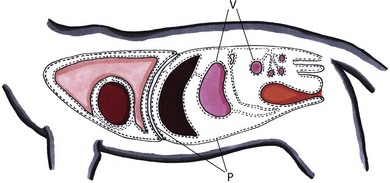
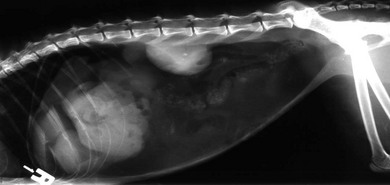
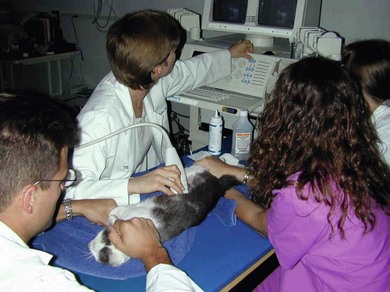
Chapter 26 The peritoneum is a closed cavity that contains all of the abdominal organs except for the kidneys and the adrenal glands. The parietal peritoneum covers the abdominal wall and diaphragm. The visceral peritoneum covers the abdominal organs (Fig. 26-1). The peritoneum consists of a single layer of mesothelial cells covering a basement membrane.1 The peritoneal mesothelial cells have the same embryological origin as vascular endothelial cells2 and produce surfactant that acts as a lubricant.3 The basement membrane is a fibroelastic tissue containing glycosylated proteins, mast cells, macrophages, and lymphocytes and it covers a well-defined network of elastic fibers. The basement membrane is absent in parts of the diaphragm, omentum, and mesentery.1 The absence of the basement membrane in the diaphragm allows large gaps between the mesothelial cells (‘stomata’) to communicate directly with underlying lymphatic channels, facilitating absorption of fluid and particulate matter. The majority of the peritoneum behaves as a semi-permeable membrane allowing bidirectional diffusion of water and solutes. A small amount of serous fluid, formed by the action of Starling forces across the peritoneal capillaries, is normally present in the space between the parietal and visceral peritoneal surfaces. The fluid has a low protein concentration (<3 g/dL protein) and resembles an ultrafiltrate of plasma. In humans, normal peritoneal fluid has antibacterial activity against Gram positive and Gram negative bacteria.4 A normal peritoneal cavity contains approximately 300 cells/mm3, which are largely lymphocytes and macrophages. Fluid injected into a normal peritoneal cavity is dispersed throughout the entire peritoneal space over a period of between 15 minutes and two hours.5 Contraction of the diaphragm promotes a cranial flow of peritoneal fluid.6 Relaxation of the diaphragm generates a sub-atmospheric pressure that draws fluid and small particles into the large openings between the peritoneal mesothelial cells covering the diaphragm (‘stomata’) that communicate with the underlying lymphatic system. Contraction of the diaphragm closes the stomata and forces fluid into the lymphatic system.1 This lymphatic system transports fluid and other absorbed material into the internal thoracic (or ‘parasternal’) mediastinal lymphatic system and mediastinal and parasternal lymph nodes, then to the thoracic duct system, and eventually to the systemic venous circulation.7 Fluid is normally also absorbed through other sites in the peritoneum, including the omentum.1,8 A physical, chemical, or microbiological injury to the peritoneum activates local defense mechanisms. These defense mechanisms fall into three broad categories (Table 26-1).9–18 The inflammatory response within the peritoneal cavity and the associated increased peritoneal vascular permeability result in the effusion of a large volume of protein-rich fluid into the peritoneal space. This results in a substantial loss of intravascular volume. In addition, systemic activation of the inflammatory and coagulation cascades causes further loss of fluid and protein and often disregulation of vascular tone and decreased tissue perfusion.19 Systemically, macrophages and other cells in the immune system respond to endotoxin and other bacteria-derived molecules (‘pathogen-associated molecular patterns’ or PAMPS) and endogenous mediators released from dead or dying cells20 through Toll-like receptors to initiate a complex systemic inflammatory response (SIRS). The SIRS response is referred to as ‘sepsis’ when initiated by bacteria. Binding of endotoxin from Gram negative bacteria to the lipopolysaccharide (LPS)-binding protein on macrophages stimulates the release of multiple cytokines.21 The mechanisms by which Gram positive bacteria induce systemic inflammation are less well characterized but several substances they contain, including peptidoglycan, teichoic acid, and their polysaccharide-hyaluronic acid capsules have pro-inflammatory effects, including the production of cytokines (tumor necrosis factor [TNF], interleukin-1 [IL1]) and nitric oxide.22,23 The macrophage is the main cell initiating the systemic response to bacteria and proteins from damaged cells.19 IL1 and TNFα are rapidly released after macrophage stimulation and promote activation of many of the inflammatory and coagulation cascades. A detailed discussion of cytokines involved in sepsis is beyond the scope of this chapter and the reader is referred elsewhere for a more detailed review.19 The net result of sepsis in peritonitis is a decrease in intravascular volume, decreased cardiac output as a result of decreased preload, abnormal vascular tone, and vascular thrombosis, leading to maldistribution of blood flow, poor tissue perfusion, and organ damage. For more information on shock and SIRS the reader is referred to Chapter 3. The gastrointestinal tract is the most frequent source of peritoneal contamination in cats.24 Usually, only two or three bacterial species are grown in culture, although numerous species of bacteria are found in the gastrointestinal tract. Polymicrobial infections present a significant problem because of bacterial synergism. This appears to be most important in mixed infections with Gram negative and anaerobic bacteria. Anaerobic bacteria alone (such as Bacillus fragilis) seem to cause minimal pathology in experimental models. However, when combined with Gram negative enteric bacteria, the resulting infection is considerably more severe.25 Ingesta, exfoliated cells, blood, gastric mucin, and bile salts are adjuvant substances that worsen peritonitis. Blood is an excellent growth medium for bacteria and hemoglobin interferes with chemotaxis and phagocytosis. Bile salts destroy peritoneal mesothelial cells and inhibit neutrophil function. Uroperitoneum occurs when the bladder or peritoneal urethra is damaged by external or iatrogenic trauma. Severe trauma to the kidneys or ureters often damages the peritoneum, allowing urine to leak into the abdominal cavity from the retroperitoneal space. Urine is hyperosmotic and pulls fluid from the vascular space into the peritoneal cavity, resulting in dehydration. Failure to excrete urine also causes acidosis and progressively worsening hyperkalemia. Severe hyperkalemia is truly life threatening. For the effects and treatment of hyperkalemia see Chapters 1 and 3. The historical and physical examination findings of cats with peritonitis are usually associated with the SIRS syndrome. The historical findings most commonly reported by owners tend to be non-specific. However, a history of trauma or penetrating abdominal wounds may increase the clinician’s suspicion of peritonitis. Gastrointestinal signs may be associated with primary gastrointestinal problems (i.e., foreign body) or may solely be due to peritonitis. An icteric animal may suggest primary biliary disease or associated pancreatitis. In both primary (not associated with feline infectious peritonitis [FIP]) and secondary bacterial peritonitis in the cat, common historical findings include lethargy, vomiting, anorexia, and diarrhea.26 Weight loss is another clinical sign reported that appears to be more frequently associated with secondary peritonitis than with primary.26 On physical examination, pertinent findings in cats with peritonitis tend to be associated with dehydration and shock, and can include depressed mentation, inadequate hydration, pale mucus membranes, tachypnea, tachycardia, weakness, hyperthermia, and poor nutritional condition.26–28 Cats with peritonitis and sepsis are often bradycardic and/or hypothermic.26–28 Abdominal distension, which has been reported to be a common finding in cats with peritoneal effusion, can be identified by owners or on palpation during physical examination.29 In humans, abdominal pain is an early clinical sign of primary peritonitis,30,31 but it appears not to be a good indicator of peritonitis in cats.24,27 The overall incidence of abdominal pain elicited in cats with peritonitis ranged from 38–62%.24,27 Survey abdominal radiographs are often taken to evaluate cats with clinical signs of gastrointestinal, urinary, or non-specific abdominal disease. The presence of free abdominal gas with no history of recent abdominal surgery or penetrating trauma warrants immediate stabilization and surgical exploration (Fig. 26-2).32 It can be difficult to definitively diagnose small volumes of free peritoneal gas; close attention should be given to the diaphragmatic crura, as they are often highlighted by gas in the lung cranially and in the peritoneal cavity caudally. Loss of serosal detail and ileus also suggest intraperitoneal disease. Plain abdominal radiography may also diagnose diseases underlying peritonitis, including gastrointestinal foreign bodies, pyometra, cholelithiasis, and abdominal neoplasia. Abdominal ultrasonography (Fig. 26-3) is very useful for obtaining samples of free peritoneal fluid. Blind, four quadrant tapping and diagnostic peritoneal lavage have been described for the same purpose. Although these three techniques have not been rigorously compared, ultrasound-guided aspiration has a subjectively higher diagnostic yield than a four quadrant tap and allows fluid to be directly analyzed without the dilutional effect of diagnostic peritoneal lavage. The hair over the abdomen is clipped and prepared with aseptic technique. A fluid accumulation is located with ultrasound and aspirated using a syringe and needle. The fluid’s pH, glucose, lactate, and cytology are evaluated (Table 26-2).33 Ultrasonography can also evaluate the abdominal organs for masses and abscesses and evaluate the urinary and biliary systems. Cytological analysis has traditionally been used to evaluate peritoneal fluid (Table 26-2). Septic peritoneal effusions generally contain degenerate neutrophils and macrophages. The presence of intracellular bacteria is diagnostic for septic peritonitis.34 A comparison of peritoneal fluid and peripheral blood lactate and glucose concentrations is also useful for confirming the diagnosis of septic peritonitis. Bile peritonitis is diagnosed based on cytological and biochemical analysis of peritoneal fluid. The diagnosis of uroperitoneum is based on a history of either blunt abdominal trauma, recent catheterization, or recent expression of the bladder. In the majority of cats with uroperitoneum, the peritoneal fluid creatinine and potassium levels are higher than those in the serum (see Chapter 3).35 Peritonitis (inflammation or infection of the peritoneum) is the most common disease affecting the peritoneum in the cat. It occurs subsequent to systemic infection or leakage of gastrointestinal tract contents. Peritonitis can be subdivided into primary, secondary, or tertiary peritonitis, reflecting the cause or source of the infection. Neoplasia of the peritoneum is rare, but both primary mesothelioma36–38 and secondary metastases to the peritoneum have been reported in the cat. Primary or spontaneous peritonitis is defined as an infection of the peritoneal cavity where no cause for the intraperitoneal bacterial contamination has been identified.30,31,39 In primary peritonitis, bacterial contamination of the peritoneal cavity occurs via blood-borne or lymphatic spread or by bacterial translocation from the intestinal tract.30 The true origin of bacterial contamination is difficult or impossible to determine in many instances. Primary peritonitis is more commonly reported in human than veterinary patients and is usually associated with diseases that cause ascites, such as cirrhosis.30,31,40,41 In veterinary medicine, FIP is the most common cause of primary peritonitis.39,42 FIP is caused by infection with a feline coronavirus and presents in two forms, the granulomatous (dry) form or the effusive (wet) form, which is the form that results in abdominal effusion and peritonitis.43 In a retrospective study on cats with peritonitis, four out of 65 cats had primary peritonitis resulting from the effusive FIP.44 There are numerous reports of primary peritonitis in cats due to other pathogens besides FIP; however, the true source of the infection in most cases is unclear.45–49 Secondary peritonitis is the most common form of peritonitis seen in veterinary medicine.31 Bacterial contamination results from penetrating abdominal trauma, bite wounds, contamination from surgery or diagnostic aspiration procedures. Leakage of bacteria from abdominal organs into the peritoneal cavity is another frequent source of contamination in secondary peritonitis.31 While all of the abdominal organs can be potential septic sources of contamination, leakage of contents from the gastrointestinal tract is the most common cause of peritonitis.27,50–53 In cats, gastrointestinal leakage causing septic peritonitis is most often the result of neoplasia.27 In one study, neoplasia was the underlying cause of gastrointestinal leakage and subsequent septic peritonitis in 54% of cats.27 Adenocarcinoma and lymphosarcoma were the most common neoplasms identified.27 Other reported causes of gastrointestinal leakage include perforation of the bowel due to a foreign body (Fig. 26-4), leakage from a previous surgical site, and perforation of the colon due to megacolon and enteritis.27 Figure 26-4 Peritonitis secondary to intestinal perforations from a linear foreign body. (Courtesy of Dr Karen Tobias.) Leakage from an enterotomy, gastrotomy, or a resection and anastomosis site is another potential cause of secondary peritonitis.54,55 Dehiscence occurred in 14% of cases in one study of dogs and cats, although none of the 25 cats had surgical dehiscence and leakage.55 It is difficult to interpret the significance of these findings due to the limited numbers of cases in this study specifically evaluating cats. In a second study, no anastomosis or biopsy site leaked in 70 cats with alimentary lymphosarcoma.56 In dogs, factors suggesting an increase in the risk of postoperative dehiscence include presence of a foreign body or a traumatic injury, preoperative peritonitis, and hypoalbuminemia.54,55 Perforation of a gastrointestinal ulcer, perhaps associated with carprofen ingestion, has been reported in one cat with peritonitis.57 Secondary peritonitis in cats may occur following bite wounds, gunshot wounds or vehicular trauma.27 Bacteria enter the peritoneal cavity directly from the environment or from trauma resulting in rupture of a viscus. Penetrating wounds warrant emergency surgical exploration to evaluate for possible bowel damage, wound debridement, and lavage. Septic peritonitis from urinary tract infection is rare and urine is not considered an adjuvant substance in peritonitis.58 In a retrospective study of 51 cases of septic peritonitis in cats, only four cases of peritonitis appeared to have resulted from a urinary tract infection.27 Feline pyometras occasionally rupture and cause secondary peritonitis. In a retrospective study of cats with pyometra, seven out of 183 cats (3.8%) had a uterine rupture, and four of the seven cats died as a result of the septic peritonitis.59 Secondary septic peritonitis can also occur because of biliary leakage resulting from trauma anywhere along the biliary tract, necrotizing cholecystitis, cholelithiasis, or iatrogenic injury from aspiration or surgical manipulation.24,60–63 In dogs, it appears that animals with septic bile peritonitis had a decreased survival rate and increased mortality as compared to those with aseptic peritonitis.60,63 Other potential sources of secondary peritonitis include abscessation of the pancreas, liver, or spleen64 or iatrogenic contamination of the abdominal cavity after abdominal surgery. In 51 cats with secondary peritonitis, three developed septic peritonitis due to septic pancreatitis or a pancreatic abscess, and one due to liver lobe necrosis.27
Peritoneum
Surgical anatomy
Physiology
Pathophysiology
Gastrointestinal tract leakage
Urinary tract leakage
Diagnosis and general considerations
Physical examination findings
Diagnostic procedures
Radiography
Ultrasonography
Cytological analysis
Surgical diseases of the peritoneum
Primary peritonitis
Secondary peritonitis
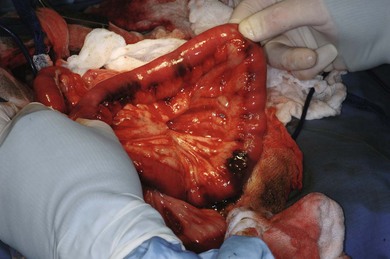
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Veterian Key
Fastest Veterinary Medicine Insight Engine