Sarah E. Powell
Magnetic Resonance Imaging of the Fetlock Joint
Magnetic resonance imaging (MRI) has been available to equine veterinarians for more than two decades. Despite this technique becoming the gold standard for sports imaging in the human field, progress in horses has been comparatively slow. The technique initially remained the domain of a few centers worldwide, and the cost implication and need for general anesthesia meant that horses frequently were first imaged many months after the onset of lameness, when other imaging methods and treatments had been explored.
A system that used a low-field magnet but was capable of imaging standing sedated horses was released in 2000 (Figure 192-1). The lower image resolution and small imaging volume produced by the system disappointed veterinarians, who were used to images from a high-field magnet. The tradeoffs between low- and high-field systems still remain, but improved versions of the standing equine system have since been produced, most recently in 2006, that allow for a larger imaging volume, an improved range of pulse sequences, motion correction functionality, further image quality improvements, and a 50% reduction in scan times. As a result, MRI is now a financially feasible option for any equine practice with a caseload to support the investment; the system has been installed in about 50 centers around the world, and more than 33,000 clinical cases have been imaged with this system at the time of writing. The result is a near exponential increase in the number of horses undergoing MRI investigation. The challenge now is to reap the rewards of this progress by making sure that the experience and expertise gained with the earlier systems is applied to this advanced technique.
The main application of equine MRI, irrespective of magnet type and field strength, is foot imaging (see the sixth edition of Current Therapy in Equine Medicine, in Chapter 125). Information generated from the fetlock joint has been slower to emerge, in part because of the comparatively lower number of horses with lameness originating in the fetlock joint. However, MRI is proving to have great potential in horses that perform at maximum speeds, such as racing Thoroughbreds, high-level endurance horses, and horses in other disciplines that necessitate repetitive hyperextension of the fetlock joint, which makes these animals prone to load-related bone damage and stress injuries of the fetlock. Thoroughbreds are overrepresented as subjects of fetlock disease and are the primary focus of this chapter, although the mechanism of fetlock loading is common to all horses and mild forms of these injuries may be found in all breeds. Racehorses are more susceptible to catastrophic injury and end-stage disease as the fetlock joints are pushed to their biomechanical limits.
Indications and Case Selection for Fetlock Magnetic Resonance Imaging
Magnetic resonance imaging of the fetlock region is perhaps best used as a method of further evaluating known or suspected pathology, to assist in arriving at a definitive diagnosis, and to formulate the most appropriate treatment regimen. As a diagnostic modality, MRI is not a time- or cost-effective screening technique, which prohibits its use for scanning multiple areas in cases of nebulous or poorly localized lameness. With MRI, it is essential that the lameness be comprehensively localized, particularly when a low-field system is being used and the imaging volume is relatively small, and also because MRI findings do not always correlate with clinical relevance. Magnetic resonance imaging should be used in conjunction with, rather than as a replacement for, conventional imaging techniques. There is a rationale for choosing between nuclear medicine and MRI, but radiographs and ultrasound remain essential for comprehensive imaging of the joint. Not all horses are amenable to standing examinations, and it is best to make this known to the client before the scan. Examination of the hind limb is not without risk, especially in temperamental horses, during limb and coil positioning. Light sedation, to a level necessary to prevent excessive motion, is insufficient to prevent unpredictable or dangerous behavior. Racehorses in training make good candidates for standing MRI of the fetlock joint for a number of reasons. First, diagnostic imaging under general anesthesia is not practical or desirable close to a racing start or even during full race training. There is a general reluctance, particularly among owners and trainers in the United Kingdom, to anesthetize horses purely for diagnostic purposes. Second, horses in training may have a range of subchondral bone lesions, which are difficult to detect and interpret radiographically, and these disease entities cannot be distinguished on the basis of patterns of increased radionuclide uptake on nuclear medicine images. These bone injuries can be detected and differentiated with MRI, with low-field standing images being perfectly adequate to make a specific diagnosis. Horses competing in other disciplines, such as dressage and show jumping, can have similar forms of palmar osteochondral disease, condylar fissures, and proximal phalangeal fractures, but more commonly have dorsal osteochondral damage, predominantly affecting the medial condyle and medial dorsal eminence. Imaging the articular cartilage of the fetlock joint is problematic with any system, but in standing horses, articular cartilage damage has to be inferred from subchondral bone changes, with only the most severe changes being visible on clinical images. Soft tissue injuries of the fetlock region are accessible to ultrasonographic evaluation, and this remains the author’s preferred technique for their evaluation. Although MRI is occasionally useful, the presence of motion artifact, volume averaging, and a lack of contrast between the soft tissues can confound comprehensive evaluation, create lesions where none exist, and prevent detection of subtle damage to the soft tissues of this region in standing horses.
Condylar Pathology
Patterns of Condylar Densification (Sclerosis)
Altered density of the subchondral bone (SCB) within the fetlock joint represents an adaptive response to loading and is essential to accommodate exercise-associated loads when training commences at a young age. Extension of the fetlock joint in the sagittal plane during maximal weight bearing at fast gaits hyperextends the joint such that the lower aspect of the third metacarpal or metatarsal (MC3 or MT3) bone and the proximal phalanx (P1) approach right angles to each other. Contact is made between the palmar or plantar region of the condyles of MC3/MT3, the proximal sesamoid bones (PSB), and the intersesamoidean ligament, with the suspensory apparatus countering the load and restricting the range of extension. Extreme extension of the joint transfers loading to the proximal phalanx as it impinges on the dorsal articular aspect of condyles. The dynamics of this joint are common to all horses, but it is the cyclic repetition of limb loading in racing Thoroughbreds that leads to the common patterns of bone densification seen radiographically, and more recently, reported on computed tomography and MRI tests.
Subchondral bone pathology is known to play a role in the pathogenesis of osteoarthritis, osteochondral fragmentation, fractures, and SCB necrosis, which are the most common causes of fetlock lameness in all types of athletic horse. Changes in SCB are most frequently seen as a consequence of repetitive microtrauma, particularly in Thoroughbreds. Repetitive trauma leading to a progression from adaptive to nonadaptive bone modeling occurs at distinct anatomic locations within the fetlock joint—typically the palmar or plantar region of the condyles of the third metacarpal or metatarsal bones and the dorsal aspect of the distal epiphysis along a defined loading path.
Patterns of densification of the condyles of MC3 and MT3 are well recognized. The pattern, extent, and clinical relevance of subchondral bone densification in Thoroughbreds in training varies among individuals and is likely to reflect many factors, including age, conformation, and training regimen. Some degree of increased densification of the palmar aspect of the condyles is common to virtually all horses that enter training. In contrast, the densification pattern in noncompetition horses rarely goes beyond mild subchondral bone plate thickening, which rarely extends significantly into the trabecular bone.
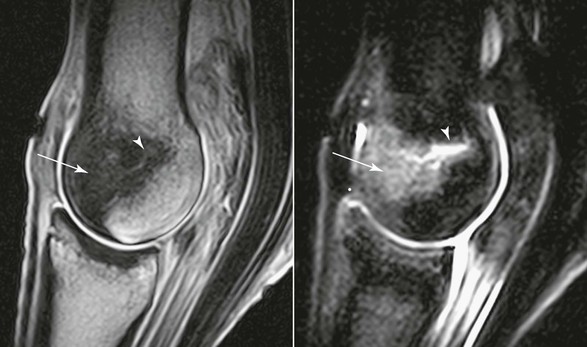
The dorsal region of the condyles is also commonly affected in racehorses and, in some cases, shows a marked degree of loading damage, densification, and inflammation, with the palmar or plantar aspect of the condyles remarkably spared (Figure 192-2). Magnetic resonance imaging has been instrumental in revealing changes in this group of horses that develops primarily “dorsal joint disease,” and would otherwise have been assumed to have palmar osteochondral disease. These horses show a combination of impact injury to the dorsal articular margin of P1, the SCB of the dorsal aspect of the condyles, and the soft tissue of the dorsal aspect of the joint, including the articular and nonarticular cartilage and capsule. In more advanced cases, the pattern of densification may extend to include the palmar or plantar aspects of both medial and lateral condyles, the dorsal aspect of the condyles, the dorsal articular margin of the proximal phalanx, and even the PSBs. Marked condylar densification is commonly seen in the absence of joint distension or other palpable abnormalities, making these joints unremarkable on clinical examination.
Areas of trabecular bone densification (condylar sclerosis) are hypointense on all MRI sequences because the marrow space between the trabeculae is obliterated and the normal signal from adipose tissue within the marrow space is lost. The most useful sequences for detecting densification as a function of loss of marrow space are those in which adipose tissue appears hyperintense, including T1-weighted, T2-weighted, and proton density sequences. Fat-suppressed or short tau inversion recovery (STIR) sequences (sometimes referred to as fluid-sensitive sequences) do not allow identification of increased bone density because the signal from adipose tissue is suppressed in these sequences.
More Advanced Stages of Condylar Pathology
Extensive densification of the condyles in racehorses is so common that making assumptions about joints on this basis alone is not practical or helpful for case management. It is logical, though, to attempt to identify horses with a greater degree of bone damage, in which bone changes have crossed the line to being maladaptive, potentially irreversible, bone pathology; in this situation, areas of increased signal within the dense bone seems to be a useful tool. Detecting focal signal increases within areas of condylar densification is pertinent in detecting and staging subchondral bone injuries in racehorses because it suggests progression from bone densification to more sinister pathologies, such as trabecular microfracture and necrosis, and detecting signal hyperintensity on fluid-sensitive sequences within, or adjacent to, the dense bone adds additional relevance. This is the basis for detecting palmar or plantar osteochondral defects and cortical fissure fractures in the metacarpal or metatarsal condyles, although the hyperintensity is likely to represent different pathologic processes in these two distinct disease entities.
A Note on Bone “Edema” or “Bruise”
It is worth noting that a “fluid” signal pattern within bone refers to the characteristic signal pattern of interosseous T2-weighted and STIR hyperintensity with corresponding T1 hypointensity. When this pattern is seen on MRI, it is widely (and frequently erroneously) referred to as “bone edema” or “bone bruise.” This characteristic pattern is observed when bone marrow is replaced by material containing hydrogen ions, in the form of water. The hydrogen ions may be contained within cells or may be free, for example, in intraosseous necrosis or hemorrhage. Causes of this signal pattern include avascular necrosis, osteochondral defects, infection, and arthritis, and use of the generic terms bone edema or bruise is misleading. Various alternatives have been suggested, of which bone marrow lesion is perhaps the most appropriate and user friendly. Bone marrow lesions have been reported in horses undergoing MRI of a number of sites, including the fetlock, and the characteristic appearance histologically represents a number of pathologic processes in horses.
Palmar or Plantar Osteochondral Disease
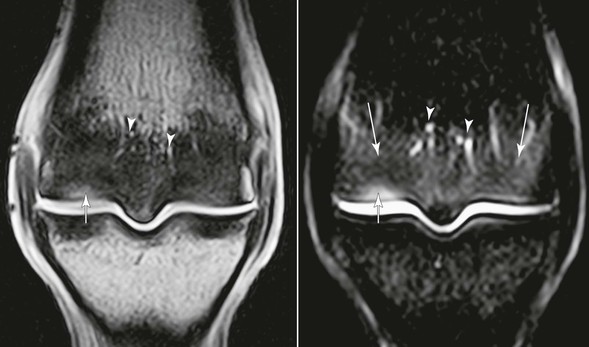
For the purposes of this chapter, palmar or plantar osteochondral disease lesions will be used to refer to a stage of palmar or plantar osteochondral disease in which areas of transverse trabecular fracture and osteonecrosis within the SCB are detected on MRI as focal hyperintensity on all sequences, surrounded by areas of increased bone density at predefined sites in the metacarpal or metatarsal condyles. Such palmar or plantar osteochondral lesions, which are seen commonly in racing Thoroughbreds, exhibit progressive changes (Figure 192-3). These lesions are distinct from condylar fissuring and parasagittal condylar fractures. Osteochondral disease lesions in the distal metacarpal or metatarsal condyles are traditionally diagnosed scintigraphically on bone-phase images as focally increased radionuclide uptake within the palmar or plantar condyle. Achieving a definitive radiographic diagnosis is difficult and may only be possible in advanced cases that have progressed to SCB collapse. Magnetic resonance imaging enables earlier detection and better characterization than that afforded by scintigraphic images, on which palmar or plantar osteochondral disease lesions and parasagittal fissure are poorly differentiated. Differentiating these two conditions is extremely important because they have different implications when assessing risk for catastrophic injury. Although difficult to assess on MRI images, the overlying articular cartilage may be largely unaffected in early lesions and, if caught early, may have the potential to resolve, at least partially.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree