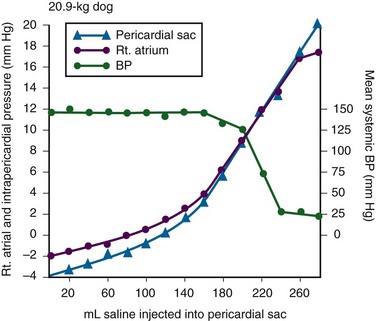
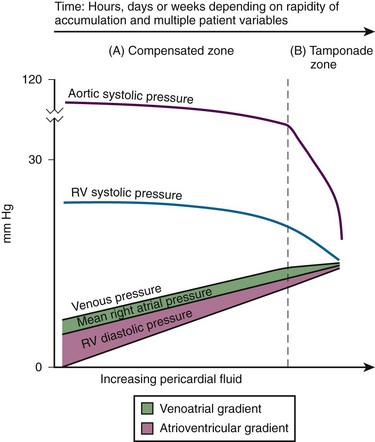
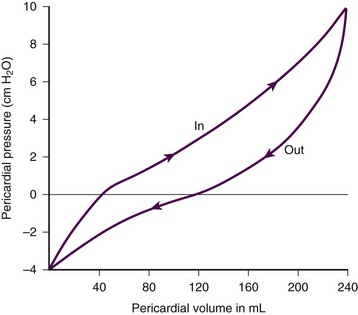
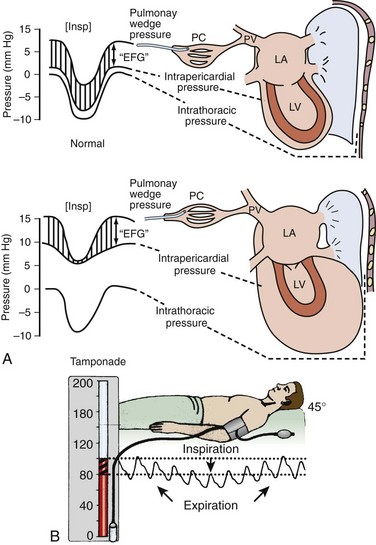
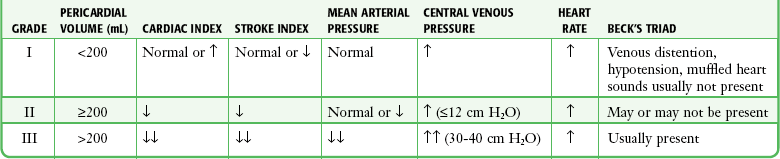
Chapter 16 Pericardium and Pericardial Space The pericardium is a two-layered fibroelastic sac surrounding the heart.1 The pericardium is avascular but well innervated, so inflammation induces severe pain. The visceral pericardium is a single-cell layer that adheres to the epicardium. The outer parietal pericardium consists mostly of collagen with some elastin. These two layers create the pericardial space, which normally contains 15 to 50 mL of serous fluid.2 Pericardial fluid provides lubrication for cardiac contractility and acts as a “shock absorber” for deceleration forces. The pericardium is a tense structure, but it also has some elasticity. These properties limit the amount of cardiac dilation that is possible during diastole and enhance mechanical interactions between the atria and ventricles during systole.3 This semi-elastic property can also tolerate an acute (i.e., over a period of hours to days) accumulation of pericardial fluid (80 to 120 mL) without significantly increasing intrapericardial pressure, which is the flat portion of the pressure-volume curve for pericardial pressure (Fig. 16-1).4,5 Once a critical volume is reached, adding as little as 20 to 40 mL can double intrapericardial pressure (the steep portion of the pressure-volume curve [see Fig. 16-1]) and cause clinical decompensation (cardiac tamponade). Cardiac tamponade typically occurs with an intrapericardial pressure of 15 to 20 mm Hg.6 Elevated intrapericardial pressure (i.e., early tamponade) results in abnormal RV filling and then abnormal LV filling. In this situation, the free wall of the RV cannot expand against the pericardial fluid during inspiration. To accommodate the filling, the interventricular septum bows abnormally into the LV, which reduces its volume. LV filling, stroke volume, and ultimately cardiac output are reduced.7 This phenomenon is responsible for pulsus paradoxus (PP, described later in this chapter), which is sometimes observed with tamponade.8 LV filling is also reduced by the collapse of right-sided structures. After a critical volume is reached on the pressure-volume curve (see Fig. 16-1), the intrapericardial pressure is transmitted to the inferior vena cava (IVC) and right atrium. These thin-walled structures then become compressed and reduce filling of the RV. The atria and pulmonary circulation are at much lower pressure than systemic arterial pressure and are also vulnerable to rising intrapericardial pressure (Fig. 16-2). Late in tamponade a “pressure plateau” occurs in which right atrial pressure, RV diastolic pressure, pulmonary artery diastolic pressure, and pulmonary capillary wedge pressure are virtually identical. This equalization of chamber pressure leads to a reduction in venous return and the echocardiographic hallmark of tamponade: diastolic collapse of the RV. At this point, hemodynamic collapse is imminent, with severe hypotension, bradycardia, and potentially pulseless electrical activity (PEA) developing. Unless intrapericardial pressure is decreased immediately, cardiac arrest will ensue.9 Compensatory mechanisms also preserve normal cardiac contractility and myocardial perfusion.7,10,11 However, when pericardial pressure overwhelms the compensatory mechanisms, coronary perfusion pressure is reduced, which leads to myocardial ischemia. Experimental induction of severe tamponade demonstrated microscopic ischemic cardiac injury.12 Lactic acidosis (resulting from reduced cardiac output and systemic hypoperfusion) may directly cause cardiac depression and thereby reduce cardiac contractility and, ultimately, cardiac output.9 Removal of pericardial fluid (i.e., pericardiocentesis) reverses the pathophysiologic processes just described by improving cardiac filling and output. Interestingly, the pressure-volume relationship of the pericardial space demonstrates hysteresis; that is, withdrawing a certain quantity of fluid reduces intrapericardial pressure more than addition of the same amount of fluid increases intrapericardial pressure. This effect, however, is not universal and may vary among patients and in various disease states (Fig. 16-3).2 Under normal circumstances, positive pressure ventilation (e.g., mechanical ventilation) reduces venous return to the right side of the heart by increasing intrathoracic pressure. This could be detrimental for patients with tamponade because right-sided filling is already compromised and further reductions can lead to severe hemodynamic instability.13 Therefore, positive pressure ventilation should be avoided in patients with known or suspected tamponade unless it is absolutely necessary. Low-pressure pericardial tamponade is defined as a hemodynamically significant effusion with lower than expected intrapericardial pressure.14 This category of tamponade occurs in certain hypovolemic patients with subacute or chronic effusions (e.g., associated with long-term diuretic use, dehydration, excessive dialysis).15 The diagnosis may be challenging because the classic symptoms and findings on physical examination (e.g., distended neck veins) may be absent.16 Fluid boluses may temporize the hemodynamic compromise while pericardial decompression is being arranged. The major categories of pericardial effusion include infection, malignancy, trauma, and metabolic abnormalities. Effusion may also be associated with aortic disease, connective tissue disease, or idiopathic causes. It is often difficult to report the exact incidence of each type of pericardial effusion because of variations in patient populations, local epidemiology, and the diagnostic protocols used during evaluation. The prevalence of a chronic effusion is also difficult to ascertain because it is often asymptomatic and underreported. General autopsy studies demonstrate an overall prevalence of 3.4%.17 Penetrating Trauma: Penetrating cardiac trauma can cause acute hemopericardium by either external forces (e.g., a stab wound to the heart) or internal forces (e.g., iatrogenic injury during placement of a pacemaker). Cardiac perforation can lead to rapid clinical deterioration and PEA. External cardiac puncture is associated with stab wounds or projectile injuries (e.g., gunshot wounds). Tamponade develops in 80% to 90% of patients with cardiac stab wounds as opposed to 20% of those with gunshot wounds.18,19 Stab wounds cause tamponade more frequently because if the pericardial injury is small, it can reseal and trap blood within the pericardial space.20 On the other hand, a gunshot typically produces large pericardial wounds that allow continuous drainage into the pleural space.21 Clinical deterioration is usually secondary to hypovolemia.21 Any penetrating injury to the chest, back, or upper part of the abdomen may injure the pericardium and cause tamponade. Internal penetrating trauma is typically caused by invasive diagnostic or therapeutic procedures. The procedures most often associated with this injury are pacemaker insertion and cardiac catheterization (angioplasty or valvuloplasty).22–24 Hemopericardium results from puncturing the cardiac chamber, a coronary artery, or a great vessel (e.g., the superior vena cava). Ironically, pericardiocentesis itself (treatment of a pericardial effusion) can cause hemopericardium if coronary vessels or the myocardium is injured during the procedure.25,26 Internal jugular and subclavian venous catheters (e.g., central venous or hemodialysis catheters) are commonly inserted in the emergency department (ED). During such procedures, hemopericardium results from perforation of the superior vena cava, right atrium, or RV. Hemopericardium can occur immediately or can be delayed up to 2 days subsequent to erosion of the catheter through myocardial or vascular tissue.27,28 Although this complication seldom occurs, it should always be considered when a patient experiences sudden hemodynamic deterioration following an invasive procedure. Blunt Trauma: Major blunt chest trauma can cause hemopericardium with or without obvious signs of injury.29 Myocardial rupture can be uncontained or contained.30,31 Patients with uncontained rupture do not typically survive long enough to reach the hospital.32 Contained rupture may be found soon after injury or may be a late finding (in some cases up to 12 days later).33 Tamponade can also be caused by a deceleration mechanism of injury that induces either aortic or vena caval disruption.34 In one case series the incidence of tamponade following deceleration injury was found to be 2.3% (1 in 43 patients).35 Miscellaneous Trauma: Chest compressions during cardiopulmonary resuscitation (CPR) can also cause hemopericardium from broken ribs, bleeding intercostal vessels, or penetrating injury (e.g., intracardiac injection of medication, which is rarely performed today).36 Hemopericardium following CPR has been described in case reports37,38 but is unlikely to be significant, much less to cause tamponade. Bleeding diathesis is an important cause of spontaneous hemopericardium and may be associated with the use of anticoagulants (reported incidence of 2.5% to 11%)22 or thrombolytic therapy (incidence <1%).39 Patients who have undergone cardiac surgery are at increased risk because of the anticoagulative effects of the cardiopulmonary bypass machine and medications started postoperatively (e.g., clopidogrel, warfarin).40 Fortunately, tamponade has a low incidence and is generally detected in the postoperative period before discharge.41,42 This complication is usually prevented by the intraoperative placement of mediastinal or pericardial drains.22,43 Hemopericardium can develop following MI. Early after a transmural MI (1 to 3 days), the necrotic myocardium causes inflammation of the overlying pericardium and then effusions can form. Late-developing effusions (weeks after an MI) are caused by an autoimmune pericarditis called Dressler’s syndrome.1 Improved reperfusion techniques have reduced the incidence of post-MI pericarditis and effusion.44 Ascending aortic dissection causes rapid and usually fatal hemopericardium. The dissection may expand in a retrograde fashion by extending to the base of the aorta and into the pericardial sac. Risk factors for aortic dissection include hypertension, atherosclerosis, vasculitis (e.g., giant cell arteritis, syphilis), collagen vascular disease (e.g., Marfan’s syndrome), and the use of sympathomimetics (e.g., cocaine).45–47 Ventricular free-wall rupture is a rapidly fatal cause of acute hemopericardium that can occur after MI. This complication is less common today than in the past (<1%)39 secondary to improved revascularization techniques, better therapeutic medications, and faster intervention times (shorter door-to-balloon times) for coronary ischemia. Despite a reduction in its overall incidence, 7% of all deaths related to MI are caused by this complication.48,49 Survival is possible with prompt recognition and treatment, but the prognosis is grim once tamponade occurs.50,51 Nonhemorrhagic pericardial effusions usually accumulate slower than acute hemopericardium does (over a period of weeks to months). Chronic fluid accumulation allows the pericardium to stretch circumferentially and accommodate up to 2000 mL of fluid without any hemodynamic compromise.52 Effusions that grow slowly allow the circulatory system to adapt to the intrapericardial pressure, thereby further maintaining hemodynamic stability. Thus, asymptomatic patients with moderate to large effusions may not need emergency pericardiocentesis, in contrast to patients with acute hemopericardium.53,54 Nonhemorrhagic effusions have several causes (see Box 16-1), and the exact one may not be obvious during the initial evaluation without diagnostic pericardiocentesis. Common causes of nonhemorrhagic effusions are discussed in the following sections. Most idiopathic effusions are believed to be viral in origin and most commonly caused by infection with coxsackievirus, echovirus, or enterovirus. Idiopathic pericardial effusions may be asymptomatic or have an associated component of pericarditis (e.g., positional pain or diffuse ST-segment changes on the electrocardiogram [ECG]).55 These effusions are often labeled “idiopathic” because the diagnosis cannot be made noninvasively (i.e., based on the history, physical examination, or serum testing) and the risk associated with diagnostic pericardiocentesis outweighs the risk of observation in asymptomatic adults who appear to be well.56 Diagnostic pericardiocentesis may be recommended for idiopathic effusions that are persistent or symptomatic without a known cause.57 Tumors of the pericardium or myocardium may cause nonhemorrhagic effusions.54,58 Primary cardiac tumors are less common (0.001% to 0.003%) than metastases from another site (2% to 18%), but either may cause a malignant effusion.59 Although no malignancy preferentially metastasizes to the heart, certain tumors commonly involve the heart when they metastasize; frequently implicated are lung cancer, breast cancer, mediastinal tumors, malignant melanoma, leukemia, and lymphoma.46 Cardiac metastasis is usually a late finding in cancer; other foci are generally evident first.47 The classic signs and symptoms of tamponade (e.g., chest pain and dyspnea) may not be obvious with malignant tamponade. When present, they may be mistakenly attributed to the underlying malignancy.46 Thus, in the relevant clinical scenario, consider screening patients with malignancy for pericardial effusion (e.g., ultrasound) before the clinical findings of tamponade appear. Congestive heart failure (CHF) is a cause of pericardial effusion. Diagnosis may be difficult because of overlapping signs and symptoms with exacerbations of CHF (e.g., chest pain or dyspnea). Adding to the diagnostic complexity is that 12% to 20% of patients with CHF have a coexisting pericardial effusion.60 Fortunately, treatment of CHF-associated pericardial effusion does not differ from that for an effusion from other causes: treat the underlying cause unless the patient has evidence of hemodynamic compromise. Pericardial effusions (secondary to radiation-induced pericarditis) can develop acutely during radiation therapy or nay be delayed for years. Risk factors include the radiation dose, duration of exposure, and age of the patient. Patients treated with radiation for Hodgkin’s disease have the highest association of radiation-induced pericarditis and subsequent effusions.22 These effusions can be serous, hemorrhagic, or fibrinous.57 Human immunodeficiency virus (HIV) can cause nonhemorrhagic pericardial effusion and tamponade (see Review Box 16-1).61,62 The incidence has been reported to be approximately 11% in patients with HIV infection or acquired immunodeficiency syndrome (AIDS), and 13% of cases are classified as moderate to severe. It is unclear whether antiretroviral therapy has affected these data.17 HIV-related effusions have been attributed to bacterial (e.g., Staphylococcus aureus), viral (e.g., cytomegalovirus), fungal (Cryptococcus neoformans), and mycobacterial causes (e.g., tuberculosis, which is the most common cause of HIV-related effusions worldwide).63 Kaposi’s sarcoma and lymphoma can cause noninfectious pericardial effusions in HIV patients.64,65 Pericardial effusion develops in approximately 15% to 20% of dialysis patients, and tamponade may eventually occur in as many as 35% of that group.66,67 Up to 7% of chronic dialysis patients have effusions with volumes of 1000 mL or greater.68 In many cases, effusions secondary to renal failure can be managed solely with aggressive dialysis without pericardiocentesis. Any sign of hemodynamic compromise, however, warrants strong consideration of pericardiocentesis. Hypothyroid patients are at risk for pericardial effusions (up to 30%), but the fluid accumulates gradually, so tamponade develops in only a few patients.54 If pericardial effusions are present, other areas of the body usually demonstrate serositis (e.g., pleural effusions). Treating the underlying hypothyroidism often reverses the effusion without the need for pericardiocentesis. Pericardial tamponade is classically described as being secondary to circumferential effusion, which causes a generalized increase in pericardial pressure and compression of multiple cardiac chambers. Loculated effusions (caused by a local hematoma or an infectious process) or pericardial adhesions (from previous inflammation) can compress one or two cardiac chambers and thus reduce both cardiac filling and cardiac output.69,70 Constrictive pericarditis occurs following chronic pericardial inflammation, infection, or mediastinal irradiation. These processes cause scarring, fibrosis, or calcification, and the pericardium eventually becomes a nonelastic and “constrictive” sac around the heart. Myocardial relaxation and cardiac filling are impaired, and diastolic dysfunction ensues. Without echocardiography, constrictive pericarditis can be difficult to distinguish from pericardial tamponade.1 Effusive-constrictive pericarditis is defined by the presence of both pericardial effusion and pericardial constriction. It may be quite difficult to differentiate between effusive-constrictive pericarditis and pericardial tamponade in stable patients because both are associated with effusions.71 Fortunately, distinguishing between these diagnoses is less important in hemodynamically unstable patients because they are treated identically (i.e., with pericardiocentesis).72 Pneumopericardium is an interesting, though rare cause of cardiac tamponade. It is most commonly associated with pneumothorax caused by barotrauma (e.g., mechanical ventilation).73 It also occurs spontaneously during acute asthma exacerbations,74 and it can follow blunt chest injury.75,76 Although typically benign, tension pneumopericardium has been reported as a cause of life-threatening tamponade after blunt77,78 and penetrating chest trauma.79,80 Diagnosis of pericardial effusions requires integration of the patient’s history, findings on physical examination, and diagnostic testing. Unfortunately, even experienced clinicians may not initially consider pericardial effusion because the clinical findings are often vague and nonspecific. Nonspecific symptoms, such as chest pain and dyspnea, can be ascribed to more common conditions (e.g., CHF or pulmonary pathology), so the diagnosis might be delayed until diagnostic testing is performed (e.g., computed tomography [CT] of the chest for pulmonary embolism)81 or until hypotension develops and bedside ultrasound is performed.82 Acute pericardial tamponade (e.g., secondary to blunt chest wall trauma) is usually challenging to diagnose because the findings on physical examination may resemble those of other life-threatening conditions (e.g., tension pneumothorax, hemothorax, hypovolemia, pulmonary edema, severe contusion of the RV, aortic dissection, or pulmonary embolism).67 In hemodynamically unstable patients, diagnostic (e.g., bedside ultrasound) and therapeutic (e.g., pericardiocentesis) interventions must be performed even with a paucity of findings on physical examination because rapid clinical deterioration and cardiac arrest can occur before a definitive diagnosis can be made. Once a pericardial effusion is suspected (or diagnosed), the next step is to determine its size and hemodynamic significance and presence of underlying or associated diseases.83 Specific therapy will hinge on this information and is discussed in the following sections. The historical features of pericardial effusions are nonspecific and the diagnosis may be overlooked initially. However, an astute clinician might be suspicious based on comorbid conditions (e.g., warfarin therapy or a history of myxedema) and the time course of the symptoms (e.g., free-wall rupture several days after MI, dyspnea in a patient with uremic pericarditis). Box 16-2 lists important details to be ascertained from the history when pericardial effusion is suspected.84 In 1935, Beck characterized the physical manifestations of tamponade with two triads—one for chronic and one for acute tamponade.85 Beck’s “chronic” triad consists of increased central venous pressure (CVP) (i.e., distended neck veins), ascites, and a small, quiet heart. “Beck’s triad” is a classic description of acute cardiac compression, which includes increased CVP, decreased arterial pressure, and muffled heart sounds. Almost 90% of patients have one or more of these “acute” signs,86 but only about 33% demonstrate the complete triad.9,87 The utility of this triad is further limited because all three signs are usually observed shortly before cardiac arrest. It would be clinically desirable to identify patients in early tamponade, before hemodynamic collapse. Unfortunately, findings on physical examination in early tamponade are nonspecific and may be indistinguishable from those of other critical diseases (e.g., septic shock, right heart failure).57 Patients initially seen in late tamponade also have nonspecific findings. They may be agitated, panic-stricken, confused, uncooperative, restless, cyanotic, diaphoretic, acutely dyspneic, or hemodynamically unstable. Such patients should undergo a brief and focused physical examination because the time between initial evaluation and full arrest may be brief. Some of the findings on physical examination associated with tamponade are described below. A more comprehensive list is presented in Box 16-3. The three stages occur sequentially and reflect the natural history of acute tamponade (Table 16-1).88 The time course within each stage varies from patient to patient. Some patients are stable within a given stage for hours, whereas others proceed through all three stages and cardiac arrest within minutes.9,88 Grade I tamponade is characterized by normal blood pressure and cardiac output with an increase in the heart rate and CVP (measured invasively with a central venous catheter). Grade II tamponade is defined by normal or slightly reduced blood pressure; CVP and the heart rate remain increased. Grade III tamponade is identified on the basis of Beck’s triad: hypotension, tachycardia, and elevated CVP. TABLE 16-1 Shoemaker System of Grading Cardiac Tamponade From Shoemaker WC, Carey SJ, Yao ST, et al. Hemodynamic monitoring for physiologic evaluation, diagnosis, and therapy of acute hemopericardial tamponade from penetrating wounds. J Trauma. 1973;13:36. Nearly all patients with tamponade have sinus tachycardia, although its specificity is low.89 The physiologic purpose of tachycardia is to maintain normal cardiac output despite reductions in stroke volume from worsening tamponade. Exceptions to the pairing of tachycardia with tamponade usually relate to the underlying cause of the effusion (e.g., myxedema) or the concomitant use of certain medications (e.g., β-blockers). Adding to the diagnostic complexity, not all patients in tamponade have a reduction in blood pressure. In fact, Brown and coworkers90 described several tamponade patients with elevated blood pressure. These patients were previously hypertensive and paradoxically had reduced systolic blood pressure following pericardiocentesis. PP is an exaggerated decrease in systolic blood pressure (>12 mm Hg) during inspiration secondary to reduced stroke volume (Fig. 16-4A).30,91,92 Patients with moderate to severe tamponade typically demonstrate PP greater than 20 mm Hg.9,86,93 Unfortunately, PP is not pathognomonic for tamponade. It is observed in other conditions, such as hypotension associated with labored breathing (secondary to extreme reductions in intrathoracic pressure [Fig. 16-4B]), severe emphysema, severe asthma, obesity, cardiac failure, constrictive pericarditis, pulmonary embolism, and cardiogenic shock.9,86,93 The absence of PP does not rule out tamponade because it can occur with several conditions: atrial septal defects, aortic insufficiency, positive pressure ventilation, loculated pericardial effusions, and elevated left ventricular diastolic pressure (e.g., poor left ventricular compliance secondary to chronic hypertension).14 Finally, PP should be interpreted with caution in patients with traumatic tamponade because it can be unreliable.94–96 In a study of 197 patients with traumatic tamponade, only 8.6% had PP.97 Neck vein distention (a surrogate for measuring CVP) occurs late in tamponade, when right-sided chambers (e.g., the RV) collapse. Neck vein distention may be obvious on examination (Fig. 16-5A), but visualization of such distention is less accurate than measuring CVP by central venous catheter or evaluation with ultrasound (Table 16-2). Patients with significant tamponade typically have a CVP of 12 cm H2O or higher.95 Finally, although initial CVP readings are useful and diagnostic when grossly elevated (e.g., 20 to 30 cm H2O),95,98 upward CVP trends can be a more sensitive diagnostic tool.95 TABLE 16-2 Noninvasive Estimation of Right Atrial Pressure with Ultrasound Based on Rudski LG, Lai WW, Afilalo J, et al. Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr. 2010;23:685.
Pericardiocentesis
Anatomy and Physiology
Pathophysiology of Pericardial Tamponade
Compensatory Mechanisms and Pericardiocentesis
Special Considerations in Patients with Pericardial Effusion and Tamponade
Epidemiology
Causes of Pericardial Effusion (Box 16-1)
Traumatic Hemopericardium
Atraumatic Hemopericardium
Nonhemorrhagic Effusions
Idiopathic Effusions
Neoplastic Effusion
Congestive Heart Failure
Radiation
HIV-Associated Effusions
Renal Failure and Uremia
Hypothyroidism
Special Considerations in Pericardial Disease
Diagnosing Cardiac Tamponade
History: Patient Profile and Symptoms
Physical Examination
Vital Sign Abnormalities
Pulsus Paradoxus
Neck Vein Distention and Elevated CVP
DIAMETER (cm) OF INFERIOR VENA CAVA
CHANGE IN DIAMETER WITH RESPIRATION
ESTIMATED RIGHT ATRIAL PRESSURE (mm Hg)
Normal (<2.1)
Decrease >50%
~3 (normal, 0-5)
Dilated (<2.1)
Decrease <50%
~8 (normal, 5-10)
Dilated (>2.1)
Decrease >50%
~8 (normal, 10-15)
Dilated (>2.1)
Decrease <50%
>15
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Pericardiocentesis