CHAPTER 42 The Musculoskeletal System
Musculoskeletal Development
Most longitudinal bone growth takes place between 12 and 26 weeks of age. During this period, mineralization of all epiphyseal growth centers is completed at predetermined times, and growth plates have their most rapid rate of longitudinal bone growth and endochondral ossification. At the end of this period, epiphyseal bone growth is completed. Following this period, growth plate–associated bone length continues at a much slower rate until it ceases completely. At cessation of growth, growth plates fuse at predetermined times (Table 42-1). The rate of skeletal development and the final size of bones vary widely among canine and feline breeds. The optimal rate of skeletal development for a given breed is rarely known. An indicator for skeletal growth is body weight. Unfortunately puppy and kitten growth (weight) curves have only been established for a few breeds.
TABLE 42-1 Age when ossification centers appear and growth plate fusion occurs in immature dogs
| Anatomical site | Age when ossification center appears | Age when fusion occurs |
|---|---|---|
| Scapula | ||
| Body | Birth | |
| Tuber scapulae | 7 wk | 4-7 mo |
| Humerus | ||
| Diaphysis | Birth | |
| Proximal epiphysis | 1-2 wk | 10-13 mo |
| Distal epiphysis | 6-8 mo to shaft | |
| Medial condyle | 2-3 wk | 6 wk to lateral condyle |
| Lateral condyle | 2-3 wk | |
| Medial epicondyle | 6-8 wk | 6 mo to condyle |
| Radius | ||
| Diaphysis | Birth | |
| Proximal epiphysis | 3-5 wk | 6-11 mo |
| Distal epiphysis | 2-4 wk | 8-12 mo |
| Ulna | ||
| Diaphysis | Birth | |
| Olecranon | 8 wk | 6-10 mo |
| Distal epiphysis | 8 wk | 8-12 mo |
| Carpus | ||
| Ulnar | 4 wk | |
| Radial | 3-4 wk | |
| Central | 4-5 wk | |
| Intermediate | 3-4 wk | |
| Body | 2 wk | |
| Epiphysis | 7 wk | 4 mo |
| First | 3 wk | |
| Second | 4 wk | |
| Third | 4 wk | |
| Fourth | 3 wk | |
| Sesamoid bone | 4 mo | |
| Metacarpus/Metatarsus | ||
| Diaphysis | Birth | |
| Distal epiphysis (2-5)* | 4 wk | 6 mo |
| Proximal epiphysis (1)* | 5 wk | 6 mo |
| Phalanges | ||
| First Phalanx | ||
| Diaphysis (digits 1-5) | Birth | |
| Distal epiphysis (digits 2-5) | 4 wk | 6 mo |
| Distal epiphysis (digit 1) | 6 wk | 6 mo |
| Second Phalanx | ||
| Diaphysis (digits 2-5) | Birth | |
| Proximal epiphysis (digits 2-5)* | 5 wk | 6 mo |
| Third Phalanx | ||
| Diaphysis | Birth | |
| Volar sesamoids | 2 mo | |
| Dorsal sesamoids | 4 mo | |
| Pelvis | ||
| Pubis | Birth | 4-6 mo |
| Ilium | Birth | 4-6 mo |
| Ischium | Birth | 4-6 mo |
| Os acetabulum | 7 wk | 5 mo |
| Iliac crest | 4 mo | 1-2 yr |
| Tuber ischii | 3 mo | 8-10 mo |
| Ischial arch | 6 mo | 12 mo |
| Caudal symphysis pubis | 7 mo | 5 yr |
| Symphysis pubis | 5 yr | |
| Femur | ||
| Diaphysis | Birth | |
| Proximal epiphysis (head) | 2 wk | 7-11 mo |
| Trochanter major | 8 wk | 6-10 mo |
| Trochanter minor | 8 wk | 8-13 mo |
| Distal epiphysis | 8-11 mo to shaft | |
| Trochlea | 2 wk | 3 mo condyle to trochlea |
| Medial condyle | 3 wk | |
| Lateral condyle | 3 wk | |
| Patella | 9 wk | |
| Tibia | ||
| Diaphysis | Birth | |
| Medial condyle | 3 wk | 6 wk to lateral |
| Lateral condyle | 3 wk | 6-12 mo to shaft |
| Tuberosity | 8 wk | 6-8 mo to condyle |
| 6-12 mo to shaft | ||
| Distal epiphysis | 3 wk | 8-11 mo |
| Medial malleolus | 3 mo | 5 mo |
| Fibula | ||
| Diaphysis | Birth | |
| Proximal epiphysis | 9 wk | 8-12 mo |
| Distal epiphysis | 2-7 wk | 7-11 mo |
| Tarsus | ||
| Talus | Birth-1 wk | |
| Fibular | Birth-1 wk | |
| Tuber calcis | 6 wk | 3-8 mo |
| Central | 3 wk | |
| First | 4 wk | |
| Second | 4 wk | |
| Third | 3 wk | |
| Fourth | 2 wk | |
| Sesamoids | ||
| Fabellar | 3 mo | |
| Popliteal | 3 mo | |
| Plantar phalangeal | 2 mo | |
| Dorsal phalangeal | 5 mo |
* Second phalanx absent or fused with first phalanx in first digit.
Modified from Owens JM: Radiographic interpretation for the small animal clinician, St. Louis, Ralston Purina Company, 1982, p 8; and Ticer JW: Radiographic technique in small animal practice, Philadelphia, 1975, Saunders, p 101.
Orthopedic Examination
The Young Dog with Hindlimb Lameness
Most clinicians start their orthopedic examination distally and proceed proximally. An overview of common causes for hindlimb lameness in pediatric dogs can be found in Box 42-1. Lameness isolated at the level of the foot may result from foreign bodies or lacerations of the digital pads or the tarsal pad, subluxated interdigital joints, paronychia, and sesamoiditis/fractured sesamoid bones. Lameness localized at the metatarsus includes fractures, ruptured flexor tendons (if the toes are off the ground), or some other soft tissue injury (like a failure of the flexor and retinacular support of the caudal distal hock joints or tarsal luxation). The latter condition will result in roundness of the caudal distal hock and proximal metatarsus with an overflexed hock. Tarsocrural luxation would result in non–weight-bearing lameness. All the normal bony protuberances around the hock should be palpable, and there should be no swelling or effusion of any joint. Hock swelling and effusion in a young dog could indicate talar osteochondrosis (OC) or infectious or inflammatory arthritis. Dogs with infectious arthritis will usually be extremely lame. Tibial distal metaphyseal bone pain and swelling, especially in a large or giant breed dog, could indicate hypertrophic osteodystrophy. Tibial diaphyseal bone pain on digital pressure could indicate panosteitis. German Shepherd Dogs are predisposed to panosteitis, but any large or giant breed dog can succumb. Lameness associated with stifle pain and effusion could be associated with rupture of the cranial (or caudal) cruciate ligament, patellar luxation (medial or lateral), infectious arthritis, patellar luxation, or OC of the femoral condyles. Femoral diaphyseal pain could indicate panosteitis. The most common cause of lameness in the hindlimbs or hindlimb dysfunction in the young or growing dog is synovitis and osteoarthritis secondary to hip dysplasia. Pain on hip extension or a positive Ortolani test indicates hip dysplasia and secondary osteoarthritis. The Ortolani maneuver detects the palpable “click” of femoral head reduction into the acetabulum. Mild pain on hip extension also may be associated with iliopsoas muscle trauma. Severe pain during hip extension with simultaneous internal rotation of the hip joint is further indication of this condition. Infectious arthritis should always be on the list of differentials in an immature dog with marked lameness and joint pain or effusion. In small breeds, Legg-Calvé-Perthes disease or avascular necrosis of the femoral head will present as a mostly unilateral lameness that develops during growth and worsens with time. In cats with hip pain, slipped capital femoral epiphysis (SCFE) or hip dysplasia should be considered. Lumbosacral malformation or hemivertebra could also result in neurologic impingement and secondary hindlimb weakness or lameness. Neurologic conditions are discussed in greater detail in Chapter 40.
BOX 42-1 Hindlimb lameness
| Source of Lameness | Possible Etiologies | Diagnostic Tests |
|---|---|---|
| Isolated in the foot | Foreign bodies or lacerations of the digital pad and tarsal pad, subluxated interdigital joints, paronychia, sesamoiditis/fractured sesamoid bones | Palpation and radiography |
| Localized in the metatarsus | Fractures, ruptured flexor tendons (if the toes are off the ground) or some other soft tissue injury | Roundness of the caudal distal hock and proximal metatarsus with an overflexed hock |
| Hock swelling and effusion | Tarsal osteochondrosis or infectious or inflammatory arthritis | Radiography and synovial fluid analysis |
| Tibial distal metaphyseal bone pain and swelling | Hypertrophic osteodystrophy | Palpation and radiography |
| Tibial diaphyseal bone pain | Panosteitis | Digital pressure |
| Radiography | ||
| Stifle pain and effusion | Rupture of the cranial (or caudal) cruciate ligament, patellar luxation | Palpation and radiography |
The Young Dog with Forelimb Lameness
Similar conditions of the digits and metacarpus occur in both the lower forelimbs and hindlimbs. An overview of common causes for forelimb lameness in pediatric patients can be found in Box 42-2. Angular limb deformities will be a cause of lameness more often in the forelimb than in the hindlimb. Angulation can be observed at the carpus in either a valgus or a varus direction, resulting in carpal pain on flexion from effusion and secondary osteoarthritis. Elbow subluxation as a result of premature closure of the distal ulnar physis is particularly debilitating and will be accompanied by effusion of the elbow joint, restricted range of motion, and sometimes crepitation. Distal metaphyseal bone pain and swelling indicate hypertrophic osteodystrophy in large and giant breed dogs. Radial diaphyseal pain on pressure indicates panosteitis. Panosteitis can also occur in the proximal ulna and humerus, especially around the region of the nutrient foramen. Shoulder pain in young dogs could indicate OC of the humeral head, shoulder instability, or biceps tendonitis.
BOX 42-2 Forelimb lameness
| Source of Lameness | Possible Etiologies | Diagnostic Tests |
|---|---|---|
| Carpal pain on flexion | Effusion or secondary osteoarthritis | Angulation at the carpus in either valgus or varus direction |
| Elbow subluxation | Premature closure of the distal ulnar physis | Effusion of the elbow joint, restricted range of motion, and sometimes crepitation; radiography |
| Distal metaphyseal bone pain and swelling | Hypertrophic osteodystrophy in large and giant breeds | Palpation and radiography |
| Radial diaphyseal pain | Panosteitis | Palpation and radiography |
| Shoulder pain in young dogs | Osteochondrosis of the humeral head, shoulder instability, or biceps tendonitis | Palpation and radiography |
Congenital Musculoskeletal Diseases
Retarded Growth
Many patients presenting with a congenital musculoskeletal condition display retarded growth. Retarded growth of a puppy or kitten may be defined as reduced growth in terms of skeletal development or body weight when compared with normal littermates or a failure to attain the weight and/or height standards characteristic of a given breed. Selected puppy and kitten growth curves, as well as weight and/or height standards, can be found in Chapter 5 for some dog and cat breeds. Abnormal prenatal growth may affect the size of newborns and results in so-called “runts.” Runts may not attain normal adult size because of inadequate compensatory growth after birth. Many diseases may result in retarded growth (Table 42-2).
TABLE 42-2 Classification of growth retardation
| Osteochondrodysplasia | |
| Endocrinopathy | |
| Congenital disorders of cell metabolism | |
| Nutritional deficiency | |
| Chronic inflammation | |
| Congenital or acquired major organ failure or insufficiency |
Modified from Lorenz MD: Retarded growth. In Lorenz MD, Cornelius LM (eds): Small animal medical diagnosis, Philadelphia, 1993, JB Lippincott, pp 83-90.
Osteochondrodysplasia
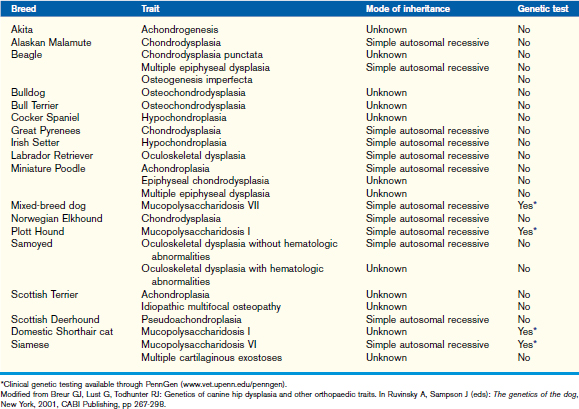
Most patients with osteochondrodysplasias have reduced skeletal growth, characterized by abnormal endochondral and/or intramembranous ossification. The diagnosis of osteochondrodysplasias is based on the patient’s signalment (breed), history (identifiable at birth or later in life, family, and nutrition history), clinical findings (proportionate or disproportionate, mentation), CBC, serum chemistry, urine analysis, endocrine evaluation as recommended for retarded growth, radiography (skull, appendicular and/or axial skeleton, epiphyseal growth centers, and/or growth plates), and growth plate histopathology (rib resection, percutaneous growth plate biopsy, or postmortem specimen) (Table 42-3). Chondrodystrophic dogs likely have an osteochondrodysplasia that can result in a tendency for relatively long trunks and short legs.
Dysostosis
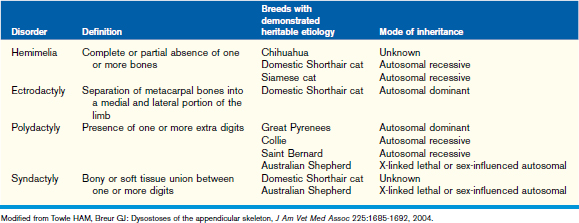
Dysostoses may occur in the skull, axial, and/or appendicular skeleton. Diseases of mesenchymal bone formation in the appendicular skeleton are characterized by overrepresentation or a partial or complete absence of one or more bone elements. Patients usually present with a limb deformity and a severe, often non–weight-bearing lameness. The disease may be bilateral. Dysostoses can be diagnosed using radiography of the affected body region. Dysostoses of the appendicular skeleton may be subclassified as amelia, dimelia, hemimelia, ectrodactyly, polydactyly, and syndactyly (Table 42-4). The most commonly seen dysostosis is radial hemimelia. Treatment may be conservative (bandage or splint to prevent muscle contractures and bone deformities during rapid growth) or surgical (amputation, bone repair, or arthrodesis).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree