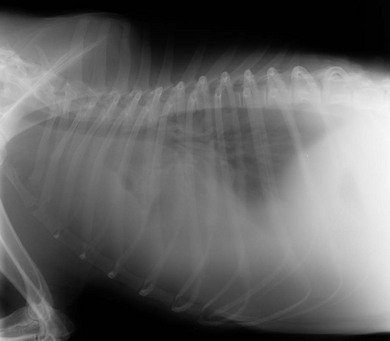
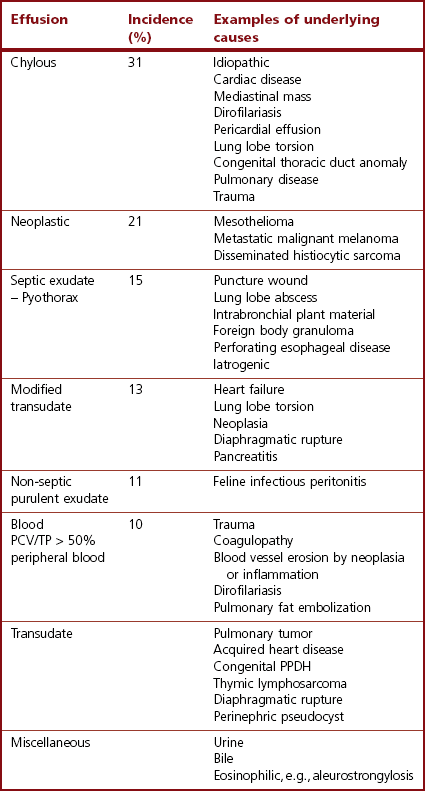
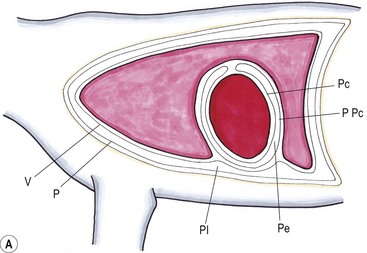
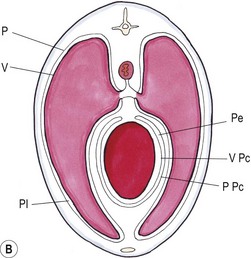
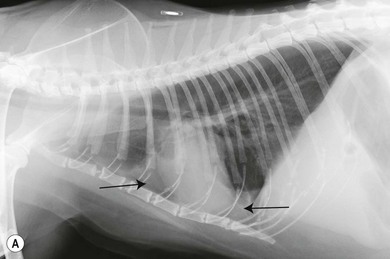
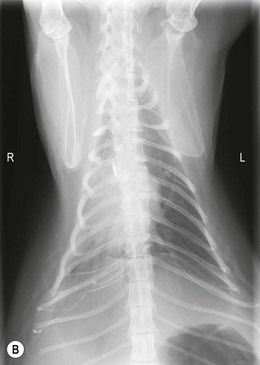
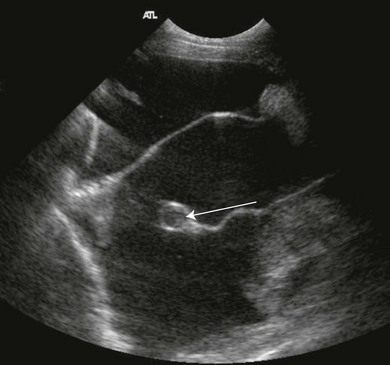
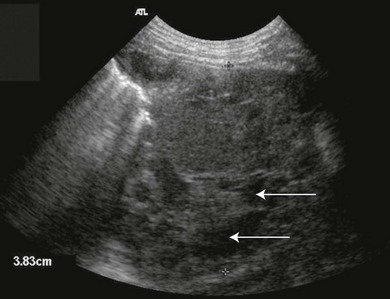
Chapter 44 Pleural diseases are a significant cause of respiratory distress in cats and while life-threatening if severe, they can often be managed successfully. Thirty years ago, all cats with pleural fluid due to thymic lymphosarcoma or pulmonary carcinoma were immediately euthanized according to one study.1 Now, advancements in diagnostic procedures, critical care management, surgical techniques, and adjunctive therapies mean that most pleural diseases have a better outcome. The pleura is a serous membrane that lines the mediastinum, pericardium, diaphragm and thoracic wall (parietal pleural), and the lungs (visceral or pulmonary pleura) (Fig. 44-1). A fold of pleura encloses the caudal vena cava. The left and right pleural cavities are potential spaces in normal cats, containing a small volume of serous fluid for lubrication. The mediastinal pleura is often incomplete, allowing communication between the pleural cavities. The blood supply to the parietal pleura is from the arteries of the thoracic wall, i.e., the intercostal, diaphragmatic, and pericardial arteries. The pulmonary and bronchial circulation supplies the visceral pleura. The pulmonary ligaments are fairly avascular. Figure 44-1 Pleural anatomy. P, parietal pleura; Pl, pleural cavity; P Pc, parietal pericardium; Pe, pericardial cavity; V, visceral pleura; V Pc, visceral pericardium. While pleural disease in cats will have the same clinical effects as in dogs, the symptoms are often less apparent due to the differences in physical activity, and as such, owners will usually not notice the early changes in behavior and respiration that may be detected in dogs. Signs are often not detected until the disease is in the advanced stages and causing respiratory distress, open mouth breathing, and cyanosis. Indeed, one study reported that breathing abnormalities were not noted by 40% of owners with dyspneic cats.2 In one study of 73 cats with pleural fluid, the main presenting complaints were dyspnea/tachypnea (69%), lethargy/weakness (36%), inappetence/weight loss (27%), dysphagia/regurgitation (12%), and cough (12%). Physical examination findings included dyspnea (59%), muffled cardiac and breath sounds (41%), tachypnea (38%), poor body condition (33%), hypothermia (21%), pyrexia (18%), reduced thoracic compressibility (14%), and tachycardia (8%).3 In light of the fragile nature of many of these patients, the first part of the examination of a dyspneic cat should just involve careful observation of the respiratory pattern and an assessment of the cat’s general condition, taking care to minimize stress and handling. If necessary, oxygen support should be provided before any physical examination is undertaken. Careful auscultation should then be performed to elucidate whether or not there is a reduction/absence of normal lung sounds and if so, in which areas of the thorax such deficits are present. Gentle percussion over all the lung fields can also be extremely useful, with dull, hyporesonant or absent sounds being suggestive of pleural fluid or a large mass lesion, in which case these abnormalities are likely to be in the dependent part of the thorax. Hyper-resonance is indicative of a pneumothorax and this is most likely to be heard in the dorsal part of the thorax (see Chapter 41, Table 41-2). Radiography is often not performed until after thoracocentesis. Movement artifacts are a significant problem and may limit the film’s diagnostic quality. Other imaging modalities, e.g., ultrasonography, may be preferred or used concurrently. Radiography can identify pleural fluid and air, with the dorsoventral view the most useful for detection of small volumes of air or fluid (Figs 44-2 and 44-3). Ventrodorsal radiography is avoided as it can lead to marked respiratory distress by causing collapse of the dorsal part of the lungs. Underlying causes of large volume pleural fluid cannot often be identified radiographically prior to thoracocentesis as fluid obscures soft tissue structures. Radiography is, however, useful for evaluating underlying diseases after fluid has been removed and can also be used to assess the completeness of thoracocentesis. Due to superimposition of mediastinal structures, especially in the cranial mediastinum, small mediastinal masses may be difficult to detect radiographically although there may be widening of the mediastinum. Large masses can usually be easily identified. Figure 44-2 (A) Lateral and (B) dorsoventral (DV) radiographs of the thorax. A small volume pleural effusion is evident in the lateral radiograph (arrows). In the DV radiograph, a mediastinal shift of the cardiac silhouette is present. The right lung lobes are collapsed causing an even increase in soft tissue opacity throughout the right hemithorax. The most likely cause is a right lateral recumbent position for some length of time before the DV radiograph was taken. A small amount of pleural effusion cannot be excluded on this radiograph. Ultrasound is the diagnostic imaging modality of choice to identify the presence of pleural fluid and has the advantage that it can be performed with the cat in almost any body position to minimize further respiratory difficulties (Fig. 44-4). Ultrasound also facilitates guided thoracocentesis of small volumes of fluid and identification of possible underlying causes, e.g., abscess. Ultrasound is an excellent way to assess mediastinal masses and is a simple method to aid procurement of cells or tissue by aspiration or needle core biopsy techniques. It can also be a useful way to assess the progression of treatment, as a cadaver study of estimation of fluid volume showed a good correlation with absolute volume,4 making it an effective method for comparing sequential fluid volumes. Ultrasound is less useful in the presence of air as the ultrasound waves are reflected at the soft tissue–air interface. Figure 44-4 Ultrasound images showing bilateral pleural effusion. The caudal vena cava is centrally located within its corresponding plica (arrow). Advanced imaging techniques can offer the most information on pleural disease. Computed tomography scans are generally significantly faster to perform than MRI and movement of the thorax is a particular problem with MRI, resulting in distracting artefacts (see Chapter 8). There will inevitably be some movement blur during computed tomography (CT), as there is for radiography and MRI, but this can be minimized by hyperventilation prior to scanning during the expiratory pause. CT allows effective visualization of the entire thoracic cavity for assessment of the volume of fluid/air, possible underlying causes, and other concurrent mediastinal diseases (see Chapter 8). The CT findings in normal cats and those with pleural, pulmonary, and mediastinal diseases have been described.5,6 Pus, blood, and chyle generally have a characteristic gross appearance, but laboratory testing is always necessary to confirm the diagnosis. Pleural fluid analysis should include cell count, cytology, aerobic and anaerobic bacteriological and fungal culture. Transudates and exudates can be accurately differentiated, with pleural fluid lactate dehydrogenase concentration and pleural fluid/serum total protein ratio being reported as the most the reliable parameters (sensitivity of 100% and 91% and specificity of 100%, respectively). However, additional parameters such as the pleural fluid/serum cholesterol ratio and serum/effusion cholesterol gradient can also help in classifying the effusion if necessary.7 Tables 44-1 and 44-2 show the incidence and underlying causes of pleural fluid submitted to a laboratory for analysis.3 No underlying cause was identified in 23% of cats. As expected, FIP affected predominantly young cats and neoplastic conditions, other than thymic lymphoma, older cats. Table 44-2 Underlying causes of pleural effusion based on pleural fluid analysis (n = 82 cats)3 Simultaneous pleural and peritoneal effusion is most common in dogs and cats with neoplastic and cardiovascular diseases and non-neoplastic liver disease. It develops most rapidly in those with infectious causes of pleuritis and/or peritonitis and pancreatitis, although the description in the literature does not differentiate dogs and cats.8 The prognosis of double effusions is grave as it is usually an indication of severe disease. Thoracoscopy (see Chapter 42) has been described in the cat as a less invasive technique than thoracotomy to examine the thoracic cavity and obtain tissue for histologic evaluation.9 However, this can be challenging in the face of a pleural effusion and full thoracic drainage is required before thoracoscopy is performed, to enable maximum visualization of the pleural cavity. Pyothorax is an inflammatory disease of the pleural space caused by inoculation of the pleural space with microorganisms. Cats with pyothorax may be younger than the general hospital population and more likely to come from multi-cat households and there is no sex predisposition. The causative organism is usually a bacterium and it was originally thought these came from the oral cavity following a cat bite, or the environment by direct penetration of the thoracic wall. However, in one large study only 14% of cats had a history of a bite or other trauma10 and the inciting cause often remains undetected. It has been recently suggested that the most common cause (56%) is parapneumonic spread of infection after colonization and invasion of lung tissue by oropharyngeal flora.2 Postmortem studies of cats with pyothorax showed underlying causes in less than half of cases and these included: lung lobe abscesses (Fig. 44-5), puncture wounds, intra-bronchial plant material, and foreign body granuloma.10 Iatrogenic infection occurred in 14% of cats with chylothorax treated by thoracocentesis or surgery.11 Figure 44-5 Ultrasound image showing multiple fluid-filled cavities (arrows) consistent with pulmonary abscessation. A positive bacterial culture was obtained in 96% of cats clinically suspected to have pyothorax, of which 89% were obligate anaerobes.12 Forty-four per cent of these cats had a mixture of facultative bacteria and obligate anaerobes and an average of 2.1 obligate anaerobic and 1.2 aerobic bacterial species were isolated per cat. Table 44-3 shows the bacterial isolates from feline cases of pyothorax from two studies. Of the facultative bacteria isolated, most were non-enteric (especially Pasteurella), in contrast to dogs in which the Enterobacteriaceae (especially E. coli) were more common.12 At the time of the study, the Pasteurella species were sensitive to those antibiotics they were tested against. Eight percent of cats with pyothorax had Actinomyces species isolated, which is usually considered to be associated with grass awns in dogs, but there appears to be less of an association in cats. Almost a quarter of cats are reported to have unusual causative organisms isolated including Candida albicans, Salmonella, Cryptococcus, Rhodococcus, Blastomyces, and Mycoplasma species.2,13,14 Table 44-3 Bacterial isolates from cases of feline pyothorax10,12 Typical clinical signs and physical examination findings are shown in Table 44-4. In one study 30% of cats had a current or recent history of upper respiratory tract infection.2 Dehydration in most cats and poor body condition in up to two-thirds suggest the disease is not immediately apparent to owners.10 Average time from onset of clinical signs to referral was up to 40 days.2,15 Changes on serum biochemistry and hematology included hyponatremia, hypochloremia, hypocalcemia, hypoalbuminemia, high aspartate aminotransferase activity, high total bilirubin concentration, elevated mean white blood cell and neutrophil counts, presence of band neutrophils, and anemia.2,10,15,16 Hypersalivation and a lower heart rate were significantly related to mortality, as was cholesterol and lower white blood cell count.10 Just over half of cats that had enough information for assessment were diagnosed with sepsis or systemic inflammatory response syndrome.10 Table 44-4 Typical clinical signs and physical examination findings in cats with pyothorax2,10,15 Radiography was used for the diagnosis of pleural fluid in all cases;2,10 76–89% had bilateral effusion, and 40% in one study also had evidence of concurrent pneumonia.2 Abnormalities of color, texture, and odor will be noted on gross fluid samples but the definitive diagnosis of an exudate requires pleural fluid analysis including cell counts and cytologic identification of degenerate neutrophils with or without the presence of intracellular bacteria. Cats are more likely than dogs to have bacteria identified on pleural fluid cytology (91% compared with 68% dogs with positive bacterial culture).2,12 Failure to demonstrate bacteria cytologically may be related to recent antibiotic administration or inappropriate techniques. Fluid samples should be submitted for aerobic and anaerobic bacteriologic and fungal culture and antimicrobial sensitivity testing to identify causative organisms and provide the appropriate treatment. Feline pyothorax is usually treated medically as a first-line approach. Intravenous fluid therapy is instituted to correct dehydration and electrolyte abnormalities, and thoracocentesis is performed to relieve dyspnea and obtain a fluid sample prior to antibiotic administration. Results of bacterial studies recommend that initial antibiotics be directed against both anaerobic bacteria (e.g., ampicillin, amoxicillin-clavulanic acid, clindamycin or metronidazole) and facultative bacteria (e.g., ceftizoxime).10 These drugs have good pleural penetration and low toxicity. Although amikacin and gentamicin have good activity against E. coli and other Enterobacteriaceae, their poor pleural penetration and the low incidence of Enterobacteriaceae in feline pyothorax limits their usefulness as a first-line antibiotic, although retrospective case series show that veterinarians have been prescribing them clinically.10 Obligate anaerobic bacteria are inherently resistant to aminoglycosides and quinolones and there may be inhibition of trimethoprim-sulfonamide by inhibitors within the exudate.12 The most commonly used antibiotics in a published retrospective study pending culture results were ampicillin (83%), gentamicin (62%), metronidazole (42%), the cephalosporins (23%), enrofloxaxin (19%), and clindamycin (17%).10 Most cats were treated with multiple classes of drugs but further information on specific combinations was not reported.10 Antibiotic use is prolonged, with a mean duration of 30–50 days.2,15 Thoracostomy tube use has been related to increased survival.10 Bilateral tube use has been recommended for bilateral effusion.2 The use of routine lavage in feline pyothorax is controversial; however, the cats in the more recent studies underwent therapeutic lavage with isotonic saline or compound sodium lactate, with the occasional addition of an antibiotic solution.2,15 The authors argued that lavage may decrease the time that tubes need to be maintained, thus reducing tube-associated pain, risk of nosocomial infection and hospitalization times. Cats have been successfully treated with small-bore wire-guided chest tubes placed by a Seldinger technique, which can usually be placed with the cat sedated rather than anesthetized.17 Lavage was performed two to six times daily and none of the tubes became occluded, suggesting that they can be successfully used and potentially reduce the morbidity associated with the large-bore tubes. Indications for surgery include failure of medical therapy or identification of an obvious surgical cause such as an abscess on diagnostic imaging (see Fig. 44-5). It appears that the requirement for surgery following medical management is low; of 35 cats that underwent thoracic lavage and survived the placement of drains in one study, only one cat required thoracotomy.2,15 In the largest case series of pyothorax in cats, 26% of cases were euthanized without treatment, due to either financial constraints or a poor perceived prognosis, and a further 11% died within 24 hours of presentation.10 Overall, 66% of cats that were treated survived, with 78% survival of those still alive 24 hours after admission. Of those cats available for follow up, only one (5%) had disease recurrence. Better survival rates were reported in studies where thoracostomy tubes were placed in all patients and thoracic lavage was performed (77–93%).2,15
Pleura
Surgical anatomy and pathophysiology
General considerations and diagnostic procedures
Clinical signs and physical examination
Diagnostic imaging
Laboratory analysis and etiology
Underlying disease
Incidence (%)
Pyothorax
18
Mediastinal lymphoma
17
Feline infectious peritonitis
10
Trauma
10
Pulmonary neoplasia
6
Hypertrophic cardiomyopathy
6
Unspecified mediastinal mass
5
Pulmonary thromboemboli
1
Pericardioperitoneal hernia
1
Heartworm disease
1
Restrictive cardiomyopathy
1
Unknown
23
Thoracoscopy
Surgical diseases
Pyothorax
Bacteria
Incidence (%)
Aerobic
Pasteurella
13–39
Unidentified non-enteric bacteria
5
Actinomyces spp.
4–15
Other
5
Anaerobic
Clostridium
0–48
Bacteroides spp.
19–20
Peptostreptococcus anaerobius
17
Fusobacterium spp.
14–21
Porphyromonas spp.
10
Prevotella spp.
8
Other
16
Negative culture
4
Clinical signs
Incidence (%)
Decreased appetite/anorexia
21–83
Lethargy
36–80
Dyspnea
59–79
Hypersalivation
18
Weight loss
14–36
Cough
26
Vomiting
15
Pain
13
Ocular/nasal discharge
9
Physical examination findings
Increased respiratory effort
81–94
Abnormal lung sounds
97
Abnormal mucous membrane color
97
Poor pulse quality
68
Mental dullness
96
Dehydration
7–93
Poor body condition
37–67
Tachycardia
21–64
Pyrexia
29–50
Hypothermia
15
Muffled heart sounds
14–37
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Veterian Key
Fastest Veterinary Medicine Insight Engine