Chapter 68Pathophysiology of Tendon Injury
Functional Anatomy of Equine Tendons and Ligaments
Tendons passively transfer force generated by muscle to bony attachments on the opposite side of a joint, or joints, to provide movement. In contrast the function of a ligament is to resist distraction of its two bony attachments (e.g., collateral ligaments and suspensory ligament [SL]). Although this function is true for most tendons and ligaments, the horse has evolved its digital flexor tendons and SL to exhibit additional functions. These tendons and ligaments, situated on the palmar distal aspect of the equine limb (Figure 68-1), receive large weight-bearing loads because of the hyperextended metacarpophalangeal and metatarsophalangeal joints. As a result the tendons and ligaments on the palmar aspect of the distal limb act to support the metacarpophalangeal and metatarsophalangeal joint during normal weight bearing.
In addition, the equine digital flexor tendons exhibit considerable elasticity that is used to store energy for energy-efficient locomotion.1 In the case of the superficial digital flexor tendon (SDFT), its muscle is highly pennate (the muscle fibers are arranged at an oblique angle to the line of pull of the muscle, which maximizes power and minimizes contraction distance) and is unable to contract by more than a few millimeters.2 Therefore the action of the muscle, together with its accessory ligament, is largely passive to fix the origin of the SDFT in space. Although the muscle contracts only a short distance, its action, together with the tendon elasticity, also provides shock absorption.2 The gait of a horse at speed can be compared with a weight (the horse’s body) bouncing up and down on elastic springs (the digital flexor tendons and SL) in a similar fashion to a pogo stick’s bouncing.3 This arrangement allows horses to reach and maintain high speeds while minimizing energy expenditure (see Chapter 26).
Functional Characteristics
Biomechanical Properties
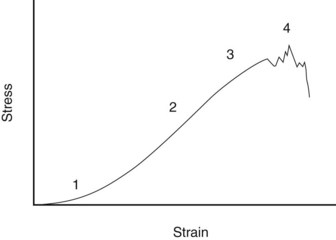
When a tendon is loaded, it stretches. The relationship between the force and the elongation defines the structural properties of the specific tendon or ligament. Because these properties depend on the size of the structure, comparison between tendons and ligaments is better made from the material properties of the tissue, which are determined by plotting the force per unit area (stress) against the percentage elongation (strain). A stylized example of such a stress-strain curve for tendon is shown in Figure 68-1. The curve has the following four regions:
Biomechanical Parameters
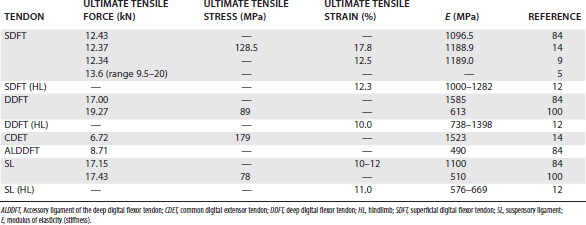
A number of simple biomechanical parameters, which are derived from their structural and material properties, can be ascribed to tendons. Some of the values of these parameters for the palmar supporting structures of the distal limb are shown in Table 68-1. A parameter not frequently calculated, but probably more relevant to the in vivo situation, is the force or stress at the yield point, after which irreversible damage is occurring.
Material Properties
The property, ultimate tensile strain, is the percentage extension of the tendon at its breaking point. In vitro testing of equine digital flexor tendons indicates that they usually extend by 10% to 12% of the original length before they rupture, although values of up to 20% have been reported.8 However, the ultimate tensile strain reflects only the final strain before rupture and includes that yield portion of the stress-strain curve that represents irreversible damage to the tendon tissue (see Figure 68-1). In addition, the ultimate tensile strain is not constant along the length of the SDFT in vitro9; the highest ultimate tensile strain occurs in the metacarpal region (the region most frequently injured).
Hysteresis
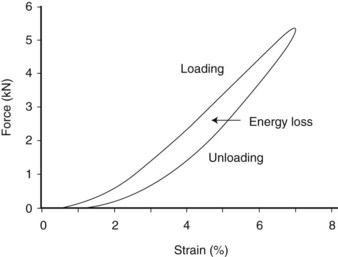
Hysteresis refers to the energy loss between the loading and unloading cycles of tendon (Figure 68-2), determined from the area between these two curves. Hysteresis is usually about 5% in equine tendons.12 Some of this energy is responsible for the rise in temperature within the tendon core associated with repeated loading (as in an exercising horse), which has been suggested as a causative factor in equine superficial digital flexor tendonitis13 (see the following discussion).
Anatomical Structure
Associated Structures
Blood Supply
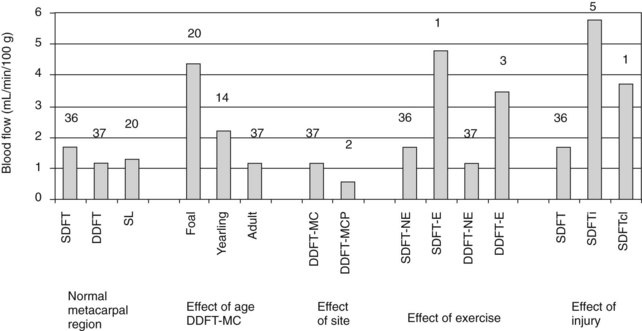
Differences in blood flow between the SDFT and DDFT are affected by age, exercise, and injury (Figure 68-3).24 The SDFT has a slightly higher blood flow than the DDFT, which reflects its good vascular anatomy (see the previous discussion). However, studies showed similar functional blood flow throughout the metacarpal region of the SDFT, although histologically and microangiographically the middle and distal regions were less well vascularized.25 However, not surprisingly, the DDFT in the metacarpophalangeal joint region has a significantly lower blood flow, associated with its fibrocartilaginous phenotype, with few blood vessels because of the high compressive forces in this region, which would limit any blood flow.
Cellular Components
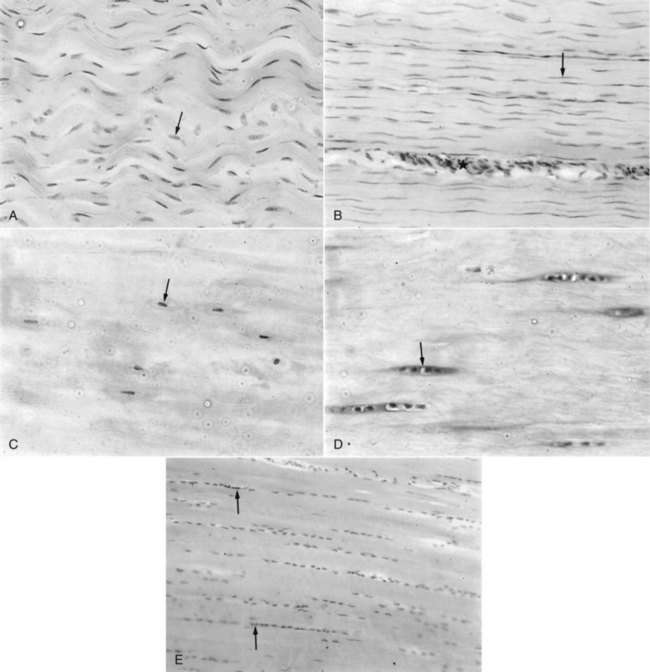
Although the biomechanical characteristics of tendon are determined by the composition and organization of the extracellular matrix, tenocytes are essential for the formation and maintenance of tendon tissue. At least three different populations of tenocytes are identifiable within the fascicles of normal equine tendon and ligament8,26 (Figure 68-4):
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree