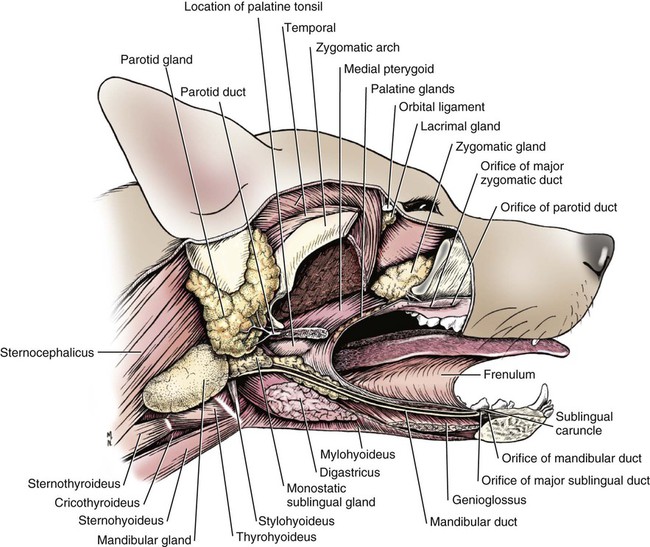
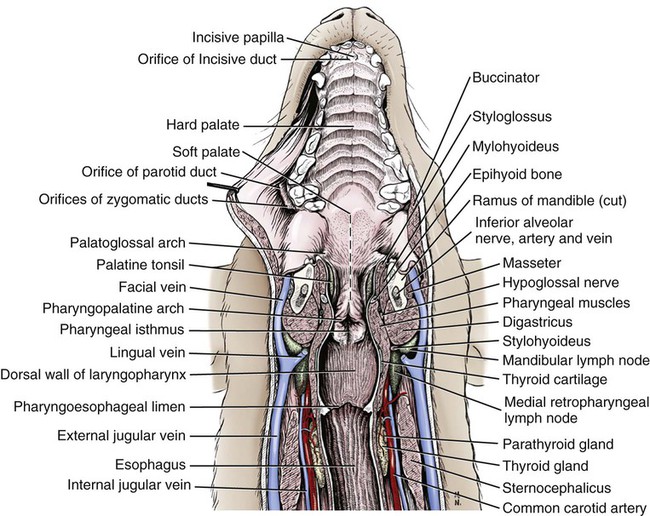
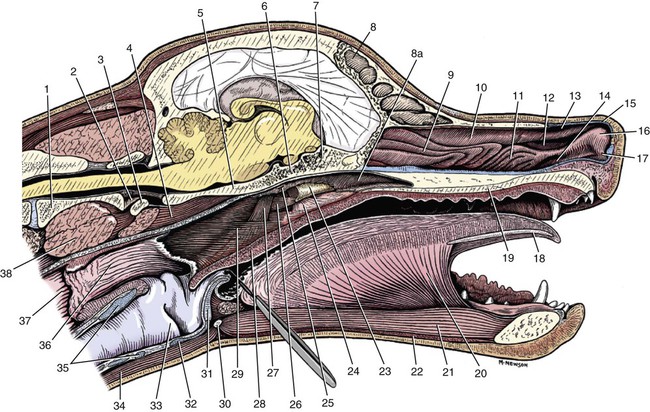
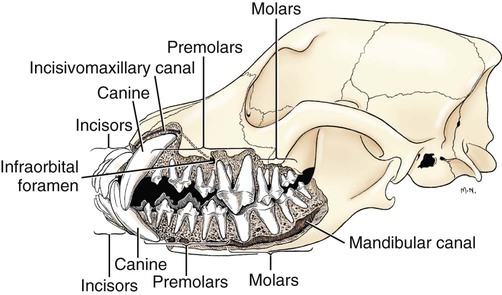
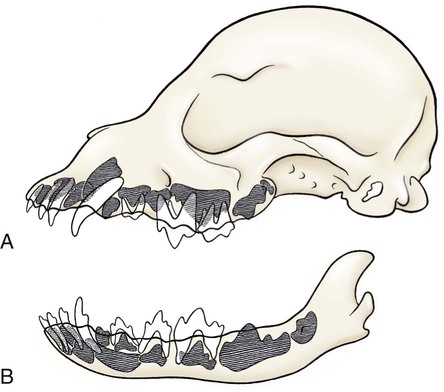
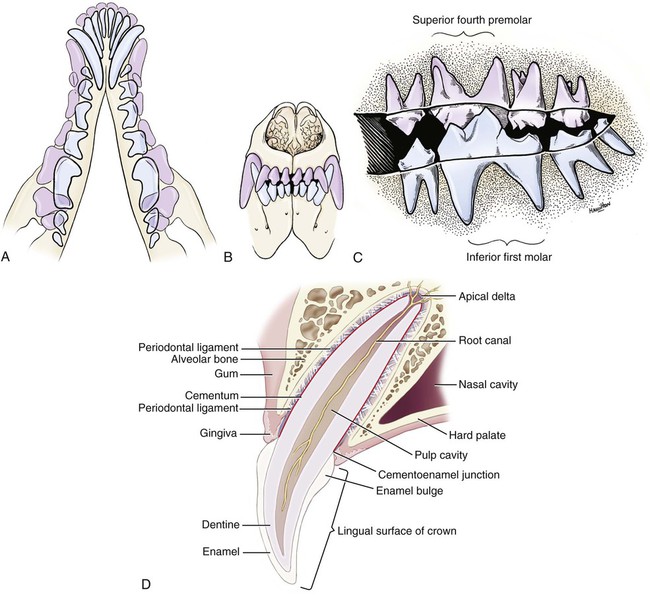
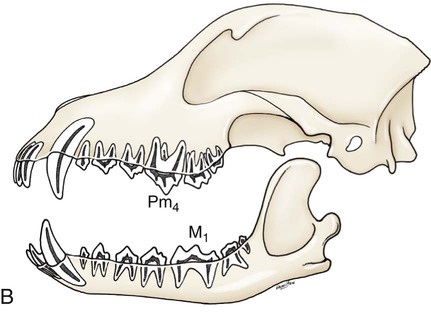
The parotid and zygomatic salivary ducts open into the dorsocaudal part of the vestibule. The parotid duct opens through the cheek on the small parotid papilla (papilla parotidea), located opposite the caudal part of the superior fourth premolar tooth, approximately 5 mm from the fornix of the vestibule, which is formed by a reflection of the mucosa from the cheek to the gum. The main duct of the zygomatic gland opens lateral to the caudal part of the superior first molar tooth on a small papilla near the vestibular fornix (Fig. 7-1). A small mucosal ridge connects the main zygomatic and parotid duct openings. Usually one to four small accessory ducts from the zygomatic gland open caudal to the main duct. The submucosal labial and ventral buccal glands are few and are confined to the inferior lip and the adjacent part of the cheek. The secretion of these glands is discharged through approximately 10 openings located opposite the four inferior premolar teeth near the fornix of the vestibule. The oral cavity is continuous caudally with the isthmus of the fauces and with the oral pharynx. The palate (palatum) (Fig. 7-2) is a partly bony, partly membranous partition separating the respiratory and digestive passages of the head. The nasal cavities and nasal pharynx lie above it; the oral cavity and oral pharynx lie below it. The bony hard palate is rostral, and the membranous soft palate caudal. The hard palate (palatum durum) is formed by processes of the palatine, maxillary, and incisive bones on each side. The mucosa on the nasal side consists of pseudostratified ciliated columnar epithelium; that on the oral side consists of stratified squamous epithelium that is cornified. The hard palate is nearly flat. Laterally and rostrally it inclines slightly ventrally, and it is continuous with the portions of the incisive and maxillary bones that contain the alveoli of the superior teeth. Six to ten ridges and depressions cross it transversely on the oral side. Not all of these ridges are complete. In extremely brachycephalic heads they become nearly straight. Close inspection of the ridges reveals small blunt eminences. The muscles of the soft palate consist of the paired palatine muscles (mm. palatini) and the end ramifications of the paired tensor and levator veli palatini muscles (Fig. 7-3), which are nearly equal in size. These muscles are described with the muscles of the head. The right and left intrinsic palatine muscles lie close together on each side of the median plane, ventral to the palatine aponeurosis. The end ramifications of the paired extrinsic muscles, the levator and tensor veli palatini, blend with the palatine aponeurosis. The right and left pterygopharyngeal muscles pass lateral to the caudal part of the soft palate from origins on the pterygoid bones. Right and left palatopharyngeal muscles arise near the median plane from the palatine aponeurosis. They sweep laterally and dorsocaudally in the pharynx, forming the bases for the palatopharyngeal arches. The teeth (dentes) (Fig. 7-4) are highly specialized structures that serve for the procuring, cutting, and crushing of food as well as for social interaction. Each tooth is divided into three parts. The crown (corona dentis) is the exposed portion of tooth that extends beyond the gums (gingiva) and is covered by a thin layer of white enamel. With the exception of the canine teeth, all crowns in the dog end in tubercles (tubercula dentis). Below the enamel bulge, a circumferential widening at the base of every crown, lies the neck (cervix dentis) of the tooth. The neck includes the most coronal region of the root between the enamel bulge and the attached gingiva. The cementoenamel junction is located within the neck. The root (radix dentis) is the portion of the tooth located beyond the level of the attached gingiva, embedded in alveolar bone. The root tip is termed the apex of the root (apex radicis dentis). Many teeth have more than one root. A dog’s dentition is diphyodont, meaning there are two sets of teeth that develop sequentially. Dechambre (1912) reported total absence of teeth in a dog. Once teeth are fully erupted in the dog they cease growing. The first set is fully erupted and functional early in the second month after birth (Fig. 7-5). These teeth, known as deciduous teeth (dentis decidui), serve the animal during its most active puppyhood. Lawson et al. (1967) described the development and eruption of teeth and illustrated several stages by a series of radiographs. They found that eruption of deciduous teeth begins on approximately the twentieth day and is completed by the thirty-fifth day. Esaka (1982) studied the development of premolar tooth germs and their rotation by means of radiographs, dissections, serial sections, and reconstructions. He found that the direction of rotation of the teeth varied along the arcade. His accompanying table showed the rotation and contact relationship of permanent tooth germs with deciduous teeth from birth to 6 months. Upon approaching maturity at 6 months of age, when the bones that contain the teeth have become larger, the small deciduous teeth are no longer adequate; they are shed (exfoliated) and replaced by the permanent teeth (dentes permanentes) (Fig. 7-6), which last throughout adult life. Permanent teeth are larger and stronger than deciduous teeth. As the maxilla and mandible continue to grow over the next two to three months permanent molar teeth erupt caudal to the premolar teeth. There are two molar teeth in each maxilla and three in each mandible. In an experimental study of diet and teeth, Mellanby (1929) began with a detailed review of dental structure in dogs that included the development of the teeth. Observations on the development of the deciduous teeth in the fetal dog have been reported by Hörmandinger (1958), Satrapa-Binder (1959), Williams (1961), and Williams and Evans (1978). Postnatal calcification and eruption of the deciduous and permanent teeth have been studied by Meyer (1942), Höppner (1956), Arnall (1961), and Kremenak (1967). Cahill and Marks (1982) studied exfoliation of the third deciduous mandibular premolar in mongrels and purebred Beagles from the thirteenth to the twenty-seventh postnatal week. They used radiographs and histologic sections to correlate bone, tooth, and soft-tissue events. The timing and sequence were the same in Beagles and mongrels. Eruption of the permanent third premolars began during the sixteenth postnatal week and was completed in 7 weeks. The central and middle deciduous incisors and the canine teeth of both superior and inferior arcades have usually erupted by the end of the first month. The lateral incisors erupt during the fifth or sixth week: the deciduous premolars between the fourth and eighth weeks. All permanent teeth, with the exception of the superior canine teeth, erupt lingual to their deciduous counterparts. The permanent superior canine teeth erupt mesially. Teeth erupt earliest in the large breeds. Table 7-1 gives the normal range of time for the eruption of each of the permanent teeth, which erupt at approximately the same time in each arcade. Eruption of the permanent canine teeth can slightly precede the shedding of the corresponding deciduous teeth. TABLE 7-1 Tetracycline antibiotics concentrate in growing bones and teeth and can inhibit bone growth or interfere with enamel formation. Because they fluoresce yellow or orange under ultraviolet light, they have been used to study incremental growth of bones and teeth. Owen (1963) studied the effects of tetracycline ingestion on the teeth of dogs and noted that dentine, cementum, and enamel can all incorporate the fluorescent component. Therefore the use of tetracycline or its derivatives is discouraged in animals younger than 6 months of age. The teeth are arranged as superior (maxillary) and inferior (mandibular) dental arches (arcus dentalis superior et inferior). The inferior arch is anisognathic, narrower and shorter than the superior arch; therefore superior incisors are located slightly rostral to the inferior incisors. Incisors function mainly to nibble or nip during mastication, grooming, and social interactions. From the superior third incisor to the superior fourth premolar, superior and inferior teeth alternate in position along the dental arch resulting in a scissors bite. Canine teeth are used for puncturing and grasping. Stockard (1941) referred to the area between the canine and carnassial teeth as the “premolar carrying space,” in reference to hunting breeds. Overlap of the carnassial teeth, the superior fourth premolars, and the inferior first molars allows for optimal shearing of food. Direct occlusal contact of the superior and inferior molars provides for grinding of food (Fig. 7-7A). In dogs, food is mostly swallowed without mastication and complete occlusal contact is not necessary. Wood and Wood (1933) commented on the genetic and phylogenetic significance of a third superior molar in the modern dog. The dense, pearly-white outer layer of the crown is enamel (enamelum). It is the hardest substance in the body, being 96% inorganic, composed of millions of crystals of hydroxyapatite. It cannot regenerate when damaged. It is thickest on the occlusal surfaces of the teeth, and its hardness gradually increases in the first year of life. Skobe et al. (1985) studied the ultrastructure of dog enamel and found three layers to be present: a rodless (aprismatic) surface layer, a middle layer of parallel rods that was not constant at all sites, and an inner layer with prominent Hunter-Schreger bands. Enamel rods of the dog tooth, as seen after acid etching of the surface, appear to be primarily hexagonal, although semicircular and spiral shapes are also present. The enamel rod is a column of mineral that extends from the dentinoenamel junction to the coronal surface of the tooth. They are perpendicular to the surface and each rod has two parts: a core of hydroxyapatite and a sheath of organic fibrous substance. A study of dental enamel in dogs was made by Glock et al (1942). The pulp (pulpa dentis), is soft tissue contained in a tooth (see Fig. 7-4). It is composed of sensory nerves, arteries, veins, lymphatic capillaries, and a connective tissue that holds those structures together. The pulp is contained in the pulp cavity (cavum dentis) of the crown and in the root canal of the root. An apical delta at the apex of each root consists of multiple small channels that allow free passage of the vessels and nerves in and out of the root canal (canalis radicis dentis). The superior teeth are attached in the alveoli of the incisive and maxillary bones. Those with roots embedded in the incisive bones are the incisor teeth (dentes incisivi). The incisor tooth nearest the midplane on each side in each incisive bone is incisor 1, the central incisor. The second is incisor 2, the middle incisor, and the third is incisor 3, the lateral incisor. They are long slender teeth, arched slightly rostral and laterally compressed. Superior incisors increase in size from the central to the lateral incisor. The crowns of the superior lateral incisors are largest and slightly hooked caudally with its conformation more anatomically similar to a small canine tooth. The superior central and middle incisors have three tubercles each. Of the three incisal tubercles, the central one is largest and extends farthest coronally; the small mesial and distal tubercles are called mamelons, and a V-shaped ridge of crown with its apex nearest the gingiva, connects the side tubercles on the lingual surface. The lingual crown enlargement at the most apical aspect of the superior central and middle crowns is termed the cingulum, and when the mouth is closed the inferior incisor occlusal surfaces rest on the cingulum. Inferior incisors are similar in size and shape to the superior central and middle incisors. Depending on the occlusion and chewing habits of the dog, the tubercles and crown may slowly wear throughout life. The canine teeth (dentes canini) are separated from the lateral superior incisors by an interdental space matching the width of the inferior canine tooth (approximately 4-10 mm) and from the lateral inferior incisors by a space of roughly 1-3 mm. The canine teeth are by far the longest teeth in the dog having large roots that are nearly two times as long as their crowns. All four canine teeth are similar in length and width. They are transversely compressed with an oval cross-section. When the mouth is closed, the crown of the inferior canine tooth occupies the interdental space between the superior lateral incisor and the superior canine tooth (Fig. 7-7B). The superior roots produce an arciform alveolar juga and all four canine roots are located adjacent to the root apices of the first and second premolar teeth. Canine tooth roots are slightly wider in the midalveolar region and taper down to a rounded apex. Whereas the superior canine tooth root runs parallel to the maxillary bone, the inferior canine tooth root crosses the width of the rostral mandible making extraction challenging. The size, shape, and course of canine tooth roots necessitate a surgical approach for extraction unless significant alveolar bone loss has occurred. Lorber et al. (1979) found differences between male and female canine teeth in the dog. In the male, crowns were 23% longer, and roots were 40% longer than in females. Likewise, in the male, the width of the crown of the canine tooth was 26% wider, and the width of the root was 50% wider. The smaller crown-to-root ratio in males, together with greater root width, indicated to the authors a difference in the tooth anchorage mechanism. All teeth caudal to the canines are often referred to as the “cheek teeth” (St. Clair & Jones, 1957). They are divided into premolars and molars. In the permanent dentition, there are four premolar teeth (dentes premolares) in each of the four dental quadrants. There are no deciduous first premolar or molar teeth. The first premolar erupts between the fourth and fifth postnatal month and usually remains throughout life. The placement of the premolar teeth may be altered by changing the shape of the head through selective breeding, but regardless of head shape the teeth remain relatively constant in form and size (Stockard, 1941). This leads to tooth crowding in smaller breeds, which predisposes them to periodontal disease. The first premolar is the smallest, and has a single root. The fourth premolar is the largest. In the superior arcade, it has three roots—mesiovestibular, mesiolingual, and distal—and in the inferior arcade only two roots—mesial and distal. The second and the third premolar teeth are similar in all quadrants. Each premolar has two roots, mesial and distal. The crowns of the first three premolar teeth are similar in all quadrants. All three have a singular pyramidal shape. The first premolar tooth has one tubercle. The second and third premolars have similar crowns with an additional small tubercle on the distal aspect. The enamel bulge at the neck of the tooth is more prominent on the lingual surface (Fig. 7-7). On each tooth the mesial border slopes slightly distal. The distal border of the largest tubercle is steep. Adjacent to the base of the main tubercle on this border is a second smaller tubercle 1 or 2 mm high. The most distal surface of the crowns of the second and third premolars can be irregular with additional small tubercles located in this region. The superior fourth premolars and the inferior first molars are the largest shearing teeth in the mouth. They are the carnassial or sectorial teeth (dens sectorius) (Fig. 7-7C). The superior fourth premolar teeth have three stout diverging roots. The mesiovestibular and mesiolingual roots are more slender than the wide distal root. The space between all roots is referred to as the furcation, which is normally filled with interradicular bone. The mesiovestibular and distal roots form prominent alveolar juga that end just ventral to the ventral border of the infraorbital canals. The distal root of each superior fourth premolar tooth is triangular and somewhat transversely flattened, becoming wide at the neck. The mesiolingual root is flattened in an oblique plane; a small tubercle extends lingually off the mesial developmental ridge of the crown indicating the position of this root. According to Annis (1974), the superior fourth premolar is the tooth most commonly involved with root abscesses. An abscess of one of the roots can manifest by either the formation of a suborbital swelling on the face rostroventral to the eye with or without a fistula or by fistula formation within the mouth at the mucogingival junction. A permanent cure involves extraction or endodontic therapy of the affected tooth. The fourth premolar tooth in the superior arcade is similar in form to the two teeth mesial to it but it is larger. A developmental groove separates the largest mesial tubercle from a more prominent distal tubercle. The inferior first molar tooth is greater than twice the size of the second and third molar teeth. The distal third of the first molar inferior teeth is adapted for crushing and grinding; the mesial portion is sharp and pointed, and is well-suited for shearing. The shearing portion of each inferior first molar possesses the largest tubercle of any inferior cheek teeth and it is roughly quadrilateral in form. A small fossa formed in the hard palate mucosa slightly lingual to the midportion of the superior fourth premolar tooth receives the coronal tip of this large tubercle when the mouth is closed. For a discussion of the terminology used for tooth cusps and crests, see Szalay (1969) and Every (1972, 1974). Szalay uses a modified Cope-Osborn terminology for mammalian tritubercular cusps and crests, whereas Every proposes a new term for each.
The Digestive Apparatus and Abdomen
Oral Cavity
Vestibule
Oral Cavity Proper
Palate
Structure of the Soft Palate
Palatine Muscles
Teeth

GROUP
TOOTH
ERUPTION PERIOD
Incisors
Central
2 to 5 months
Intermediate
2 to 5 months
Corner
Most breeds 4 or 5 months
Canine
5 or 6 months
Premolars
First
4 or 5 months
Second
6 months
Third
6 months
Fourth
4 or 5 months
Molars
First
5 or 6 months
Second
6 or 7 months
Third
6 or 7 months
Tooth Structure
Tooth Groupings
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


The Digestive Apparatus and Abdomen
Only gold members can continue reading. Log In or Register to continue