CHAPTER 38 The Urinary System
Normal Development
The development of the kidney is complex. There are three stages of development: the pronephros, mesonephros, and metanephros (Table 38-1). A schematic diagram of this process is illustrated in Figure 38-1. Nephrogenesis continues for at least 2 weeks postnatally in the dog. In newborns, the inner glomeruli are larger than those in the outer cortex. However, all cells within the proximal convoluted tubules have a significantly smaller membrane area than the adult. These differences gradually disappear as the kidney grows.
TABLE 38-1 Developmental embryology of the kidney
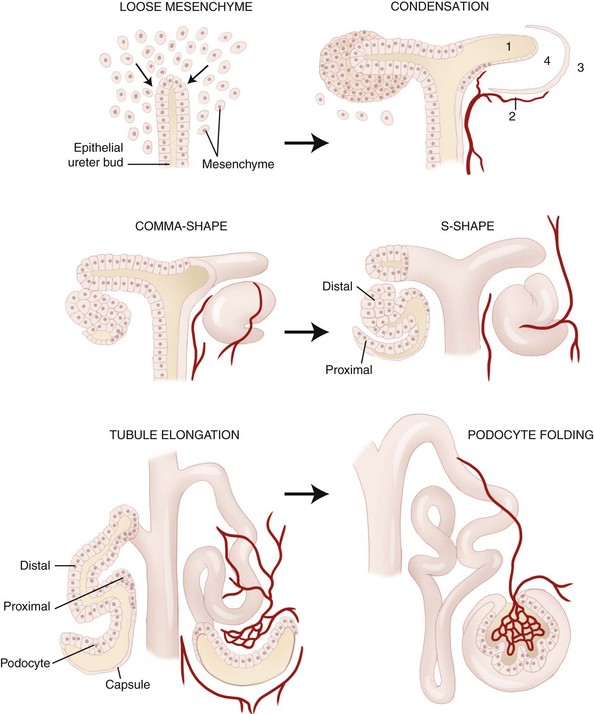
Figure 38-1 Schematic representation of nephrogenesis.
(Modified from Zoetis T, Hurtt ME: Species comparison of anatomical and functional renal development, Birth Defects Research 68(B):112, 2003.)
Congenital Disorders
Congenital disorders of the urinary tract may be caused by the presence of one or more abnormal genes. The end result may be production of an abnormal protein, failure to produce a normal protein, or production of excess amounts of a normal protein. In some instances, these mutations result in clinical syndromes that are sometimes identified in related animals. Familial or hereditary genetic diseases occur when a disease-causing gene is passed from one or both parents to offspring. The mode of inheritance is known for some of these diseases but not for others. Heritable diseases of the urinary tract have been identified in several breeds of dogs (Table 38-2) and cats (Table 38-3) and should be suspected when any of these breeds present with signs consistent with urinary tract disease. Newly affected breeds continue to be identified, and some of these conditions have been found in mixed-breed dogs and cats; therefore it is important that heritable diseases are not ruled out exclusively by signalment.
TABLE 38-2 Canine familial or heritable urinary tract disorders
| Breed | Disorder | Trait |
|---|---|---|
| Alaskan Malamute | Renal dysplasia | |
| Basenji | Fanconi’s syndrome | Familial |
| Bernese Mountain Dog | Membranoproliferative glomerulonephritis | Autosomal recessive |
| Beagle | Renal agenesis | Familial |
| Membranoproliferative glomerulonephritis | Familial | |
| Border Terrier | Renal dysplasia | |
| Fanconi’s syndrome | ||
| Boxer | Renal dysplasia | Familial |
| Brie Sheepdog | Renal dysplasia | |
| Brittany Spaniel | Membranoproliferative glomerulonephritis | Autosomal recessive |
| Bull Terrier | Glomerulopathy | Autosomal dominant |
| Polycystic kidney | Autosomal dominant | |
| Bulldog | Renal dysplasia | |
| Bullmastiff | Glomerulonephropathy | Autosomal recessive |
| Renal dysplasia | ||
| Cavalier King Charles Spaniel | Renal agenesis | |
| Chinese Shar-Pei | Amyloidosis | |
| Chow Chow | Renal dysplasia | Familial |
| Cocker Spaniel | Renal dysplasia–related? | |
| Dachshund | Xanthinuria | |
| Dalmatian | Glomerulopathy | Autosomal dominant |
| Uric aciduria | Recessive | |
| Doberman Pinscher | Renal agenesis | Familial |
| Glomerulopathy | Familial | |
| Dutch Kooiker | Renal dysplasia | Familial |
| English Bulldog | Renal and ureteral duplication | |
| Uric aciduria | ||
| Ectopic ureter | ||
| Urethrorectal fistula | ||
| English Cocker Spaniel | Glomerulopathy | Autosomal recessive |
| English Foxhound | Amyloidosis | |
| Finnish Harrier | Renal dysplasia | Familial |
| Fox Terrier | Ectopic ureter | |
| German Shepherd Dog | Multifocal renal cystadenocarcinoma | Autosomal dominant |
| Golden Retriever | Renal dysplasia | Familial |
| Ectopic ureter | ||
| Great Dane | Renal dysplasia | |
| Labrador Retriever | Ectopic ureter | |
| Lhasa Apso | Renal dysplasia | Familial |
| Miniature Poodle | Urethrorectal fistula, urethroperineal fistula, urethral duplication | |
| Miniature Schnauzer | Renal dysplasia | Familial |
| Fanconi’s syndrome | ||
| Newfoundland | Glomerulopathy | Familial |
| Cystinuria | Autosomal recessive | |
| Ectopic ureter | ||
| Norwegian Elkhound | Fanconi’s syndrome | |
| Pekingese | Renal agenesis | |
| Pembroke Welsh Corgi | Ectopic ureter | |
| Renal telangiectasia | ||
| Poodle | Ectopic ureter | |
| Rhodesian Ridgeback | Renal dysplasia | |
| Rottweiler | Glomerulopathy | Unknown |
| Samoyed | Renal dysplasia | |
| Glomerulopathy | X-linked | |
| Scottish Terrier | Cystinuria | Autosomal recessive |
| Shetland Sheepdog | Renal agenesis | Familial |
| Fanconi’s syndrome | ||
| Shih Tzu | Renal dysplasia | Familial |
| Siberian Husky | Ectopic ureter | |
| Skye Terrier | Ectopic ureter | |
| Standard Poodle | Renal dysplasia | Familial |
| West Highland White Terrier | Ectopic ureter | |
| Soft-Coated Wheaten Terrier | Renal dysplasia | Familial |
| Membranoproliferative glomerulonephritis | Familial |
TABLE 38-3 Feline familial or heritable urinary tract disorders
| Breed | Disorder | Trait |
|---|---|---|
| Abyssinian | Amyloidosis | Autosomal dominant with variable penetrance? |
| Domestic Shorthair | Renal agenesis | |
| Oriental Shorthair | Amyloidosis | |
| Siamese | Amyloidosis | |
| Persian | Polycystic kidney | Autosomal dominant |
Ectopia
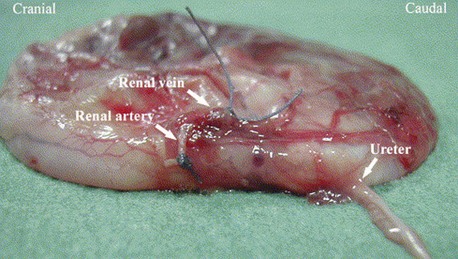
Ectopic ureters occur in both puppies and kittens as a congenital abnormal termination of the ureters with the urinary bladder. Ectopic ureters are described as intramural or extramural. Intramural ectopic ureters enter the bladder wall and run transmurally but fail to empty at the trigone and instead continue transmurally and empty in the urethra or vagina vestibule. Extramural ectopic ureters bypass the urinary bladder completely and enter the urethra, vagina vestibule, or uterus. Females are affected more often than males. Ectopic ureters empty into the urethra of most females and prostatic urethra in males. Ectopic ureters may occur alone or with other developmental abnormalities of the urinary tract. Hydronephrosis may occur if the ureter ends in a blind pouch or forms a functional stenosis. An increased incidence has been found in English Bulldogs, Fox Terriers, Golden Retrievers, Labrador Retrievers, Newfoundlands, Poodles, Siberian Huskies, Skye Terriers, and Welsh Corgis. Clinical signs are related to variable degrees of urinary incontinence. Diagnosis is made with contrast radiography (excretory urogram with retrograde urethrogram or vaginogram), endoscopy, or cross-sectional imaging (Figure 38-2). Surgery is the treatment of choice with transection and reimplantation or unilateral ureterectomy and nephrectomy. In some dogs incontinence persists, and pharmacologic management with diethylstilbestrol or phenylpropanolamine may be necessary.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree