23 Semen Collection, Evaluation, and Preservation
A rusty load is an ejaculate containing many old spermatozoa from storage, many of which are misshapen and have poor motility. If semen is collected from a dog that has not been used for a while and many of the spermatozoa appear abnormally shaped, semen should be collected again before that dog is evaluated critically.
I. SEMEN COLLECTION
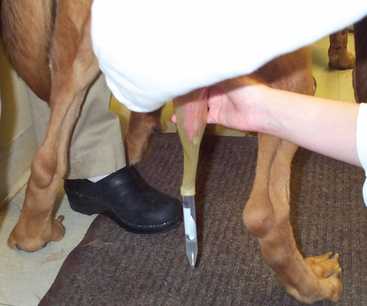
Semen can be collected into any container. I have heard of people using cups, sandwich bags, and other household items. Any container used must be very clean and contain no soap residues; any contaminant may kill spermatozoa collected into that vessel. I prefer to use a rubber collecting cone with a clear plastic centrifuge tube attached to the end (Figure 23-1) because it is a self-contained unit that does not allow semen to be spilled or ejaculated outside of the collecting instrument and because it best mimics natural service, presumably yielding a better quality ejaculate. Again, the equipment must be kept scrupulously clean. Rubber should not be cleaned with soap at any time because soap residues easily adhere to the rubber and may kill spermatozoa subsequently collected with that equipment.
Most male dogs can be collected in the absence of a teaser bitch. However, it has been demonstrated that the libido of the male and quality of the semen are improved in the presence of a teaser bitch, especially if she is in heat. Some very experienced male dogs require not just a bitch in heat but one that is at the optimal time for breeding. If a teaser bitch is not available, there are several techniques that can be used to entice the male: (1) use of a commercial product mimicking the pheromones produced by a bitch in heat, (2) allowing the male to sniff at swabs collected from an estrous bitch and frozen until needed, and (3) administration of a drug that stimulates contraction of the smooth muscle in the epididymis. In my experience, the first two techniques have not proven very successful. The last technique involves administration of prostaglandin (Lutalyse) 10 minutes before attempted semen collection. This technique has been demonstrated to increase number of spermatozoa in the ejaculate to the same extent as does the presence of an estrous teaser bitch.
If a teaser bitch is used, a handler should hold her with her hindquarters on the rug. If the bitch is not in heat, she should be muzzled or otherwise securely restrained. The male is allowed to investigate her hindquarters and to mount if he so wishes. The bulbus glandis is palpated through the prepuce. The area over the bulbus glandis is manipulated briskly and enthusiastically. As soon as the penis starts to become erect, the prepuce is pushed behind the engorging bulbus glandis. This is best accomplished by using the rubber collecting cone to push the prepuce back while “squirting” the penis out of the prepuce with the other hand. A better-quality ejaculate usually is obtained if the prepuce is pushed proximal to the erect bulbus glandis. Once the rubber collecting cone is advanced completely proximal to the bulbus glandis, the penis between the bulbus glandis and body wall is encircled tightly with the fingers. This mimics the tie. At this point, most dogs thrust vigorously and ejaculate a clear presperm fraction and then the cloudy sperm-rich fraction of the ejaculate (Figure 23-2). The dog stops thrusting and steps down. He may try to step over the collector’s arm. If the bulbus glandis is completely free from the prepuce, the collector should swing the dog’s penis 180 degrees in a horizontal plane and continue to hold. The final fraction, the prostatic fraction, is ejaculated as distinct pulses of clear fluid that will be visible filling the tube attached to the collecting cone and will be palpable in the hand holding the collecting cone and penis. The anus contracts rhythmically as prostatic fluid is ejaculated. The amount of prostatic fluid collected depends on the purpose of the semen collection (Table 23-1). At the conclusion of semen collection, the penis is released and the rubber collecting cone gently peeled off the penis. Allow the male to lick at the penis, walk him away from the collection area, or gently massage the engorged penis with towels moistened with cold water (not ice) to hasten detumescence of the penis. Ensure that the penis is completely flaccid and within the prepuce and that the prepuce is not rolled in on itself before kenneling the male dog.
Table 23-1 Prostatic Fluid Collection During Manual Ejaculation
| Purpose of Semen Collection | Amount of Prostatic Fluid to Collect |
|---|---|
| Breeding soundness exam (complete semen evaluation) | One to two good “squirts,” enough to ensure the entire second (“sperm-rich”) fraction has been collected |
| Semen collection for culture and cytology testing or other diagnostic tests (prostate disease, subfertility, or infertility) | As above or enough to provide sufficient volume for tests to be conducted |
| Artificial insemination with fresh semen (see Chapter 21) | Dependent on size of bitch to be inseminated. For bitches weighing less than 10 lb, total semen volume should be 1.5 to 3 mL; for bitches weighing 10 to 50 lb, 3 to 5 mL; and for bitches weighing more than 50 lb, 5 to 8 mL |
| Semen to be chilled and shipped overnight | One good “squirt”—amount of prostatic fluid collected should be minimized as incubation of spermatozoa with the dog’s prostatic fluid is associated with decreasing motility of those spermatozoa |
| Semen to be frozen | One good “squirt,” enough to ensure the entire sperm-rich fraction has been collected. Sample will be centrifuged and the spermatozoa removed for further processing |
II. SEMEN EVALUATION
A. VOLUME
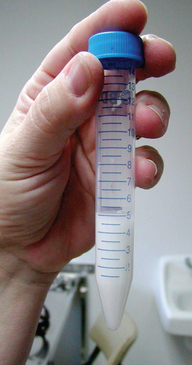
Volume is not an indicator of semen quality because it is dependent on the amount of prostatic fluid collected. However, the value for volume must be recorded to allow determination of the total number of spermatozoa in the ejaculate. A normal value for volume has been reported at 1 to 30 mL (Figure 23-3).
B. COLOR
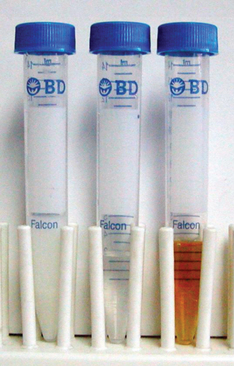
Color is a superficial indicator of semen quality and possible contamination of semen. Semen should be milky or opalescent. All milky fluid collected should be evaluated microscopically because occasionally dogs will ejaculate fluid that contains no spermatozoa but has many fat droplets that give it a milky appearance. Yellow is indicative of urine contamination. Brown or red indicates that blood is present in the semen. Brown discoloration is due to old blood and usually is associated with prostate disease. Red discoloration is due to frank blood and may be associated with prostate disease or with trauma to the penis. Occasionally young male dogs bleed from the surface of the penis the first time they become erect because small blood vessels rupture on the engorging penis. Green discoloration may be seen in animals with reproductive tract infection. Finally, clear fluid indicates lack of spermatozoa in the ejaculate (azoospermia) (Figure 23-4).
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree