Chapter 26 The Biomechanics of the Equine Limb and Its Effect on Lameness
Box 26-1 provides short definitions of biomechanical terms used in this chapter.
BOX 26-1 Definition of Common Terms in Biomechanics
Functional Anatomy of the Horse Limb
Horse Limbs Function Like Pogo Sticks
If you tried to build a horse limb with children’s blocks, the limb would collapse, because it would be impossible to build the fetlock joint in a hyperextended position. However, in a live limb the digital flexor tendons and ligaments on the flexor side of the limb prevent failure. Tendons have elastic properties and act like rubber bands or springs, providing resistance against which the limb presses when it comes under load, thus resisting further hyperextension and preventing collapse (Figure 26-1). The tendons are stretched at the same time, thus storing elastic strain energy, which can be returned in elastic recoil. During each step energy is stored, and it is returned when the limb leaves the ground. Energy is carried forward from one step to the next, thus reducing work the muscles have to do and saving metabolic energy. Indeed, the muscles associated with the main contributors of this system—the suspensory ligament (SL), deep digital flexor tendon (DDFT), and superficial digital flexor tendon (SDFT)—either are not present at all (the SL and the accessory ligament of the DDFT [ALDDFT]) or are very short in relation to the tendons (e.g., the average length of the deep digital flexor muscle-tendon unit in a Thoroughbred is 77 ± 5 cm, more than 60% of which is tendon. The flexor muscles, being highly pennate and having short muscle fibers (1 cm in length), have limited capacity to change the length of the muscle-tendon unit when contracting. Approximately 7% of the energy stored in the tendons is released as heat, and during gallop the tendons of a galloping horse reach about 45°C.4 We have reasoned that increase in temperature may account for core lesions in equine SDFTs but found that although 45°C resulted in death of some cells, tendon cells may be resistant.5 Core lesions may result from hyperthermic damage of matrix components.5
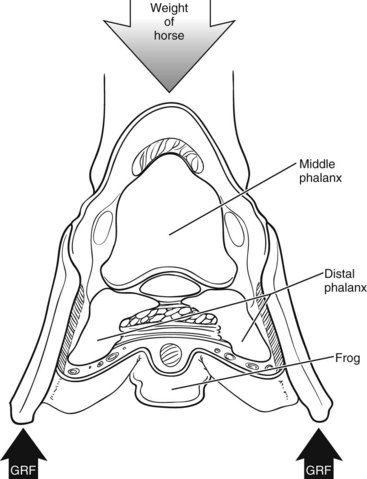
The Foot as Interface to the Ground
The foot has a series of “built-in” protective mechanisms that absorb part of the concussive forces and damp vibrations during impact (Figure 26-2):
Ability of Structures to Cope with Mechanical Demands
Structural and Material Properties
It is often of interest to know about the properties of the material per se, independent of size. This is achieved by dividing the force acting on a structure by its cross-sectional area. When normalized for cross-sectional area, a force is called stress (σ; common units would be mega Newtons per square meter [MN/m2] or Newtons per square millimeter [N/mm2]). The resulting deformation of an applied stress is expressed as strain (ε), the ratio of the change in size to the original size. Being a ratio, strain does not have a dimension; however, it is often expressed as a percentage—for example, ε = 0.1 = 10%. If we are interested in the property of a material rather than structure, we can express this relationship as a stress-strain curve. The slope of this curve is the ratio between the tensile stress and strain and is called the elastic modulus or Young’s modulus (E). The elastic modulus defines whether a material is “rigid” or “compliant.” Rigid materials have a very high elastic modulus and deform very little under load, whereas compliant materials have a low elastic modulus and require less load to deform. Bone, for example, is relatively rigid, whereas articular cartilage is more compliant and thus is able to act as an excellent shock absorber by undergoing considerable deformation when under load. Figure 68-1 shows the stress-strain curves for the digital flexor tendons of a Thoroughbred racehorse.
The ability of structures to deform when loaded and return to their original length when the load is removed allows them to store energy. The amount of energy per unit volume is the area under the linear portion of the stress-strain curve. The capacity of a material to absorb and release energy is often referred to as elastic resilience. The energy a material can absorb before failure defines whether it is “brittle” or “tough”: tough materials are able to absorb considerable elastic energy before failing, whereas brittle materials absorb very little. The digital flexor tendons of a horse are able to store and return a considerable amount of energy during locomotion. Load-deformation curves of tendons are different between loading and unloading, forming a “hysteresis” loop. The area of the loop presents the loss of energy, largely in the form of heat, that occurs during stretch and release (see Figure 68-2). It has been shown that the heat produced by repetition of this mechanism leads to an increase in core temperature, which may contribute to the pathogenesis of tendon injury through thermal damage.4 A detailed description of the pathobiology of tendon injuries is given in Chapter 68.
From an injury perspective, stress is probably the most informative mechanical measure in addition to strain. Equine digital flexor tendons, for example, have different elastic moduli and experience different strain rates in vivo, but they also have different cross-sectional areas. Both the SL and the SDFT experience high strains (up to 16%) during locomotion, but the stress in the SDFT is much higher than in the SL because its cross-sectional area is only about a third of that of the SL.11 This corresponds to the fact that the SDFT is the most commonly injured tendon or ligament (see Chapter 68).