Chapter 72The Suspensory Apparatus
Proximal Suspensory Desmitis in the Forelimb
Diagnostic Analgesic Techniques
If PSD is suspected, perineural analgesia of the lateral palmar nerve using lateral3 or medial15 approaches (2 mL mepivacaine) or the medial and lateral palmar metacarpal nerves (2 mL per site) is indicated (see Chapter 10). This should result in substantial improvement in, or alleviation of, lameness within 10 minutes, assuming PSD is the only cause of lameness. However, neither technique is necessarily specific. Blockade of the lateral palmar nerve also has the potential to alleviate pain associated with a lateral source of pain in the more distal aspect of the limb (e.g., a splint). The risks of influencing middle carpal joint pain are less than with the subcarpal approach, but with the lateral approach local anesthetic solution may diffuse and improve lameness associated with the middle carpal joint16 or with the carpal canal. Perineural analgesia of the palmar metacarpal nerves may alleviate pain associated with the middle carpal or carpometacarpal joints because of local diffusion or inadvertent deposition of local anesthetic solution into the distopalmar outpouchings of the carpometacarpal joint capsule. There is no right or wrong method, but it is important to be aware of the limitations of whichever technique is used. One author (SJD) usually blocks the medial and lateral palmar metacarpal nerves using a lateral approach, with the limb non–weight bearing. In a difficult, potentially dangerous horse that strikes out, the lateral palmar nerve is blocked with the limb bearing weight. It is easy to hit the lateral palmar nerve using the medial approach, causing the horse sudden severe pain; therefore this technique is rarely used. A false-negative result may be achieved because of inadvertent injection into the carpal sheath or failure of the local anesthetic solution to diffuse proximally to the most proximal extent of a lesion. Although the SL receives innervation from fibers from the median and ulnar nerves, perineural analgesia of the ulnar nerve usually resolves or substantially improves lameness associated with PSD. However, in a minority of horses perineural analgesia of the median and ulnar nerves is required to abolish lameness completely.
Differential Diagnosis
PSD should be differentiated from middle carpal joint pain, being aware that especially in young TB racehorses and STB racehorses lesions may occur in both locations simultaneously. Osteoarthritis of the carpometacarpal joint occasionally occurs (see page 417). Horses with pain associated with palmar cortical fatigue fractures or stress reactions of the McIII11,18-20 respond similarly to diagnostic analgesic techniques; however, in horses with fracture, lameness tends to be more severe and worse on firm ground and often deteriorates the farther the horse trots (see page 413). Avulsion fractures of the McIII at the origin of the SL (see page 417) occur less frequently and tend to be associated with more persistent and severe lameness.1,21 Pain associated with the carpal sheath or carpal retinaculum also should be considered (see Chapter 75). Perineural analgesia of the deep branch of the lateral palmar nerve or the palmar metacarpal nerves alone should not alleviate pain associated with the deep digital flexor tendon (DDFT) or its accessory ligament (ALDDFT), the superficial digital flexor tendon (SDFT), or the fetlock region, without simultaneous blockade of the palmar nerves. However, horses with proximal lesions of the SDFT or ALDDFT may show partial improvement in lameness.
Diagnostic Ultrasonography
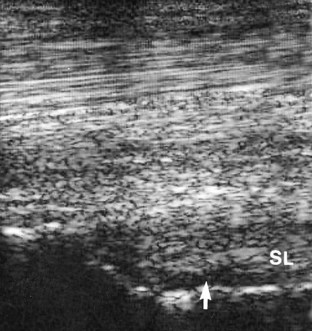
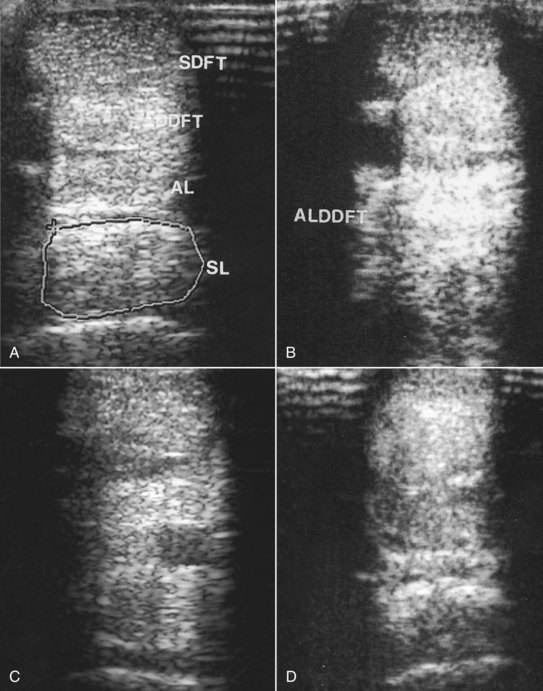
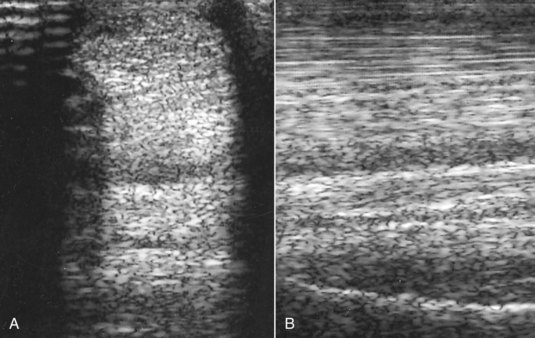
Diagnostic ultrasonography is essential for accurate diagnosis of PSD. The limb should be evaluated in transverse and longitudinal planes, and careful comparison should be made with the contralateral limb. High-quality images are required, because lesions can be subtle and easily missed if the gain controls are too high or if the transducer is not focused on the SL. Artifacts are readily created if the transducer is not in complete contact with the limb. The contours of the proximal palmar aspect of the metacarpal region can make obtaining longitudinal images difficult, especially because the proximal palmar aspect of the McIII slopes backward. Therefore creating hypoechoic artifacts at the enthesis of the SL on the McIII is easy. Cross-sectional area measurements may be extremely valuable, especially in horses with acute PSD, because enlargement of the ligament may be the only detectable ultrasonographic abnormality. Bear in mind that muscular tissue appears less echogenic than does ligamentous tissue and that proximally the SL originates in two halves. The entire cross-section of the SL cannot be seen from a palmar approach, and marginal lesions may be missed unless oblique images are also obtained. A convex transducer or virtual convex transducer allows a greater proportion of the proximal abaxial aspects of the SL to be evaluated in transverse images compared with a linear transducer. Previous injuries to the SL may not resolve fully to restore normal, uniform echogenicity. Be aware that poor diagnostic analgesic technique may result in aspiration of air, which creates artifacts. This usually resolves in 24 hours. A thin band of the SL passes proximally from the enthesis on the McIII to blend with the palmar carpal fascia. The anechogenic space seen dorsal to the SL at this level is fluid in the palmar recess of the carpometacarpal joint and should not be mistaken for a lesion (Figure 72-1).
Abnormalities associated with PSD include the following22 (Figures 72-2 and 72-3):
In horses with an avulsion fracture of the McIII at the origin of the SL, the fracture fragment is usually readily identifiable and generally is associated with only a focal lesion in the SL itself, usually restricted to the dorsal aspect (see page 417).
Proximal Suspensory Desmitis in the Hindlimb
PSD in the hindlimb occurs in horses in all athletic disciplines and of all ages and is a particular problem in dressage horses.36 There is an association between straight hock conformation and hyperextension of the metatarsophalangeal joint and hindlimb PSD (Figure 72-4). Such conformational abnormalities were identified in nine (21%) of 42 horses with hindlimb PSD but in only four (8%) of 50 horses examined consecutively with hindlimb lameness unrelated to the suspensory apparatus.37![]() Straight hock conformation may predispose to PSD or develop secondarily; hyperextension of the metatarsophalangeal joint may develop as a sequela to PSD, probably as the result of progressive degeneration of the SL. A long-toe and low-heel conformation also may be a predisposing factor, especially if associated with abnormal orientation of the distal phalanx, with the plantar aspect lower than the toe.29
Straight hock conformation may predispose to PSD or develop secondarily; hyperextension of the metatarsophalangeal joint may develop as a sequela to PSD, probably as the result of progressive degeneration of the SL. A long-toe and low-heel conformation also may be a predisposing factor, especially if associated with abnormal orientation of the distal phalanx, with the plantar aspect lower than the toe.29