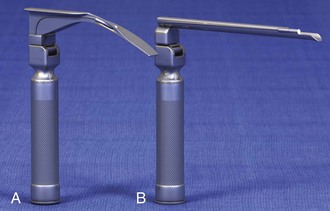
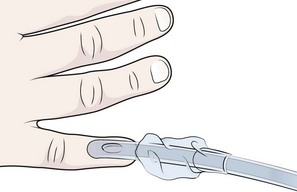
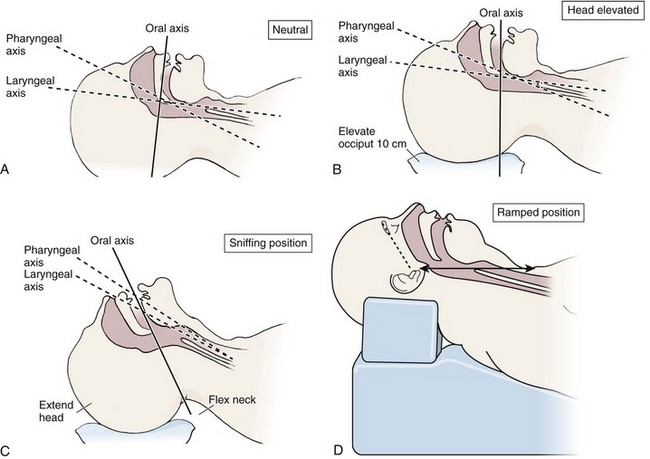
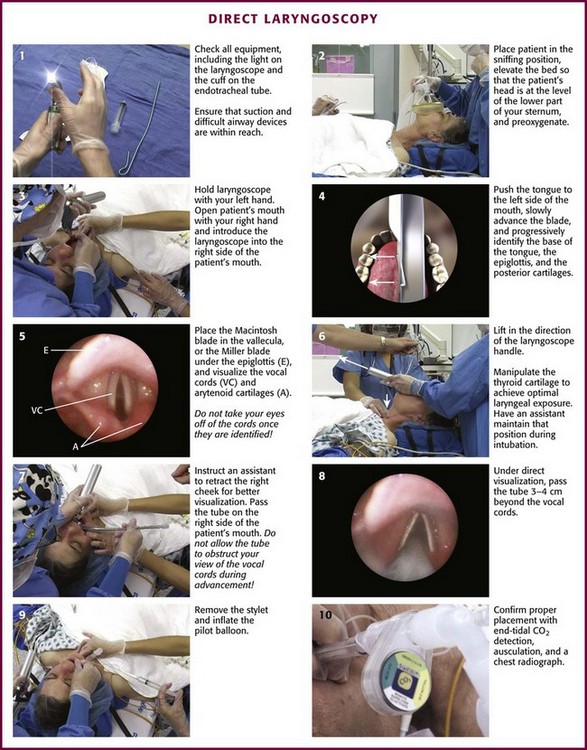
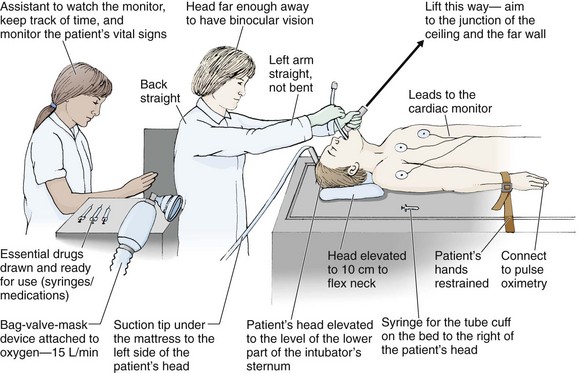
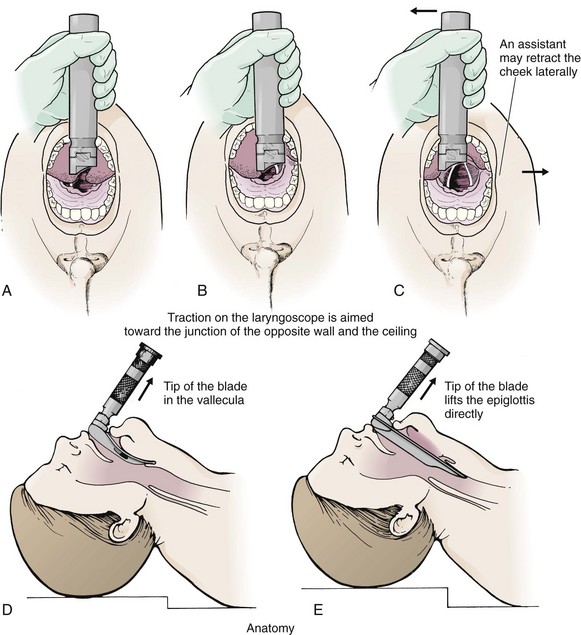
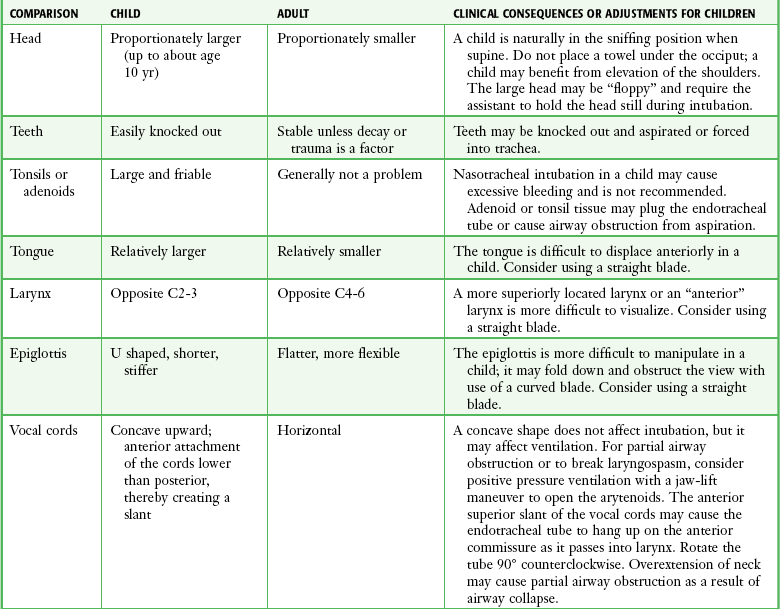
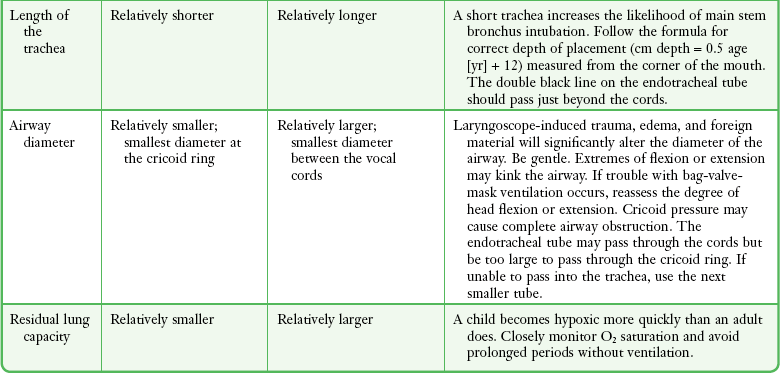
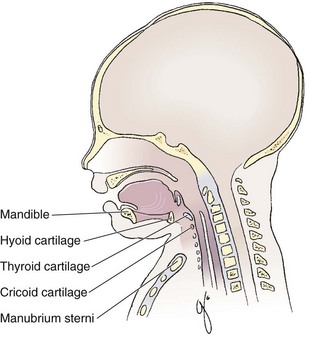
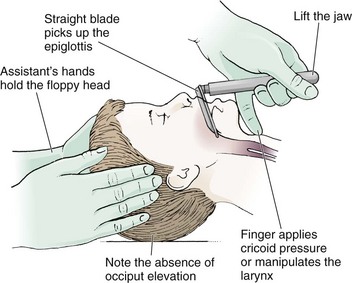
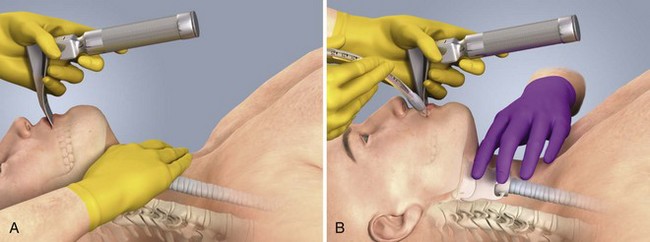
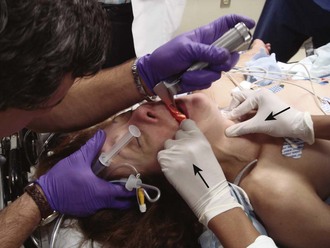
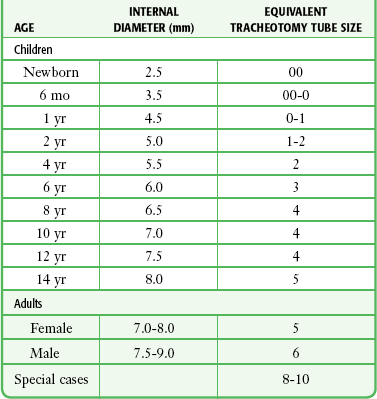
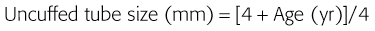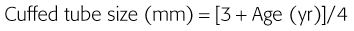Chapter 4 Intubation is often the pivotal procedure in the emergency management of critically ill patients. There are several new devices that can improve the likelihood of successful intubation, but it is important to understand that intubation is just one aspect of airway management. The primary objective of airway management is to maintain adequate oxygenation. Intubation allows oxygenation once it is completed, but not during the procedure.1 A comprehensive approach, such as the algorithm described in Chapter 3, is crucial (see Fig. 3-15).2 In the emergency setting it is important to use intubation techniques that have a high likelihood of success, critically recognizing when a given approach has failed and quickly moving to a different technique. This chapter describes nearly every possible means of tracheal intubation, with emphasis on widely used techniques. The most common means of intubation in the emergency setting is rapid-sequence intubation (RSI), but this approach must be considered very carefully.3 If a difficult intubation is anticipated, awake intubation may be preferred. With good topical anesthesia, nearly any intubating technique can be used for awake intubation, although flexible fiberoptic devices are commonly used. Because many difficult airways cannot be predicted, it is essential to have a well-defined backup plan and appropriate resources. The intubating laryngeal mask airway (LMA Fastrach) is an ideal backup device for failed RSI because it provides ventilation and oxygenation and facilitates tracheal intubation in a high percentage of patients with failed RSI.2 Preplanning is the key to successful emergency airway management. Providers should follow a clear, preconceived, practiced airway algorithm that uses readily available and familiar equipment and techniques. When encountering a difficult airway, it is more important to be comfortable with a few well-proven devices and techniques than to try new or unfamiliar devices.2,4 It is important to know when to abandon one approach and move on to the next. No single approach is mandated. The best technique is the one chosen by the clinician at the bedside based on one’s individual experience and expertise and the specific clinical scenario. A critical aspect of preparation is making sure that all essential equipment required to perform the airway maneuvers is immediately available and within easy access. This may be accomplished by wall-mounting essential equipment in the emergency department (ED) resuscitation room.5 Alternatively, equipment can be placed in dedicated adult and pediatric airway carts or tackle boxes in an open, organized, and labeled manner, and the carts and boxes checked regularly and stocked (Fig. 4-1).6 Essential equipment that is seldom used but is potentially lifesaving should be clearly identified and placed in an easily accessible location such as a dedicated difficult airway cart. The importance of this concept cannot be overstated. Technical expertise cannot substitute for the lack of essential equipment. In airway management, failure has ominous consequences. Mental, physical, and equipment preparation maximizes the chance of success. An understanding of airway anatomy and its terminology is requisite for any discussion of airway management procedures (Fig. 4-2). The following terms are used frequently in this chapter: Pharynx: the upper part of the throat posterior to the nasal cavity, mouth, and larynx 1. Nasopharynx: base of the skull to the soft palate 2. Oropharynx: soft palate to the epiglottis 3. Hypopharynx: epiglottis to the cricoid ring (posteriorly), including the piriform sinus/recess/fossa Piriform sinus/recess/fossa: the pockets on both sides of the laryngeal inlet separated from the larynx by the aryepiglottic folds Larynx: the anterior structures of the throat (commonly called the voice box) from the tip of the epiglottis to the inferior border of the cricoid cartilage, including the laryngeal inlet Laryngeal inlet: the opening to the larynx bounded anterosuperiorly by the epiglottis, laterally by the aryepiglottic folds, and posteriorly by the arytenoid cartilage Arytenoid/posterior cartilage: the posterior aspect of the laryngeal inlet separating the glottis (anterior) from the esophagus (posterior) 1. Corniculate cartilage: the medial portion of the arytenoid/posterior cartilage 2. Cuneiform cartilage: the lateral prominence of the arytenoid/posterior cartilage 3. Interarytenoid notch: the notch between the posterior cartilage Glottis: the vocal apparatus, including the true and false cords and the glottic opening Vallecula: the space between the base of the tongue and the epiglottis Hyoepiglottic ligament: anterior midline ligament connecting the epiglottis to the hyoid bone Intubation is best accomplished with two operators, one to perform the intubation and the other to handle equipment, help with positioning, observe the monitor, and keep track of time. Unfortunately, the ideal scenario and adequate time for preparation are not always available to the clinician, who has to make calculated adjustments based on the situation at hand. Before intubating, it is preferable to take the following steps in chronologic order: (1) attach the necessary monitoring devices and administer oxygen, (2) establish intravenous access, (3) draw up essential medications and label them if time permits, (4) confirm that the intubation equipment is available and functioning, (5) reassess oxygenation and maximize preoxygenation, (6) position the patient correctly, and (7) make sure that all team members are aware of the primary procedural approach and the most likely backup plan. In the haste of the moment, it is a common error to forget to preoxygenate or to position the patient optimally. Simple omissions, such as failing to restrain the patient’s hands or remove the patient’s dentures or misplacing the suction tip, can seriously hamper the success of the procedure. One suggested preintubation checklist is presented in Box 4-1. In addition, be sure to use universal precautions by wearing gloves, a gown, and eye and mouth protection. Preoxygenation is one the most important aspects of emergency airway management. Preoxygenation before RSI provides much more time for intubation before desaturation occurs and thus significantly increases the chance for successful intubation on the first attempt. Failure to preoxygenate before RSI is often a critical factor when a straightforward emergency airway becomes an unexpected airway problem. Preoxygenate by providing the maximal fraction of inspired oxygen (Fio2) with a non-rebreather mask for 3 to 5 minutes before intubation. Alternatively, give eight vital capacity breaths from a maximal Fio2 system, such as a non-rebreather mask or a bag-valve-mask device, if there is not enough time for standard preoxygenation.7 When using a bag-valve-mask device for preoxygenation it is important for the exhalation port to have a one-way valve so that room air is not drawn into the mask when the patient inhales. The purpose of preoxygenation is not solely to maximize oxygen saturation, but also to wash out nitrogen from the patient’s lungs and replace it with oxygen. This provides the maximum safe apneic time during RSI. Those at greatest risk for rapid desaturation include obese, pregnant, critically ill, and pediatric patients, and they will benefit the most from appropriate preoxygenation. Most studies addressing preoxygenation have been conducted under ideal conditions on relatively healthy individuals. It is far more challenging to effectively preoxygenate critically ill patients.8 Morbidly obese patients are best preoxygenated in a 25-degree head-up position because significantly higher oxygen tension can be achieved with this position.9 Patients who are hypoxic despite maximal oxygen delivery have been shown to benefit from 3 minutes of noninvasive positive pressure ventilation (NPPV) with 100% O2 just before intubation.10,11 Sometimes patients who will benefit the most from preoxygenation are uncooperative with a face mask because of delirium from hypoxia, hypercapnia, or other factors. These patients may benefit from delayed-sequence intubation: careful sedation without suppression of respirations, and oxygenation with a face mask or NPPV for 2 to 3 minutes before administration of a paralytic agent.11 Ketamine (1 to 1.5 mg/kg by slow intravenous push) has been suggested for this technique. Delayed-sequence intubation is a commonsense way to deal with a difficult problem, but it has not been well studied and providers using this method should be ready for hypoventilation or apnea. Another method to delay desaturation during RSI is nasopharyngeal oxygen insufflation during apnea. Many studies have shown that providing oxygen therapy during apnea is much more beneficial than previously considered.12–15 During apnea, oxygen continues to be absorbed through the alveoli, and thus air with a low oxygen concentration remains in the lower airways. Supplying oxygen to the nasopharynx during apnea allows air with a higher oxygen concentration to passively replenish oxygen in the alveoli. In two studies of patients undergoing RSI, nasopharyngeal oxygen insufflation significantly delayed desaturation after the onset of apnea in both normal and morbidly obese patients.12,15 In these studies, nasopharyngeal oxygen insufflation was accomplished by inserting a 10-Fr catheter into the nasopharynx to a distance equal to that from the mouth to the tragus of the ear and delivering oxygen at a flow rate of 5 L/min when apnea occurred.12 Using a standard nasal cannula with a nasopharyngeal airway is simpler and would probably provide the same benefit. It is important to keep the upper airway open with a jaw thrust or artificial airway for this technique to be most beneficial. When trying to predict whether there will be difficulty during emergency intubation, it is important to understand that most of the literature on prediction of difficult laryngoscopy does not apply very well in the emergency setting. A study by Levitan and colleagues in 2004 showed that two thirds of patients who were intubated in their ED via RSI could not be assessed with the most common difficult airway prediction tests (Mallampati scoring, measurement of thyromental distance, and neck mobility testing) because of altered mental status or cervical spine immobilization.16 Even in the best circumstances, only about half of cases of difficult laryngoscopy can be predicted.17 Many factors such as Mallampati scoring and measurement of thyromental distance have not been found to accurately predict difficult laryngoscopy, especially in the emergency setting.16–19 Only obvious anatomic and pathologic abnormalities and a history of difficult intubation are accurate predictors of difficult laryngoscopy.16 The American Society of Anesthesiology Difficult Airway Guidelines state that “in patients with no gross upper airway pathology or anatomic anomaly, there is insufficient published evidence to evaluate the effect of a physical examination on predicting the presence of a difficult airway.”20 This does not mean that emergency providers should ignore factors that are known to be associated with difficult laryngoscopy, but these must be placed in perspective. No examination finding alone can predict difficult laryngoscopy, but a combination of multiple factors makes difficulty more likely. The classic predictors of difficult intubation include a history of previous difficult intubation, prominent upper incisors, limited ability to extend at the atlanto-occipital joint,21 poor visibility of pharyngeal structures when the patient extends the tongue (Mallampati classification or the tongue-pharyngeal ratio) (Fig. 4-3A),22 limited ability to open the mouth (suggested by a space less than three fingerbreadths between the upper and lower incisors),23 short thyromental distance (<6 cm from the thyroid notch to the chin with the neck in extension) (Fig. 4-3B),24 and a limited direct laryngoscopic view of the laryngeal inlet (Fig. 4-4).23 A relatively new test is the upper lip bite test, which has been shown in some studies to be more accurate and specific than older tests.25–27 It is essentially a test of anterior mandibular mobility, and the less mobility, the more difficult it is to intubate the patient. Upper lip bite criteria are as follows: class I, the lower incisors can bite the upper lip above the vermillion line; class II, the lower incisors can bite the upper lip below the vermillion line; and class III, the lower incisors cannot bite the upper lip.25 Interestingly, a 2008 study by Tremblay and associates identified factors predictive of difficult GlideScope intubation, which included a high Cormack-Lehane grade during direct laryngoscopy, short thyromental distance, and high upper lip bite test score.28 As noted earlier, many of these predictors cannot be assessed in the emergency setting.16 Fortunately, some of the key predictors are apparent simply by observing the external appearance of the patient’s head and neck. Patients with neck tumors, thermal or chemical burns, traumatic injuries involving the face and anterior aspect of the neck, angioedema, infection of pharyngeal and laryngeal soft tissues, or previous operations in or around the airway suggest a difficult intubation because distorted anatomy or secretions may compromise visualization of the vocal cords. Facial or skull fractures may further limit airway options by precluding nasotracheal (NT) intubation. Patients with ankylosing arthritis or developmental abnormalities such as a hypoplastic mandible or the large tongue of Down’s syndrome are difficult to intubate because neck rigidity and problems of tongue displacement can obscure visualization of the glottis. Besides these obvious congenital and pathologic conditions, the presence of a short, thick neck is one of the more common predictors of a difficult airway. Such individuals are easily identifiable by observing the head and neck in profile. Obesity alone may not be an independent predictor of difficult intubation, but obese patients with large-circumference necks are likely to be difficult to intubate.29 Facial hair can complicate a difficult airway by rendering bag-mask ventilation ineffective because of the lack of a good mask seal. One patient type that does not immediately stand out as a difficult intubation—but can be surprisingly so—is a patient with an unusually long mandibulohyoid distance (the thyroid prominence appearing low in the neck) and a short mandibular ramus.30 Visualization of the larynx is difficult because of the distance to the larynx and the relative hypopharyngeal location of the tongue. Knowledge of the poor performance of difficult airway predictors should not make emergency providers more cavalier about using RSI; it should create more concern. The solution is not to avoid RSI, because lack of paralysis makes every intubation more difficult. Instead, it is essential to appreciate the critical importance of having a clear backup plan when intubation with RSI fails.16,31 This situation mandates the need for a preconceived algorithm that uses proven rescue techniques applicable to a broad range of clinical scenarios, such as the bougie (flexible intubating stylet) and the LMA Fastrach.2 The value of the bougie is indisputable, and it is clear that using the LMA Fastrach after failed RSI has decreased the frequency of failed airways and the need for surgical intervention.2,4,32–38 Finally, if RSI is the “go to” method for emergency intubation, providers must be prepared to perform a surgical airway when laryngoscopy, bag-mask ventilation, and backup devices fail.2,39 There are two basic blade designs for direct laryngoscopy, curved (Macintosh) and straight (Miller) (Fig. 4-5). Each comes in various adult and pediatric blade sizes. Slight variations in laryngoscopic technique follow from the choice of blade design, and it is often a matter of personal preference. The tip of the straight blade goes under the epiglottis and lifts it directly, whereas the curved blade fits into the vallecula and indirectly lifts the epiglottis via engagement of the hyoepiglottic ligament to expose the larynx. The illumination provided by the laryngoscope can make a big difference in the ability to visualize the laryngeal inlet. The importance of these factors is underappreciated, as demonstrated by Levitan40 in a survey of the Macintosh blades used in 17 Philadelphia EDs. It was found that only 24% of all blades provided the brightness necessary for fine inspection. This finding was largely explained by the fact that the majority of EDs used the A-Mac (American) as opposed to the clearly superior brightness design of the G-Mac (German) or the intermediate brightness of the E-Mac (English). The standard adult endotracheal (ET) tube measures approximately 30 cm in length. Tube size is typically printed prominently on the tube and is based on the internal diameter (ID) and measured in millimeters. The range is 2.0 to 10.0 mm in increments of 0.5 mm. The outer tube diameter is 2 to 4 mm larger than the internal diameter.41 Tubes are also imprinted with a scale in centimeters that indicates the distance from a tube’s distal tip. Correct tube size is important in the pediatric population. It is especially important when using an uncuffed tube because a good seal is needed between the ET tube and the upper part of the trachea (Table 4-1). Since tube size is based on the ID, a cuffed tube should generally be a half size (0.5 mm) smaller than an uncuffed tube. The smaller ID of an appropriately sized, small cuffed tube could theoretically make it more prone to plugging from secretions. Cuffed tubes are available down as small as 3-mm ID, although indications for these tubes in neonates and infants are rare. A cuffed tube is used in children with decreased lung compliance who may require prolonged mechanical ventilation. In a child, the smallest airway diameter is at the cricoid ring rather than at the vocal cords, as in adults. Hence, a tube may pass the cords but go no farther. If this should occur, the next smaller size tube should be passed. TABLE 4-1 Tracheal Tube Sizes for Average Patients* *A slightly smaller size may be required for nasotracheal intubation. Modified from Applebaum EL, Bruce DL, eds. Tracheal Intubation. Philadelphia: Saunders; 1976. For most clinical situations, using the width of the nail of the little (fifth) finger as a guide is sufficiently accurate and has been shown to be more precise than finger diameter (Fig. 4-6).42 A standard tracheal tube uses a high-volume, low-pressure cuff to avoid pressure necrosis the tracheal lining. A clinical test for determining correct cuff inflation is to slowly inject air until no air leak is audible while the patient is receiving bag-tube ventilation. This usually occurs with 5 to 8 mL of air if the proper size tracheal tube has been selected. Many clinicians use the tension of the pilot balloon as a guide to cuff inflation. Slight compressibility with gentle external pressure indicates adequate inflation for most clinical situations. For long-term use, cuff pressure should be measured and maintained at 20 to 25 mm Hg. Capillary blood flow is compromised in the tracheal mucosa when cuff pressure exceeds 30 mm Hg. In emergency situations, the balloon may simply be inflated with 10 mL of air and adjusted when the patient’s condition has stabilized. Interest in design of the tip of the tracheal tube has grown as the Seldinger technique is increasingly being applied to intubation. When a tracheal tube is passed over a smaller-caliber introducer (Seldinger technique), regardless of whether it is a tracheal tube introducer or a fiberoptic scope, there is a reasonable chance that the tube will get hung up on the laryngeal soft tissue.43 A tracheal tube that has been designed to overcome this problem has a bevel oriented posteriorly and a flexible tip that decreases the distance between the tube and whatever it is being passed over (Fig. 4-7). The sniffing position, with the head extended on the neck and the neck flexed relative to the torso, has traditionally been considered the best head position for direct laryngoscopy.44,45 This position aligns the oral, pharyngeal, and laryngeal axes (Fig. 4-8A–C). Horton and colleagues described the ideal sniffing position for normal patients as neck flexion of 35 degrees and atlanto-occipital extension such that the plane of the face is −15 degrees to the horizontal position (Fig. 4-8C).46 In supine patients, neck flexion is achieved by head elevation. Depending on the size and shape of the patient, the amount of head elevation may differ significantly, and the end point should be horizontal alignment of the external auditory meatus with the sternum.44,47–49 In normal-size adults it is usually possible to achieve the sniffing position with 7 to 10 cm of head elevation.50,51 Morbidly obese patients require much more head elevation to achieve the proper sniffing position. In these patients, aligning the external auditory meatus with the sternum requires elevation of the head and neck, as well as the upper part of the back (Fig. 4-8D).3,48,49,52 This can be accomplished by building a ramp of towels and pillows under the upper torso, head, and neck or by using a Troop Elevation Pillow (Mercury Medical, Clearwater, FL) or similar device (see Fig. 3-2).3,48,49,52 Alternatively, elevating the head to a 25-degree backup position may achieve the same purpose.53 Two studies have shown that elevating the head (flexing the neck) beyond the sniffing position often improves visualization of the glottis.48,54 Because the amount of head elevation needed for optimal laryngoscopy varies depending on individual patient anatomy, it is important to make laryngoscopy a dynamic procedure. This is best accomplished by putting your right hand behind the patient’s head to lift, flex, and extend the head as needed to bring the glottis into view.55 Optimal positioning of the head and neck is not possible in trauma patients who require in-line stabilization of the cervical spine. This is one of the aspects that makes trauma airways so challenging and makes other maneuvers, such as external laryngeal manipulation, even more important in these patients.55 Place the patient in the supine position with the head at the level of the lower part of the intubator’s sternum (Fig. 4-9, step 2). To maintain the best mechanical advantage, keep your back straight and do not hunch over the patient; bend only at the knees (Fig. 4-10). Keep the left elbow relatively close to the body and flex it slightly to provide better support. In a severely dyspneic patient who cannot tolerate lying down, perform direct laryngoscopy with the patient seated semi-erect and the clinician on a step stool behind the patient.56 Grasp the laryngoscope in the left hand with the back end of the blade pressed into the hypothenar aspect of your hand. Draw the patient’s lower lip down with your right thumb, and introduce the tip of the laryngoscope into the right side of the patient’s mouth (Fig 4-9, step 3). Slide the blade along the right side of the tongue while gradually displacing the tongue toward the left as you move the blade to the center of the mouth (Fig. 4-9, step 4). If you initially place the blade in the middle of the tongue, it will fold over the lateral edge of the blade and obscure visualization of the airway. Placing the blade in the middle of the tongue and failing to move the tongue to the left are two common errors that prevent visualization of the vocal cords (Fig. 4-11). As you move the tip of the blade toward the base of the tongue, exert force along the axis of the laryngoscope handle by lifting upward and forward at a 45-degree angle (Fig. 4-9, step 6). The direction of this force is critical because if the force is too horizontal or too vertical, poor visualization will result. The epiglottis should come into view with this maneuver. It may help to have an assistant retract the cheek laterally to further expose the laryngeal structures. Avoid bending the wrist because it can result in dental injury if the teeth are used as a fulcrum for the blade. The step after visualization of the epiglottis depends on which laryngoscope blade is being used. With the curved blade, place the tip into the vallecula, the space between the base of the tongue and the epiglottis (Fig. 4-11D). Continued anterior elevation of the base of the tongue and the epiglottis will expose the vocal cords. If the tip of the blade is inserted too deeply into the vallecula, the epiglottis may be pushed down and obscure the glottis.22 When using the straight blade, insert the tip under and slightly beyond the epiglottis and directly lift it up (Fig. 4-11E). If the straight blade is placed too deeply, the entire larynx may be elevated anteriorly and out of the field of vision. Gradually withdraw the blade to allow the laryngeal inlet to drop down into view. If the blade is deep and posterior, the lack of recognizable structures indicates esophageal passage; gradually withdraw the blade to permit the laryngeal inlet to come into view. It is helpful to appreciate the anatomic differences between children and adults when intubating pediatric patients (Fig. 4-12 and Table 4-2). Children’s proportionately larger heads naturally place them in the sniffing position, so a towel under the occiput is rarely necessary (Fig. 4-13). The large head of newborns can result in a posterior positioning of the larynx that prevents visualization of the vocal cords. A small towel under the infant’s shoulders should correct this problem. The head may also be floppy and may benefit from stabilization by an assistant. The child’s increased tongue-to-oropharynx ratio and shorter neck hinder forward displacement of the tongue and, coupled with a U-shaped epiglottis, can make visualization of the glottis difficult. Consequently, direct laryngoscopy in infants and young children is generally best performed with a straight blade: Miller size 0 for premature infants, size 1 for normal-sized infants, and size 2 for older children. The infant’s larynx lies higher and relatively more anterior. If no laryngeal structures are visible after laryngeal pressure, gradually withdraw the blade. Inadvertent advancement of the blade into the esophagus is a common error. Sellick’s Maneuver: There is some good evidence that Sellick’s maneuver (cricoid pressure) helps prevent gastric inflation during bag-mask ventilation.57–62 The only evidence suggesting that cricoid pressure prevents regurgitation consists of five cadaver studies,62–66 one small human study,67 and some case reports68—poor evidence by today’s standards of evidence-based medical practice.57,69 Furthermore, there are many reports of significant regurgitation and aspiration regardless of the application of Sellick’s maneuver.70–73 Despite the lack of evidence, many experts believe that Sellick’s maneuver is critical during RSI.74–77 Because aspiration has dire consequences and Sellick’s maneuver has traditionally been considered integral to patient safety during emergency airway management,69 it is reasonable to apply cricoid pressure as long as it does not interfere with ventilation and intubation. Unfortunately, there is significant evidence that it can interfere with ventilation and intubation, so it is best to apply Sellick’s maneuver on a case-by-case basis with full understanding of the benefits and drawbacks of cricoid pressure.69 When applied during bag-mask ventilation, Sellick’s maneuver reduces tidal volume, increases peak inspiratory pressure, and prevents good air exchange.58,60,61,78–85 There are mixed data about the effects of cricoid pressure during larynoscopy,86 but several studies have shown that it worsens visualization of the larynx.87–89 Cricoid pressure also decreases successful insertion of and intubation through LMAs.90–97 Because of lack of evidence of the utility of Sellick’s maneuver and evidence of adverse effects, routine use of cricoid pressure during bag-mask ventilation of patients in cardiac arrest is not recommended in the 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care.98 Also, it is recommended that cricoid pressure be released immediately if there is any difficulty either intubating or ventilating a patient in an emergency setting.69,73,81,99,100 In addition, it is reasonable to release or relax cricoid pressure during insertion of an LMA, during intubation with an intubating LMA (ILMA), or if ventilation with the LMA is difficult.57,69,96 Some authors believe that improper technique is to blame for the many reported failures of Sellick’s maneuver.57 The proper technique for applying Sellick’s maneuver is to place the thumb and middle finger on either side of the cricoid cartilage and the index finger in the center anteriorly.62 Apply 30 N (6.7 lb) of force to the cricoid cartilage in the posterior direction.57,77 As a reference, about 40 N of digital force on the bridge of the nose will usually cause pain.57 External Laryngeal Manipulation, Bimanual Laryngoscopy, and BURP: ELM is the application of pressure on the thyroid cartilage in an attempt to improve the view of the larynx during laryngoscopy. Multiple studies have shown that ELM performed by the laryngoscopist (bimanual laryngoscopy) is superior to having an assistant apply anterior neck pressure.88,101 Bimanual laryngoscopy is best because the direction and amount of force that will optimize laryngeal exposure is variable. BURP is sometimes optimal, but it often worsens the laryngoscopic view,88 so it is best to move the larynx in a variety of directions to determine the optimal ELM. The best way to quickly apply a variety of different forces to the larynx to determine the optimal ELM is by manipulation of the larynx with the laryngoscopist’s right hand (Fig. 4-14).101 Operator-directed posterior displacement of the larynx during laryngoscopy was described by Brunnings in 1912.102 After Sellick described the use of cricoid pressure to avoid regurgitation in 1961, it became common to have an assistant apply anterior neck pressure during laryngoscopy.62 In 1993 Knill reported that having an assistant apply BURP to the cricoid or thyroid cartilage improved visualization of the glottis during two cases of difficult laryngoscopy.103 In 1993 Takahata and coworkers performed a prospective study of 630 intubations and found that BURP produced better laryngeal exposure than just backward pressure did in patients with difficult laryngoscopy.104 However, a 2005 prospective crossover trial by Snider and colleagues found no benefit with routine application of the BURP maneuver.105 Studies by Benumof, Levitan, and colleagues have demonstrated that it is best to apply pressure on the thyroid cartilage (not the cricoid cartilage) and suggested that ELM should be applied by the laryngoscopist’s right hand, not by an assistant.88,101,106 They also found that the direction of force required for optimal ELM was not always upward and rightward and that the amount of backward pressure was variable.88 In a 1996 study of 181 patients, Benumof and Cooper found that external manipulation was optimal when applied to the thyroid cartilage in 88% of patients and to the cricoid cartilage in only 11% of patients.101 In a 2006 cadaveric study of 1530 laryngoscopies by 104 laryngoscopists, Levitan and associates found that bimanual laryngoscopy was more effective than both BURP and cricoid pressure for optimizing laryngeal exposure (Figs. 4-15 and 4-16; see also Fig. 4-14).88 In addition, they found that cricoid pressure worsened the view of the larynx in 29% of cases and BURP worsened it in 35%.88 Finally, in a 2002 study of eight first-year emergency medicine residents performing 271 intubations in an operating room setting, Levitan and colleagues found that bimanual laryngoscopy (ELM performed by the laryngoscopist) consistently improved laryngeal exposure by novice intubators (see Fig. 4-12).106 Once the vocal cords have been visualized, the final step is to pass the tube under direct vision through the vocal cords and into the trachea. It is best to use a malleable stylet for all emergency intubations. The best stylet shape is straight with a 35-degree hockey-stick bend at the proximal cuff (“straight-to-cuff”). In a 2006 study, Levitan and colleagues showed that stylet bend angles greater than 35 degrees made ET tube passage more difficult.107 If direct laryngoscopy does not bring the vocal cords fully into view, a tracheal tube introducer may be used to facilitate intubation. This adjunct is a long, thin, semirigid introducer that, with the aid of a laryngoscope, is passed through the laryngeal inlet and over which an ET tube is advanced through the cords and into the trachea. The technique, originally described more than 60 years ago by Macintosh,108 was recommended for patients in whom visualizing the vocal cords was difficult. It has also been shown to be effective when the laryngeal inlet cannot be visualized at all.109 It is the most common airway adjunct used in British EDs for complicated intubations.110 Its efficacy has been demonstrated prospectively during difficult intubations in the operating room, as a pivotal component of a difficult airway algorithm in the operating room, and when compared with conventional laryngoscopy in the ED.33,111,112 A variety of tracheal tube introducers are available today (Fig. 4-17). The original adjunct was called the gum elastic bougie, or simply “the bougie,” and is currently available in a reusable form for both adult and pediatric patients (Eschmann Tracheal Tube Introducer, Portex Sims, Kent, UK). The adult size comes in two forms, a 60-cm (15-Fr) version with a short, 40-degree hockey-stick curve at the end and a straight one that is 70 cm. The adult version can accommodate a 5.5-mm ET tube. The pediatric version is 70 cm (10 Fr) and straight and can accommodate a 4.0-mm tube. A polyethylene introducer designed for single use is also available and comes only in the 60-cm version (Flextrach ETTube Guide, Greenfield Medical Sourcing, Austin, TX). A variation of this concept is the FROVA Introducer (Cook Critical Care, Bloomington, IN), a plastic introducer with a similar profile to the others except that it has a hollow lumen through which the patient can be ventilated when an accompanying adapter is attached.
Tracheal Intubation
General Approach to Emergency Intubation
Airway Anatomy
Preparation
Preoxygenation
Assessing for a Difficult Airway
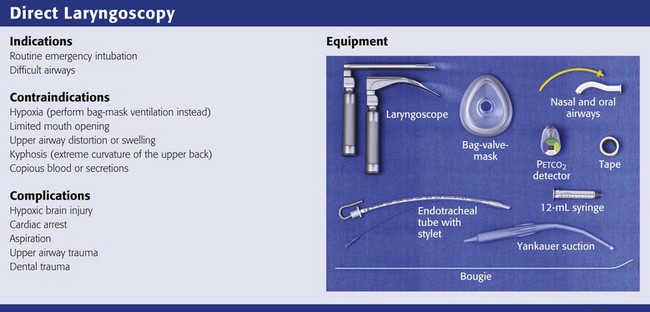
Direct Laryngoscopy
Equipment
Tracheal Tubes
Optimal Patient Positioning for Direct Laryngoscopy
Procedure and Technique of Direct Laryngoscopy
Infants and Children
Sellick’s Maneuver, External Laryngeal Manipulation, Bimanual Laryngoscopy, and BURP
Passing the Tube
Tracheal Tube Introducer (Bougie)
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Tracheal Intubation