Chapter 2. Pathologic mechanisms in ocular disease
CHAPTER CONTENTS
Objectives9
Fundamental cellular and tissue responses to injury9
OBJECTIVES
• To introduce the terminology and principles used in the morphologic interpretation of general tissue responses to disease
• To present, in a simplified manner, general pathology concepts that need to be considered when interpreting pathological changes in eyes on a routine basis
• To provide an overview of the mechanisms of disease. It is envisaged that the student may return to this chapter when, for example, evaluating a globe with suppurative inflammation, in order to better understand the morphologic features that are used in making that interpretation
• The goal is not to present a detailed discussion of the most basic principles of general pathology, such as cell degeneration or inflammation, which are addressed adequately by other texts.
FUNDAMENTAL CELLULAR AND TISSUE RESPONSES TO INJURY
Pathologic mechanisms in ocular disease are essentially the same for human and veterinary pathology. How they are grouped and taught is largely a matter of style and experience. It can be stated at the outset that the cells comprising the tissue can react to adverse events in several ways, which may be sequential. Depending on the severity and duration of the insult, cell injury may be reversible or irreversible.
The initial response is an attempt at adaptation, with potentially reversible injury, but culminating in cell death if the insult is not remediated.
Even under normal conditions, cells attempt to maintain a steady state, in response to a changing environment; these environmental factors include: changes in workload, levels of hormonal stimulation, and exposure to growth factors and other factors. Comparable adaptive mechanisms also operate in many pathologic conditions, the affected cells becoming altered but remaining viable. These mechanisms include atrophy, hypoplasia and aplasia, hypertrophy and hyperplasia, metaplasia; and dysplasia.
Comparative Comments
Comparative Comments
The principles discussed in this chapter, regarding cellular degeneration and death, tissue degeneration, inflammation and immunobiology, and abnormalities of cellular or tissue development, are identical in human and veterinary pathology. Similarly, the specific examples of cellular adaptation and reversible and irreversible cell injury in the eyes of humans are extremely similar to those given for other animals’ eyes. The causes of injury are multiple, with ischemia and toxicity accounting for most cases in humans.
CELLULAR DEGENERATION AND DEATH
Cellular degeneration
Intracellular edema/hydropic degeneration. Examples include:
• Corneal epithelial cells in corneal edema
• Lens fiber swelling in osmotic cataract
• Toxic change in feline photoreceptors in acute fluoroquinolone toxicity (Fig. 2.1).
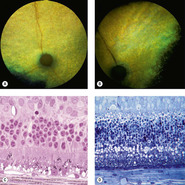 |
| Figure 2.1 Cellular degeneration from fluoroquinolone (enrofloxicin) retinal toxicity in cats. (A,B) Fundus photographs illustrating the clinical appearance of the affected retina. (A) DSH, 14 years old: the temporal hyperreflective tapetum in the left eye is totally void of vessels. (B) DSH, 10.5 years old: blind for 3 months; severe tapetal hyperreflectivity is present. (C) Photomicrograph of the retina showing degeneration of the photoreceptors 90 days after first receiving a toxic dose of fluoroquinolone. (D) Photomicrograph of the retina 3 days after administration of a known toxic dose. There is profound cellular vacuolation of the photoreceptor cells. |
Cytoplasmic lipid accumulation/fatty degeneration. Examples include:
• Keratocytes in corneal lipid degeneration (Fig. 2.2)
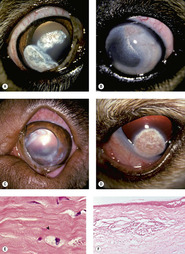 |
| Figure 2.2 Canine corneal lipid degeneration. (A–D) Clinical photographs of canine corneas with lipid deposits. (A) Golden Retriever, 9 years old: white elevated lipid deposits are present bilaterally with fine superficial vessels. (B) Golden Retriever cross, 3.5 years old: lipid deposits can be seen encircling the limbus 360° in this hypothyroid dog. (C) Dogue de Bordeaux, 3 years old: this dog was hypothyroid and has bilateral lipid deposits with superficial corneal vessels. (D) Cockapoo, 5 years old; Axial unilateral lipid deposits with corneal vascularization are present. (E) Photomicrograph showing keratocytes with lipid vacuoles (arrow) in the cytoplasm. (F) Photomicrograph of a superficial stromal cholesterol granuloma. |
• Vascular smooth muscle in atherosclerosis.
Hemosiderosis involves accumulation of an intracytoplasmic protein and iron conglomerate that results from the breakdown of hemoglobin. Hemosiderin appears as a brown pigment by microscopy and has a rusty red appearance when seen grossly. Examples include:
• Hemosiderin-laden macrophages, which accumulate within the globe in response to hemorrhage (Fig. 2.3)
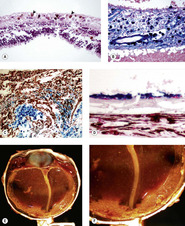 |
| Figure 2.3 Hemosiderosis of ocular tissues. (A) Hemosiderophage cells in the canine retina (arrows). (B) Prussian blue stain of retinal hemochromatosis. (C) Prussian blue stain showing hemosiderophage cells in the iridocorneal angle of a dog. (D) Prussian blue stain showing hemochromatosis of the retinal pigment epithelial cells in a canine eye with longstanding hemorrhage. (E,F) Gross photograph of a canine globe with extensive accumulation of hemosiderin-laden macrophage cells (hemochromatosis), as a result of longstanding hemorrhage into the globe. |
• Hemosiderin also accumulates in retinal pigment epithelial cells, ciliary epithelial cells, and in the neurosensory retina following intraocular hemorrhage
• Iron may bind directly to the tissue, especially in the basement membranes of the retinal blood vessels.
Lipofuscinosis involves the intracellular accumulation of a conglomerate containing oxidatively modified lipids. Lipofuscin appears as a light brown granular accumulation in the cytoplasm of affected cells and exhibits autofluorescence. Lipofuscin accumulates as a result of aging or under oxidative stress. Within ocular tissues, lipofuscin is most commonly seen in the retinal pigment epithelium. Examples include:
• Accumulation of lipofuscin in retinal pigment epithelial cells in vitamin E deficiency, or central progressive retinal atrophy/retinal pigment epithelial dystrophy (Fig. 2.4)
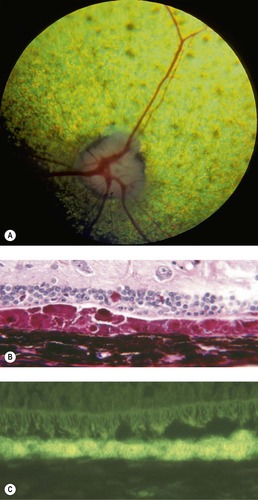 |
| Figure 2.4 Lipofuscinosis. (A) English Cocker Spaniel, 3.5 years old: fundus photograph of a dog with canine lipofuscin retinopathy, characterized by lipofuscin accumulation in the RPE. Subtle tapetal hyperreflectivity with subjective attenuation of vessels is present with multiple focal areas of tapetal pigmentation. (B) PAS stain showing abundant PAS-positive granular lipofuscin in the RPE and loss of photoreceptors. (C) Autofluorescent lipofuscin appears in the RPE when a non-stained section is examined with fluorescent light. |
• Cytoplasmic swelling in lysosomal storage diseases. There are many hereditary lysosomal storage diseases that affect the eyes of animals. These genetic disorders affect lysosomal enzymatic degradation and, for that reason, lead to the intracellular accumulation of the metabolic by-product that cannot be processed (Fig. 2.5).
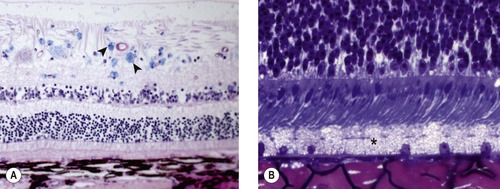 |
| Figure 2.5 Lysosomal storage disease. (A) Photomicrograph of a canine retina stained with Luxol-fast blue showing accumulation of storage product in ganglion cells and glial cells (arrows). (B) Photomicrograph of a bovine retina showing accumulation of storage product in the retinal pigment epithelium (*) in mannosidosis. |
Necrobiosis-physiologic cellular degeneration and death of cells
Examples include:
• Lens epithelial cells in the formation of the lens nucleus during normal lens development
• Epithelial keratinization.
Necrosis (oncosis)
The irreversible changes preceding cell death and necrosis are multiple, but certain key elements in all forms of potentially lethal cell injury can be identified: an influx of ionic calcium, a depletion of energy-providing enzyme systems, and an increase in cell membrane permeability.
Such irreversible defects ultimately result in cell death and necrosis through autolysis, as lytic enzymes are released from degenerating lysosomes and as cellular proteins are degraded. Necrosis is characterized by cytoplasmic expansion, followed by disruption of the nucleus, cellular swelling, membrane rupture, loss of organelles, mineralization of the mitochondria, and eosinophilic cytoplasm. Necrosis eventually excites an inflammatory reaction, as the degradative products escape outside the cell. Examples include:
• Necrosis of ganglion cells in the earliest stages of canine glaucoma (Fig. 2.6)
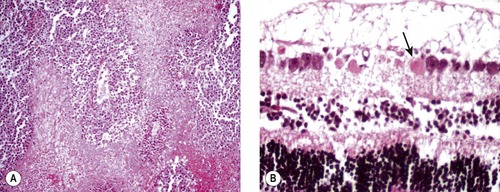 |
| Figure 2.6 Cellular necrosis. (A) Photomicrograph of feline post-traumatic sarcoma, round cell variant. Surviving neoplastic cells are only around blood vessels, and cellular necrosis occurs away from vessels. (B) Necrotic ganglion cell profiles (arrow) in acute canine glaucoma are characterized by excessive eosinophilic staining. |
• Necrosis of neoplastic cells deprived of blood supply
• Infarction of retinal tissue after vascular compromise in hypertensive vasculopathy.
Apoptosis (programmed cell death)
In contrast to necrosis, apoptosis is an essentially physiologic means of removing redundant or damaged cells on an individual basis, although it is increasingly being recognized as a feature of a wide variety of pathologic processes. The process of apoptosis is important in the regulation of tissue growth, normal development, and the elimination of damaged and potentially neoplastic cells.
Cells undergoing apoptosis are phagocytosed and removed without exciting a significant leukocytic response. Apoptosis implies the loss of individual cells within viable tissue and is characterized by nuclear pyknosis, autophagocytosis, and rapid digestion of cellular elements by surrounding tissues.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


