Other Viruses
Hepeviridae, Hepadnaviridae, Deltaviruses, Nodaviridae, and Unclassified Viruses
Abstract
This chapter describes the properties of other viruses, including hepeviruses, hepadnaviruses, nodaviruses and unclassified viruses, and features of the diseases they cause in animals.
Keywords
Hepevirus; hepadnavirus; Nodaviridae; Delatvirus; Hepeviridae; Orthohepeviruses
Inevitably, viruses with distinctive or unique properties are identified that do not readily fit into the existing taxonomic organization of major virus families. Some of these agents are identified because of the important disease syndromes with which they are associated, or because they are emerging zoonoses, whereas the pathogenic significance of many others is uncertain. The advent of next generation sequencing techniques (metagenomics) has already resulted in the identification of many new viruses, as well as increased genetic diversity of known ones.
Members of the Family Hepeviridae
Properties of HEPEVIRUSES
The family Hepeviridae currently includes two genera: the genus Orthohepevirus with four separate species (designated as Orthohepeviruses A–D) that include the so-called “hepatitis E-like viruses”—reflecting the sequential designation of human viral hepatitis—from mammals and birds, and the genus Piscihepevirus with a single virus species from fish. The genus Orthohepevirus includes Orthohepevirus A (the type species of hepatitis E virus), including hepeviruses from humans, domestic and wild pigs, deer, mongoose, rabbits, and camels; Orthohepevirus B, which includes the avian hepeviruses; Orthohepevirus C, which includes strains of hepevirus from rats, Asian musk shrews, ferrets, and mink; and Orthohepevirus D, which includes hepeviruses from bats. The genus Piscihepevirus includes only Piscihepevirus A, which is more commonly known by its historic name of cutthroat trout virus.
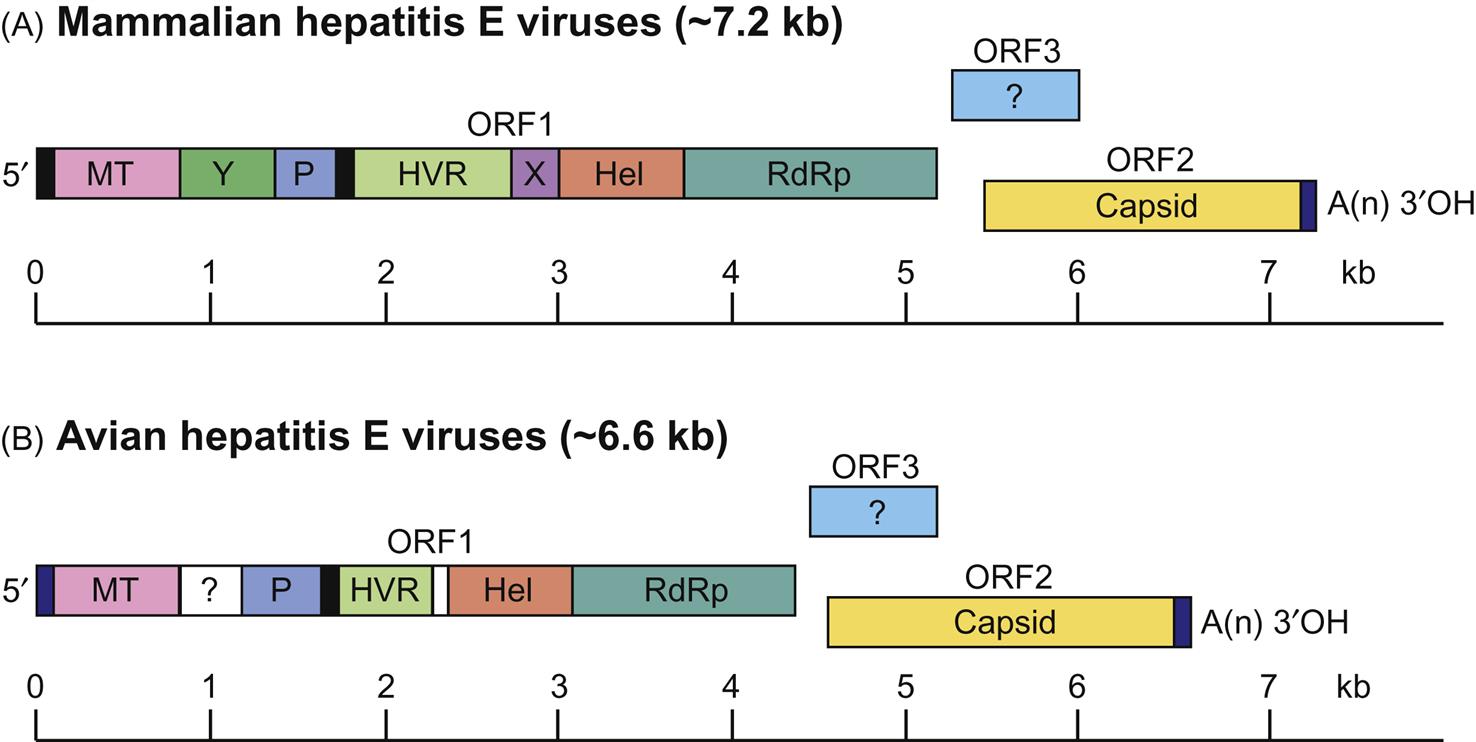
Members of the family Hepeviridae superficially resemble members of the family Caliciviridae due to their similar virion morphology and genome organization, although viruses within the Hepeviridae and Caliciviridae are genetically distinct. Hepevirus virions consist of a single capsid protein and are nonenveloped, 27–34 nm in diameter, and have icosahedral symmetry. The genome is a positive-sense, single-stranded RNA molecule of approximately 7.2 kb that includes a 5′ cap, a 3′-poly (A) tail, and three open reading frames (ORFs) (Fig. 30.1). ORF1 encodes a polyprotein that is involved in virus replication and contains several functional enzymatic domains including methyltransferease, helicase, papain-like cysteine protease, and RNA-dependent RNA polymerase. ORF2 encodes the capsid protein that is immunogenic, and ORF3 encodes a small multifunctional phosphoprotein that is involved in virus replication and assembly.
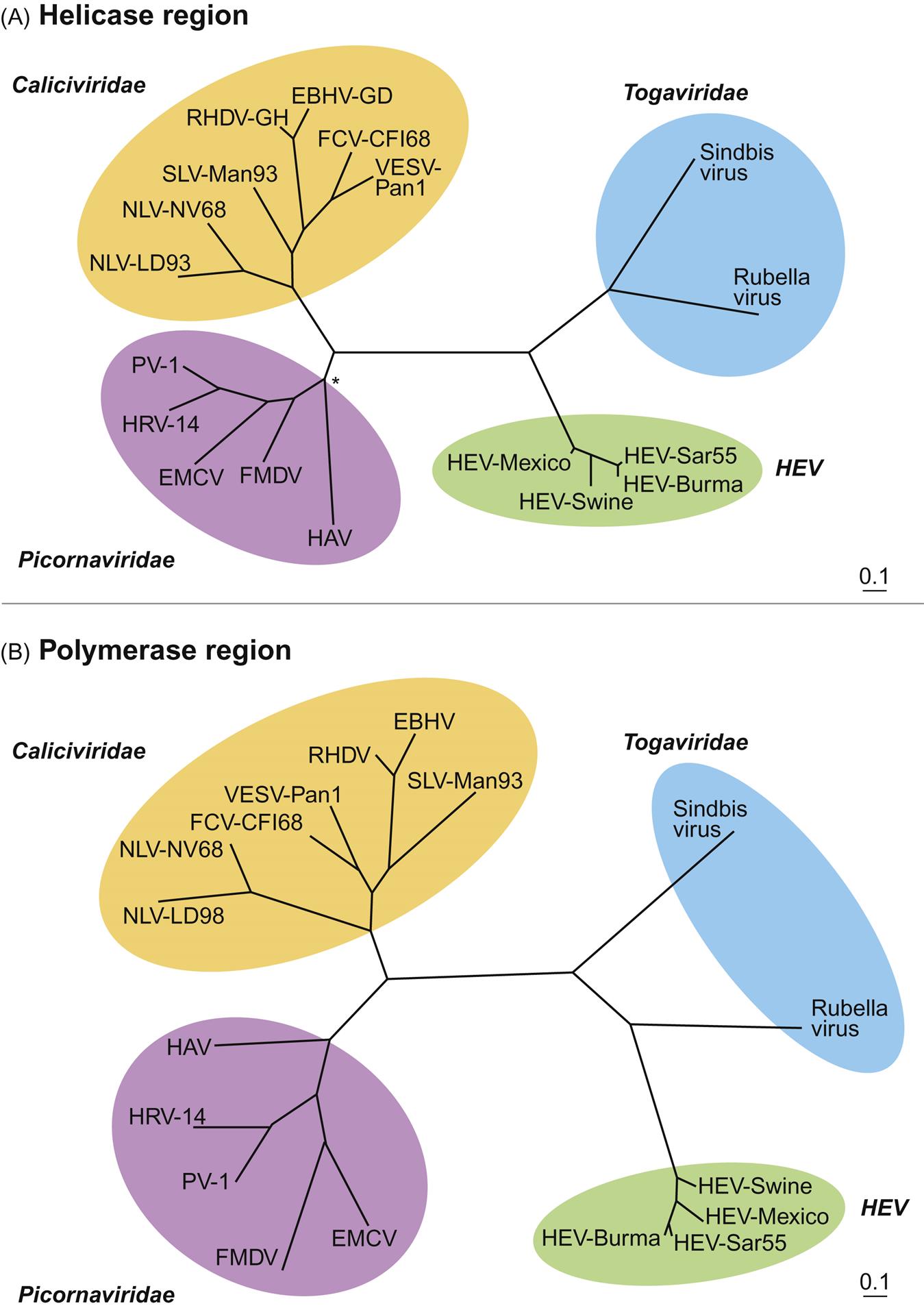
The replicative process of hepeviruses is only poorly characterized because of the lack of an efficient cell culture system, but it is thought to be similar to that of rubella virus and alphaviruses in the family Togaviridae (see Chapter 28: Togaviridae). Phylogenetic analyses of the portions of the genome encoding the helicase and polymerase confirm that strains of hepevirus segregate in a distinct clade, but are distantly related to members of the Caliciviridae, Togaviridae, and Picornaviridae (Fig. 30.2). Heparin sulfate proteoglycans likely serve as a general attachment receptor of the viral capsid protein, and virus entry into cells may be mediated by the heat shock cognate protein 70. After uncoating, viral genomic RNA is released and translation of viral nonstructural proteins is initiated and the resulting viral RNA-dependent RNA polymerase protein is used to produce progeny virions. Both ORFs 2 and 3 are translated from a single bicistronic subgenomic RNA. Hepevirus replication produces an intermediate negative-sense RNA that occurs in both the liver and extrahepatic tissues of experimentally-infected rhesus macaques, pigs, and chickens.
HUMAN HEPATITIS E VIRUS (ORTHOHEPEVIRUS A)
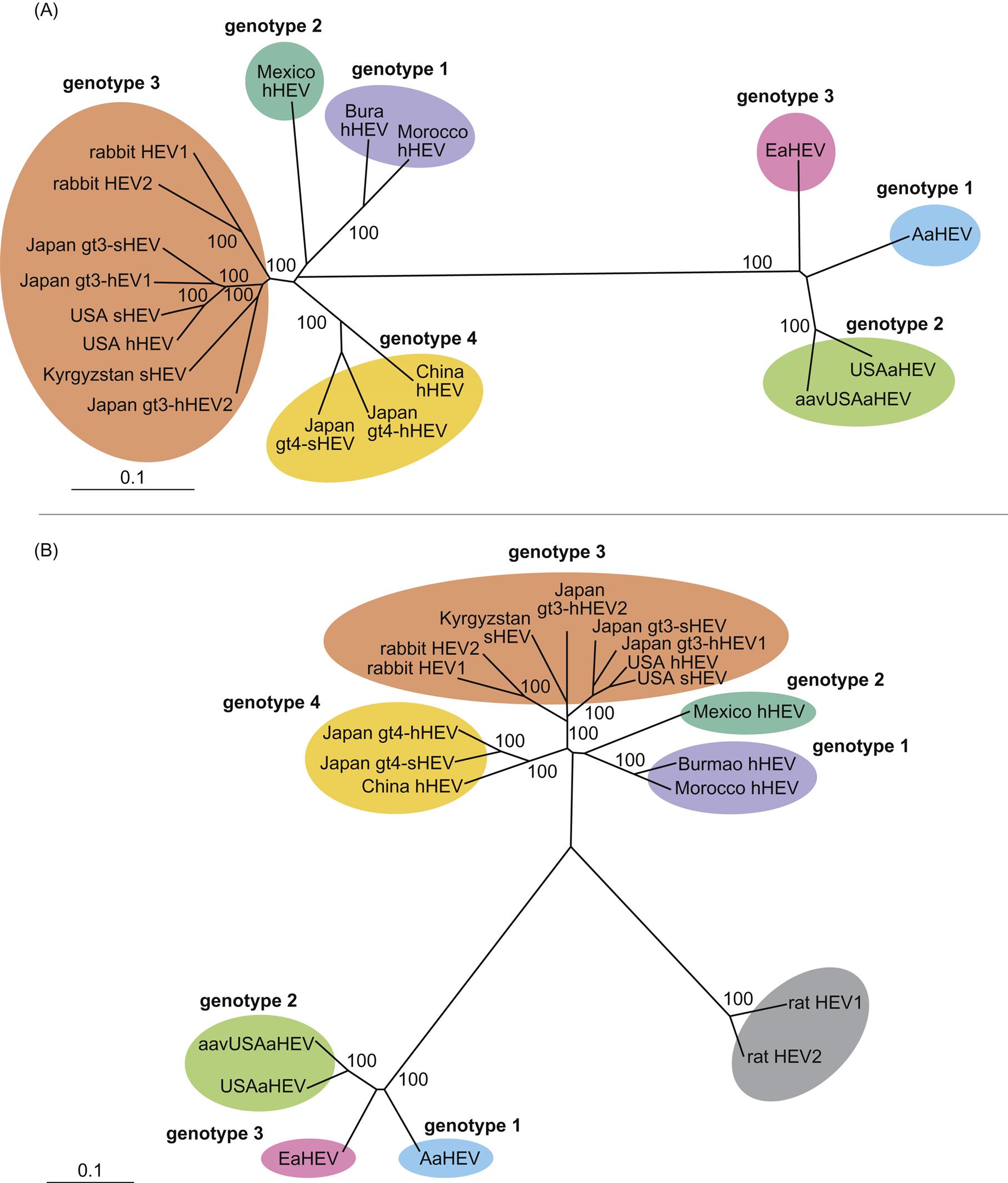
Hepatitis E virus (orthohepevirus A) is the cause of outbreaks and individual instances of acute, sometimes fatal hepatitis in humans worldwide. There are at least four distinct genetic groups (designated genotypes 1–4) of hepatitis E virus (orthohepevirus A; Fig. 30.3) that infect humans, with genotypes 1 and 2 having been associated with extensive disease outbreaks and epidemics in developing countries, and genotypes 3 and 4 with sporadic cases of hepatitis E in both developing and industrialized countries. The virus is readily transmitted during outbreaks by fecal contamination of water or food, typically because of poor sanitation. Genotypes 1 and 2 are restricted to humans whereas genotypes 3 and 4 are potentially zoonotic and infect humans as well as several other animal species including pigs, cattle, rabbit, and deer. Genotype 3 infections can progress to chronic and persistent hepatitis E in solid organ transplant recipients and other immunocompromised individuals, and are also linked to neurological disorders such as the Guillain-Barré syndrome.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree