Chapter 46 Nonsteroidal Anti-inflammatory Drugs in Raptors
Classification and Mechanism of Action
NSAIDs inhibit one or both of the cyclooxygenase (COX) enzyme systems involved in prostaglandin synthesis. COX-1 enzymes are constitutively expressed and their primary function is homeostasis through the synthesis of physiologic prostaglandins, including those involved in the protection of the gastric mucosa, renal function, and platelet aggregation.8 As such, COX-1 inhibition contributes primarily to the adverse effects of gastric bleeding, erosions, and ulcers. COX-2 enzymes are inducible and involved with synthesis of prostaglandins that initiate inflammation, including pain, swelling, and stiffness. COX-2 enzymes are the primary therapeutic targets for NSAIDs, and selective inhibitors of COX-2 have the advantage of inhibiting inflammation while sparing the adverse effects associated with COX-1 inhibition.8 Thus, attempts have been made to develop NSAIDs that are highly or preferentially selective for COX-2 enzymes. Most of the NSAIDs important to avian medicine are nonselective (e.g., diclofenac, flunixin meglumine, ketoprofen) or partially selective (e.g., carprofen, meloxicam) for COX-2 enzymes. However, the degree of selectivity can vary widely among species. Also, renal toxicity, the most significant adverse effect in birds, can occur with COX-2 preferential drugs.2
Therapeutic Uses in Captive Birds of Prey
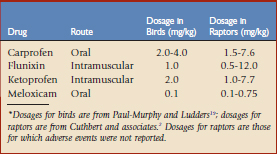
Anecdotally, the NSAIDs commonly used in raptors are similar to those used in other birds. This has been substantiated by a recent survey of veterinarians, zoos, and wildlife rehabilitation facilities that found that in descending order of use, the most commonly used NSAIDs in various raptors are meloxicam, carprofen, ketoprofen, and flunixin meglumine.2 Other NSAIDs used rarely include acetylsalicylic acid, ibuprofen, and phenylbutazone. The doses used in these studies were comparable to those previously published for avian use in general19 (Table 46-1).
No studies have been conducted in raptors that demonstrate the efficacy of analgesia with NSAID therapy. However, studies with naturally lame chickens treated with carprofen10 and with experimentally-induced arthritis in parrots (Amazona ventralis) treated with meloxicam1 have demonstrated evidence of successful analgesia at doses comparable to the usual doses used in raptors. Therefore, it is reasonable to expect that some analgesia can be expected from NSAID use in raptors although, based on work in other birds, the actual level of analgesia achieved is likely to be dose-1 and possibly species-10,20 dependent.
NSAID therapy in mammals is well known to have a number of adverse effects, including ulceration of the gastrointestinal tract, decreased platelet function, and impairment of renal function. Much less is known about the adverse effects of NSAIDs in birds. There are some reports of possible gastrointestinal disturbance and bleeding associated with the use of flunixin meglumine in budgerigars (Melopsittacus undulatus) and a crane.19 Flunixin meglumine has also been associated with localized muscle necrosis when used parenterally in mallard ducks (Anas platyrhynchos).9 However, the most common adverse effect that appears to be associated with NSAID use in birds, including raptors, is renal impairment. Unfortunately, this can be severe and rapidly lead to renal failure, visceral gout, and death. In raptors, the NSAIDs with the most reports of renal toxicity were flunixin meglumine (7 of 24 birds treated) and carprofen (5 of 40 birds treated); there were no reports of renal toxicity with ketoprofen (20 birds treated) or meloxicam (739 birds treated).2 Despite the apparent lack of problems with ketoprofen, published12 and anecdotal14 reports in other avian species have suggested that this drug may be toxic, which has been substantiated in more recent studies in Gyps vultures.17 NSAID toxicity is generally dose-dependent, although this was not evident in the above raptor survey, in which toxicity was not well correlated with higher dose ranges. However, dose-dependent toxicity is consistently seen in controlled studies, including studies of Gyps vultures treated with ketoprofen17 or diclofenac,14,24 and in other birds with ketoprofen.21 Consequently, great caution should be exercised when treating raptors at the higher end of or beyond established dose ranges. Preexisting dehydration or renal disease are additional risk factors for NSAID-induced renal toxicity in humans, and dehydration is suspected as a complicating factor in the renal toxicity of ketoprofen in birds.12,13
Of more concern with the clinical use of NSAIDs in birds, however, is the marked difference in toxicity among different groups of birds, making adverse events potentially very unpredictable. In spectacled eiders (Somateria fischeri) and king eiders (S. spectabilis) treated with ketoprofen, even gender differences in renal toxicity have been noted.12 Species differences in sensitivity to NSAIDs have been exemplified in the diclofenac-induced decline of Gyps vultures in Asia and in the subsequent research conducted to mitigate the problem.
Diclofenac and Vultures
Diclofenac is a relatively early NSAID that was, and still is, very commonly used in human medicine, primarily for rheumatoid and nonrheumatoid arthritis. After this drug was no longer covered by patent, generic forms became widely available in Asia and, in the early 1990s, found their way into veterinary medicine in India.18 Diclofenac acts on both COX-1 and COX-2 enzymes and, in humans, although quite effective therapeutically, the drug is associated with substantial adverse effects on the gastrointestinal tract, platelet function, and renal function. In livestock, however, diclofenac appeared to be an ideal veterinary drug—it was cheap and perceived to be effective and safe for clinical use in mammals. Thus, diclofenac became widely available to and widely used by veterinarians in India.
Anecdotal and newspaper reports of disappearing vultures in India began to appear in the mid-1990s, and the decline was scientifically documented in 1997 when biologists from the Bombay Natural History Society (BNHS) documented the decline in Keoladeo National Park in northern India. At this site, the breeding population of Oriental white-backed vultures (Gyps bengalensis) had gone from about 250 breeding pairs in the mid-1980s to none in 1999. Subsequent and more geographically comprehensive studies found that between the early 1990s and 2000 there was a decline of at least 96% for white-backed vultures and 92% each for the long-billed vultures (G. indicus) and slender-billed vultures (G. tenuirostris).18 Similar catastrophic declines were recorded in neighboring Pakistan and Nepal. In Pakistan, at the end of 2000, three breeding colonies being studied each had 420 to 760 active breeding pairs of white-backed vultures but, by 2003, two of these large nesting colonies were completely extirpated, followed by the third in 2007.4 White-backed and long-billed vultures were once regarded as probably the most common large raptors in the world but, in about a decade, and with a total loss estimated at tens of millions of individuals, these birds were listed as critically endangered.
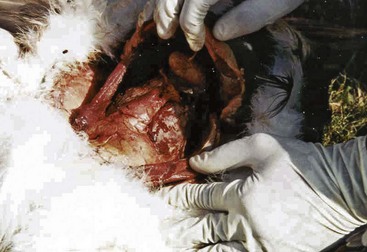
In 2003, the vulture decline in Pakistan was linked to the use of diclofenac in livestock.14,23 Key findings from ecologic studies that initially focused attention on toxins indicated that the vulture population decline was being driven by abnormal mortality in the breeding adults, and that mortality was clustered temporally and spatially, suggesting a highly lethal point source—findings most compatible with a toxic cause.4 Diagnostically, it was evident from the beginning that the abnormal mortality was associated with visceral gout (Fig. 46-1). Gross postmortem examinations done on 259 adult and subadult white-backed vultures found that 219 (85%) of these had visceral gout, indicative of renal failure. The investigation then focused on identifying the cause of renal pathology, which in wild birds would most likely be toxic or infectious. The histopathology in these affected birds was uniformly described as severe, acute, renal tubular necrosis and resultant uric acid crystal deposition in the kidneys and other tissues.11 Importantly, there were no significant inflammatory cellular infiltrates consistent with an infectious disease. Extensive testing was performed for the usual toxic causes of renal failure and/or acute death in wild birds, including the heavy metals cadmium, lead, and mercury, other metals, including arsenic, copper, iron, manganese, molybdenum, and zinc, and poisoning by organophosphate, carbamate, or organochlorine pesticides. The results of all this testing were negative.14
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree