CHAPTER 17 Fungal Infections
Cutaneous Fungal Infections
Dermatophytes
The majority of clinical cases presented are caused by three fungi: Microsporum canis, Microsporum gypseum, and Trichophyton mentagrophytes. Typically M. canis is the most common source of dermatophytosis in dogs and cats. Table 17-1 summarizes dermatophytes isolated from hair and skin of dogs and cats.
TABLE 17-1 Dermatophytes isolated from dogs and cats
| Most common | |
|---|---|
| Microsporum | Most common isolate |
| Microsporum gypseum | Generally from soil contact |
| Trichophyton mentagrophytes | Soil contact |
| Less common | |
|---|---|
| Microsporum persicolor | Sylvatic form |
| Microsporum fulvum | Rare |
| Microsporum audouinii | Rare |
| Trichophyton rubrum | Rare |
Diagnosis
Wood’s lamp
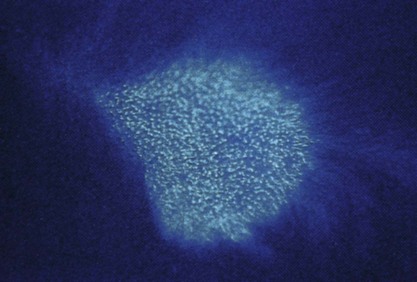
One of the quickest, easiest, and oldest diagnostic tests for fungal identification is the use of the Wood’s light or Wood’s lamp. This is a light emitting ultraviolet rays (320- to 400-nm wavelength) and is a useful initial test for certain dermatophytes. However, the Wood’s lamp is not definitive because only about one-half of all M. canis infections will show fluorescence, and other animal-infecting dermatophytes do not fluoresce at all. The characteristic “apple-green” or “Jimi Hendrix–green” fluorescence results from metabolites of the fungal organism that grow only on the hair and never on scale or claw material. In early infections, the whole hair shaft will fluoresce. As the dermatophyte infection clears, only the more distal portion of the hair glows as the proximal portion of the hair shaft is no longer infected. In later stages of infection, only the very tips of hair shafts fluoresce (Figure 17-1).
Fungal culture
Dermatophyte test medium can be easily obtained for in-house procedures and is best bought as a plate for the toothbrush technique (Figure 17-2). Dermatophyte medium containers are best incubated loosely covered or capped at room temperature protected from ultraviolet light and desiccation, and covered in a container holding a damp paper towel. Colonies of dermatophytes can appear within 5 to 7 days of inoculation. Nevertheless, plates must be maintained for at least 3 weeks before determining a culture negative. Culture plates must be examined daily for medium color change to red. Growth of a white to buff-colored, powdery to cottony mycelium also will be observed. Color change must occur simultaneously as the first colony is visible, never later. All fungi, including nonpathogen fungal growth, will produce a red color change after colonies have growth from several days to a week. Colonies of infectious dermatophytes are never black, brown, gray, or green. Often M. canis will fail to grow or sporulate on dermatophyte medium. Recently studies have demonstrated that increased sporulation and growth can be obtained when plates were incubated at higher temperatures of 70° to 73° F (21° to 23.8° C).
Treatment
Topical treatment
Miconazole can be used as a sole therapeutic agent or used together in combination with chlorhexidine. Generally it is used twice weekly as an adjunct topical to systemic therapy rather than as a sole treatment method. As with all medicated shampoos and dips, for optimal therapeutic effects a skin contact time of 10 minutes is recommended for miconazole. Synergism between miconazole and chlorhexidine has been confirmed, and shampoos with this combination have been shown to hasten mycological cure. A list of topical antifungal therapy is included in Box 17-1.
Systemic treatment
Systemic therapy of dermatophytosis is used to hasten resolution of infection. Several drugs have been shown effective in the treatment of dermatophytosis. The appropriate choice for treatment is based on fungal species present, patient species involved, age and size of the patient involved, possible adverse effects and toxicity, and cost. Dogs and cats with multifocal lesions, all long-haired animals, and those in multianimal situations are candidates for systemic antifungal treatment. Animals nonresponsive following 2 to 4 weeks of topical therapy should also be considered for systemic treatment. See Box 17-2 for dermatophyte treatment summary. Itraconazole is currently the systemic treatment of choice for dermatophytosis in dogs and cats. Griseofulvin and ketoconazole both have significant issues of safety and efficacy. Box 17-3 and Table 17-2 list systemic fungal medications and antifungal drug dosages.
BOX 17-2 Current dermatophyte treatment summary
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree