(other sum for less than 2 kg) can be used to calculate daily resting energy expenditure. The metabolic energy requirement (MER) is the amount of calories required for an animal in a thermoneutral environment with basic activity to obtain, digest and absorb foodstuffs. The IER is the energy requirement associated with illness, injury, infection and inflammation. The illness energy requirement (IER) is an arbitrary number multiplied by the RER to combat the proposed increase in caloric requirements associated with various forms of illness. However, studies have found no increase in RER in numerous hospitalised patients, so this value is generally the value selected when calculating the patient’s nutritional requirements. This is thought to occur because of the down-regulation of metabolism and associated euthyroid sick syndrome that often occurs with critical illness. Overfeeding, particularly carbohydrates, can contribute to respiratory acidosis and increased patient morbidity. Overfeeding early in the course of illness, particularly after a long period of anorexia or weight loss, can result in hyperalimentation and ‘re-feeding syndrome’. Because of this, nutritional assessment of each patient should occur on an individual basis, depending on the patient’s primary illness, the expected time that the patient will need nutritional supplementation, the patient’s tolerance to enteral or parenteral feeding, anaesthetic risks and underlying illnesses including pancreatitis, gastric stasis, oesophageal motility disorders and severe diarrhoea.
Patient Selection
As a general guide, the earlier nutritional support is commenced the better, so wherever possible the need should be anticipated. All fluid and electrolyte deficits should be addressed first and then nutritional support should be introduced over the following 2–3 days. Nutritional support should be started if the patient exhibits any of the following:
- Recent weight loss of more than 10% body weight, not due to dehydration. This is relevant even in obese animals. It should be remembered that fluid gains and losses will interfere with the assessment of weight changes so this should be borne in mind when performing the nutritional assessment.
- Partial or complete anorexia for more than 3 days, as enterocyte atrophy occurs within 48 hours of anorexia and lack of trophic stimuli within the gut lumen. Early enteral nutrition has been shown to decrease patient morbidity in patients with parvoviral enteritis. Within 24 hours of hospitalisation, fluid, acid–base and electrolyte balance should be normalised, and exogenous nutritional support should commence in any anorexic patient or patient that is not tolerating oral feeding (see Figure 16.1). This includes animals in catabolic states, e.g. patients with severe burns, draining sepsis such as pyothorax, neoplasia, major surgery or severe trauma, malabsorption (see Figure 16.2).
Figure 16.1 Staffordshire Bull Terrier post badger attack, where it was anticipated the patient would be unlikely to eat for more than 3 days, so an oesophagostomy tube was placed at the time of surgery.

How Much Food to Give?
The patient’s daily food requirement should be calculated using resting (or basal) energy requirement (RER):


Previously, we would multiply the patient’s RER by an ‘illness’ factor which varied depending on the severity of the disease. We no longer do this as it has been shown that you risk complications if you overdo the calories in hospital.
What Food Should We Give?
Anorexic patients with concurrent injury, infection or neoplasia are effectively suffering from an accelerated form of starvation. In uncomplicated starvation, food deprivation results in a decrease in blood glucose which stimulates reduced levels of insulin and increased glucagon secretion. This reduction in insulin leads to a reduction in the insulin-responsive conversion of T4 to the more active T3 which results in a lowering of RER. The increased glucagon causes hepatic glyconeogenesis and glucose release: hepatic gluconeogenesis from amino acids, lactic acid and glycerol; release of glycerol and fatty acids from fat stores (lipolysis facilitated by a reduction in insulin).
The consequences of these changes are as follow:
Initially, glucose is obtained from glycogen. This is rapidly used up and following on from this the main source is breakdown of body protein, so an initial rapid decline is seen in the first few days. Gradual changes over the following week whereby the body begins using stored fat as the main fuel. This occurs as an increase in blood ketone levels change enzymes in tissues including brain and heart to use ketone bodies more. Then, in late starvation, as fat stores are used up, there is a shift back to using body protein as the energy source. Additionally, some tissues are obligate glucose users and cannot use fat or ketones (due to low mitochondria or poor oxygenation – fat oxidation is mitochondrial and uses oxygen): red blood cells, renal medullary cells, nervous tissue, also fibroblasts for wound healing and some tumours.
Anorexic and sick animals suffer from an accelerated form of starvation; they differ from starving animals in that they are hyperdynamic. Anorexia in these animals starts the same metabolic changes but these are complicated by the neuroendocrine responses to stress (sympathetic nervous system stimulation, catecholamine, adrenocorticoid and growth hormone release) which override the normal down-regulation of RER that occurs in uncomplicated starvation, i.e. get reduced insulin secretion and increased glucagon but no reduction in the T4 to T3 conversion. Also, neuroendocrine changes make patients relatively insulin resistant, i.e. high dose glucose infusions will not completely stop gluconeogenesis in these animals whereas they may do in healthy patients.
Early feeding is vitally important. It helps immunity, wound healing and prevents the loss of lean body mass. In paitents with tumours, it may not increase survivial time but it does improve quality of life, with faster recovery after therapy and increased immunocompetence.
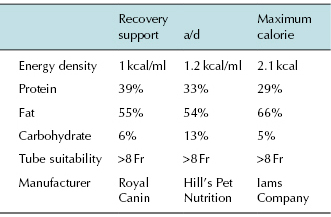
It is important not to feed a restricted protein food unless specific indication (hepatic encephalopathy or severe uraemia). Critical care diets specifically manufactured for cats and dogs should be used as human critical care foods are too low in protein (see Tables 16.1 and 16.2). The consistency of the diet is also very important. Very low viscosity foods should be used for naso-oesophageal tubes to minimise blockages. Thicker ‘gruel’ consistency foods can be used in larger tubes, such as gastrotomy tubes. The food selected should be iso-osmolar to prevent diarrhoea. The food should also be warmed, as cold food can induce either vomiting or rapid gastric emptying which may cause diarrhoea, If food is too cold, e.g. just removed from the refrigerator, the patent has to expend energy in warming it.
Table 16.1 Recommended levels of fats, proteins and carbohydrates in critical care diets

Table 16.2 Recommendations for avoiding complications of the re-feeding syndrome
Recommendations for Avoiding Complications of the Re-Feeding Syndrome
Re-feeding syndrome has been identified in humans in some starving or malnourished patients receiving total parenteral nutrition (TPN), dextrose infusions or high carbohydrate diets. The syndrome has also been reported in tube-fed cats. The mechanism of re-feeding syndrome is poorly understood (not all susceptible patients succumb) but is ascribed particularly to hypophosphataemia with or without changes in potassium, magnesium and sodium. Phosphate has important functions in glycolysis, phospholipids and ATP formation. Severe hypophosphataemia causes haemolysis, muscle weakness, leucocyte dysfunction and a reduction in red blood cell (RBC) levels of 2,3-diphosphoglycerate, which increases the affinity of RBCs for oxygen so reducing oxygen delivery to tissues. Carbohydrate and insulin release cause an increase in cellular uptake of phosphate and increased glucolysis and protein synthesis, which leads rapidly to hypophosphataemia as total body phosphate is depleted. Advice in these patients is to increase calories gradually, monitor electrolytes carefully and do not use high carbohydrate diets or dextrose infusions.
How to Feed?
There are two important rules for feeding:
Methods of Encouragement
Simple techniques such as the warming of food or using foods with strong smells such as sardines can sometimes be successful. It is important to check with the owner the type of food the patient normally eats at home as feeding the usual diet is more likely to increase the chance of the patient eating in the hospital. Cats particularly can be very fussy eaters and have differing personalities, e.g. some cats are likely to eat when being fussed and stroked, whereas others will prefer privacy. The use of medication can sometimes be successful in stimulating patients’ appetites and giving them a reminder of what they are missing. Current drugs used to simulate appetite include cyproheptadine (Periactin), a serotonin antagonist, at 2–4 mg/kg/day PO and mirtazapine (Zispin), a tetracyclic antidepressant, at one-eighth 15 mg tablet PO every 2–3 days. The administration of 0.05–0.2 mg/kg valium IV may also be used.
Force Feeding
This technique occasionally may be helpful to ‘kick start’ animals into eating, particularly cats, puppies and kittens, but it is usually poorly tolerated by the patient and stressful for both the patient and staff. Care needs to be taken with this technique to avoid aspiration, particularly in collapsed patients where it should really be contraindicated (see Figure 16.3).
It is essential if using either of the above techniques to ensure the patient is receiving more than 88% of their calculated requirements daily. It is very easy to spend long periods of time trying to syringe feed a patient and actually get very little of its energy requirements down.
Tube Feeding
Prior to each feeding through enteral tubes, tube location must be checked by gentle application of negative pressure (by aspiration) to the end of the feeding tube. The aspiration of nasogastric and gastrostomy tubes also allows the measurement of residual gastric contents, which indirectly assesses gastric motility. Oesophagostomy tubes should contain no residual material if they are correctly located in the oesophagus. Because the jejunum has a relatively small holding capacity, aspirating residual contents has little value. Radiography can also be used to verify tube location. Contrast agents are usually not required, as most tubes are radiopaque (see Figure 16.4).
Figure 16.4 Radiograph of an oesophagostomy tube post placement. Contrast medium has been used to confirm placement.

Choice of Feeding Tube
The best feeding tubes for prolonged use are made of polyurethane or silicone. For short-term feeding (<10 days), polyvinylchloride or red rubber tubes can be used. These latter tubes are not appropriate for long-term feeding because they tend to become stiff with prolonged use and may cause the animal discomfort. Silicone is softer and more flexible than other tubing materials and has a greater tendency to stretch and collapse. Polyurethane is stronger than silicone, which allows for thinner tube walls and a greater internal diameter, despite the same overall French size. Both silicone and polyurethane tubes do not disintegrate or become brittle in situ. The French (Fr) unit measures the outer lumen diameter of a tube; each unit is equal to 0.33 mm.
Naso-Oesophageal and Nasogastric Tubes
Naso-oesophageal tubes can be inserted using minimal equipment and standard techniques. Nasogastric tubes are inserted in a similar fashion as naso-oesophageal tubes, but they should be long enough to reach 7–10 cm past the last rib. Both types of tubes are useful for providing short-term nutritional support (usually <7 days). They can be used in animals with a functional oesophagus, stomach and intestines. Naso-oesophageal tubes are contraindicated in animals that are vomiting, comatose, lack a gag reflex or have respiratory diseases. Complications include epistaxis, intolerance of the insertion procedure and inadvertent removal by the animal. Nasogastric tubes increase the risk of gastro-oesophageal reflux and thus may increase the incidence of oesophageal strictures and for these reasons should not generally be placed unless gastric syphoning is required, e.g. parvovirus, gastric dilation and volvulus (GDV) patients. Because of the small internal diameter of these tubes, only liquid enteral diets can be used. Feeding may be delivered via a syringe pump as a continuous rate infusion or as bolus feedings. If a syringe pump is used, the delivery equipment must be completely changed every 24 hours to help prevent bacterial growth within the system. Clogging of these tubes is a common problem, due to the narrow bore, but the incidence can be decreased by using a syringe pump or flushing the tube well before and after bolus feedings. A column of water should always remain within the feeding tube in between feeds. If the tube becomes clogged, replacement may be necessary, or carbonated drinks may be used to remove the blockage (see Figure 16.5). Diluting the liquid diet with water may also help prevent clogging, but this decreases the caloric concentration of the diet and increases the volume necessary to meet caloric needs.
Figure 16.5 Unclogging of a blocked oesophagostomy tube using a carbonated drink.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree




