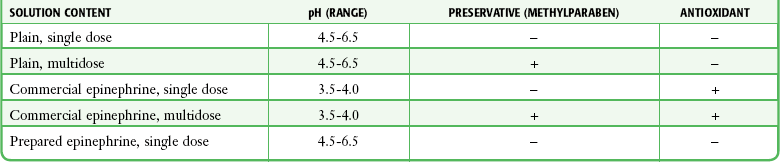
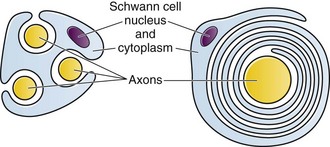
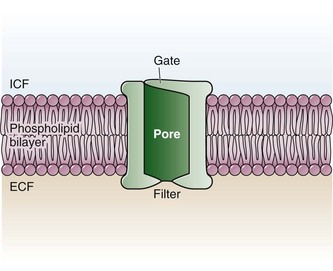
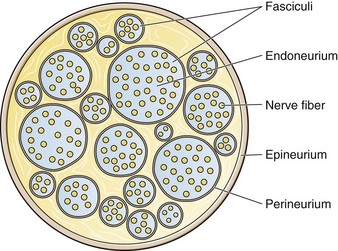
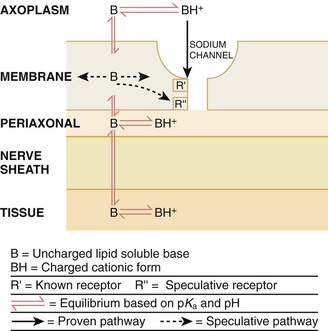
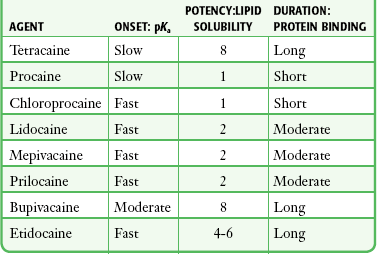
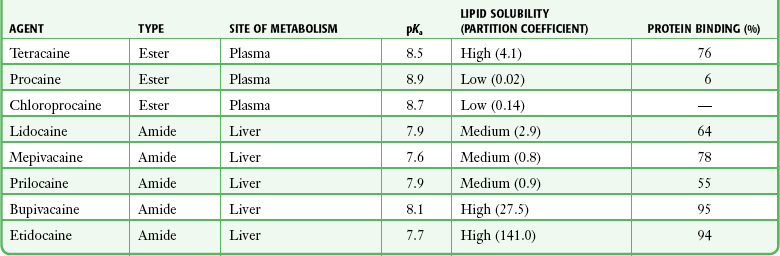
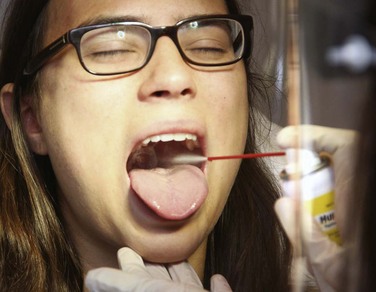
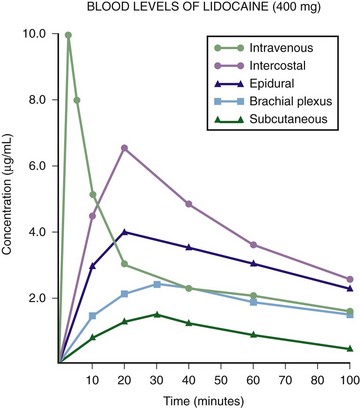
Chapter 29 The first local anesthetic was cocaine, an alkaloid in the leaves of the Erythroxylon coca shrub from the Andes Mountains. Early Incan society used cocaine for invasive procedures, including cranial trephination. In 1884, Koller1 used topical cocaine in the eye and was credited with the introduction of local anesthesia into clinical practice. In the same year, Zenfel used a topical solution of alcohol and cocaine to anesthetize the eardrum, and Hall2 introduced the drug into dentistry. In 1885 Halsted3 demonstrated that cocaine blocked nerve transmission, thereby laying the foundation for nerve block anesthesia. The search for alternatives to cocaine led to synthesis of the benzoic acid ester derivatives and the amide anesthetics used today. It was not until the 1960s that detailed understanding of the physiochemical properties, mechanism of action, pharmacokinetics, and toxicity of these agents emerged. Chemical Structure and Physiochemical Properties Most useful local anesthetic agents share a basic chemical structure: Subtle variations in this basic structure determine each agent’s main physiochemical properties: the negative log of dissociation constant (pKa), the partition coefficient (a measurement of lipid solubility), and the degree of protein binding. Each of these properties determines the drug’s potency, onset, and duration of action. However, physiochemical properties are not the sole determinant of clinical activity; other factors influence the drug’s effect. The intermediate chain between the aromatic and the hydrophilic segments is either an amino-ester or an amino-amide; these chemical structures form the basis for the two main classifications of local anesthetics. Common ester-type agents include procaine, chloroprocaine, cocaine, and tetracaine. Common amide-type agents include articaine, lidocaine, mepivacaine, prilocaine, bupivacaine, and etidocaine. Different biochemical pathways metabolize each class. Esters are hydrolyzed by plasma pseudocholinesterase. Cocaine, an ester, is also partly metabolized by N-demethylation and nonenzymatic hydrolysis. Individuals with pseudocholinesterase deficiency may have a greater potential for cocaine toxicity if large doses are used, although this has not been an issue when cocaine is used clinically as an anesthetic. Amides are metabolized in the liver by enzymatic degradation. Local anesthetics are poorly soluble weak bases combined with hydrogen chloride to produce the salt of a weak acid. In solution, the salt exists both as uncharged molecules (nonionized) and as positively charged cations (ionized). The nonionized form is lipid soluble, which enables it to diffuse through tissues and across nerve membranes. The ratio of nonionized to ionized forms depends on the pH of the medium (vial solution or tissue milieu) and on the pKa of the specific agent. The pKa is the pH in which 50% of the solution is in the uncharged form and 50% is in the charged form. When the pH of the solution or tissue is less than the pKa, more of the drug is ionized. When the pH increases, more of the drug is in the nonionized form. Because the nonionized form of drug can diffuse through tissues and nerves, manipulating the pH of the solution can alter a drug’s diffusion properties. Local anesthetics are available in single-dose vials or ampules and in multidose vials, with and without epinephrine. Most solutions have a pH higher than 5. Multidose vials contain methylparaben, an antibacterial preservative. Local anesthetics premixed with epinephrine also contain an antioxidant (sodium bisulfite or sodium metabisulfite) to prevent deactivation of the vasoconstrictor. These solutions must be adjusted to a more acidic pH, approximately 3.5 to 4.0, to maintain the stability of epinephrine and its antioxidant. These properties as they relate to the amide group are depicted in Table 29-1. Functional and Structural Components of a Peripheral Nerve The functional nerve unit includes the nerve axon and its surrounding Schwann cell sheath. The Schwann cell (Fig. 29-1) may surround several unmyelinated axons or a single myelinated nerve fiber and form a myelin sheath. Junctions between sheaths along the axon called nodes of Ranvier contain sodium channels necessary for depolarization. As myelin sheath thickness increases from autonomic to sensory to motor fibers, the nodes of Ranvier are spaced farther apart. The most important structure affecting transmission of nerve impulses is the axon membrane (Fig. 29-2). The membrane consists of a double layer of phospholipids into which are embedded protein molecules that serve as channels containing pores for the movement of ions in and out of the cell. Most pores have a filter, or gate, that controls ion-specific movement and a sensory mechanism that opens or closes the gate. Bundles of nerve fibers (Fig. 29-3) are embedded in the endoneurium, which is made of collagen fibrils, and they are surrounded by a cellular layer, the perineurium. The perineurium functions as a diffusion barrier and maintains the composition of extracellular fluid around the nerve fibers. Surrounding the entire structure is the outer layer of a peripheral nerve, the epineurium, which is composed of areolar connective tissue. How anesthetic agents prevent sodium influx is still not completely understood. It is believed that the cationic charged form blocks the action potential from inside the membrane; the agent enters the sodium channel from the axoplasmic side and binds to a receptor.4,5 This “specific receptor” theory is well accepted and is considered the predominant mechanism in preventing influx of sodium. However, this theory cannot account for the action of benzocaine and other neutral compounds or the uncharged base forms of the common local anesthetics. In summary (Fig. 29-4), when a local anesthetic (other than benzocaine) surrounds the perineurium, it equilibrates into its uncharged and charged forms based on tissue pH and pKa. In a more alkaline environment, a greater proportion of the uncharged form is present. The uncharged lipid-soluble form penetrates tissue, nerve sheath, and nerve membrane to gain access to the axoplasm and reequilibrates into both charged and uncharged forms. The charged form enters the sodium channel, decreases movement of sodium into the cell, and halts nerve transmission. The uncharged base is also involved in sodium channel blockade, but the exact nature of this mechanism is unknown. The pKa of an anesthetic is the primary physiochemical factor that determines its onset of action. Increased tissue penetration and a shortened onset of action are found in drugs with a lower pKa because more of the lipid-soluble uncharged form is present (Tables 29-2 and 29-3). Although in isolated nerve fibers the onset of action directly parallels pKa, other physiochemical factors also influence drug activity. For example, prilocaine and lidocaine have the same pKa, but lidocaine’s onset is faster because of its enhanced ability to penetrate through nonnerve tissue. The lipid solubility of an anesthetic is a primary physicochemical factor determining potency. The drug’s partition coefficient, not the concentration of the lipid-soluble form determined by the pKa or pH, confers its lipid solubility. Because the nerve membrane is lipid, lipophilic anesthetics pass more easily into the cell and few molecules are needed to block conduction (see Tables 29-2 and 29-3). The degree of protein binding of an anesthetic primarily determines its duration of action. Agents that bind more tightly to the protein receptor remain in the sodium channel longer (see Tables 29-2 and 29-3). Like potency, the duration of action is reduced by the vasodilation produced by local anesthetics. Prilocaine, which is less protein bound than lidocaine, has a longer duration of action because of its lesser degree of vasodilation. The duration of action also varies with the mode of administration. It is shorter when agents are applied topically. The duration of action may be prolonged by several methods. Increasing the dose, usually by increasing the concentration, prolongs the duration to limits imposed by toxic effects. Raising the pH of the anesthetic solution has also been shown to prolong duration.6,7 The most practical way to increase duration is to use solutions that contain epinephrine.8 Epinephrine causes vasoconstriction, decreases systemic absorption, and allows more drug to reach the nerve. The effect of epinephrine varies according to the agent. Anesthetics that intrinsically produce more vasodilation (e.g., procaine, lidocaine, mepivacaine) benefit more from epinephrine’s vasoconstrictive action. The long-acting, highly lipid-soluble agents (e.g., bupivacaine, etidocaine) are less affected because they are substantially taken up by extradural fat and released slowly. In fact, lidocaine with epinephrine may be effective for as long as bupivacaine without epinephrine. Generally, most ED procedures can be accomplished quickly before the anesthesia wears off regardless of which drug is selected. Choose agents with a long duration of action when the procedure is lengthy or if postoperative analgesia is desired. Local anesthetic agents may be applied topically to mucous membranes, intact skin, and lacerations. There are sufficient differences among these sites to merit a separate discussion of each one. Topical anesthesia of the eye is discussed in Chapter 62. Effective anesthesia of the intact mucous membranes (not intact skin) of the nose, mouth, throat, tracheobronchial tree, esophagus, and genitourinary tract may be provided by several anesthetics (Box 29-1). Tetracaine, lidocaine, and cocaine are the most effective commonly used agents (Table 29-4 and Box 29-1). Benzocaine (14% to 20%) is commonly used for intraoral or pharyngeal anesthesia (Fig. 29-5). The anesthesia produced is superficial and does not relieve pain that originates from submucosal structures. The onset of action may be slow, which limits its usefulness in urgent situations (such as passing a nasogastric tube). Agents applied topically can be absorbed systemically, and concentrated topical agents can cause toxicity. TABLE 29-4 Practical Agents for ED Use: Mucosal Application *These are conservative figures; see text for explanations. †The lower dosage should be used for a maximum safe dose when feasible. ‡The 10% cocaine solution is best avoided because of minimal additional clinical benefit and the potential for coronary vasoconstriction in patients with coronary artery disease. Cocaine is an effective, but potentially toxic topical agent that is applied to the mucous membranes of the upper respiratory tract. Although it is an ester, hepatic metabolism occurs, as does hydrolysis by plasma pseudocholinesterase. Absorption is enhanced in the presence of inflammation. Cocaine is the only anesthetic that produces vasoconstriction at clinically useful concentrations, hence its popularity for treating epistaxis. This major advantage is offset by its susceptibility to abuse and toxic potential. The toxic effects are due to direction stimulation of the CNS and blockade of norepinephrine reuptake in the peripheral nervous system. Cocaine should not be administered to patients who are sensitive to exogenous catecholamines or who are taking monoamine oxidase (MAO) inhibitor antidepressants. Clinical manifestations of toxicity include CNS excitement, seizures, and hyperthermia. Central and peripheral effects of hypertension, tachycardia, and ventricular arrhythmias may also be seen. Acute myocardial infarction has been reported after topical application.9 Cocaine is commonly used as a 4% solution with a maximum safe dose of 200 mg (2 to 3 mg/kg). A 10% solution is available, but this concentration adds little to the topical effect while enhancing the potential for toxicity. Coronary vasoconstriction may occur with doses as low as 2 mg/kg applied to the nasal mucosa. Although the clinical effect of this vasoconstriction is usually benign and without electrocardiographic changes, topical cocaine should be used cautiously in patients with known or suspected coronary artery disease. Dyclonine offers advantages over other topical anesthetic agents. It is a ketone derivative without an ester or amide linkage and may be used in patients who are allergic to the common anesthetics. Extensive experience with the topical preparation has shown it to be effective and safe.10 Dyclonine is marketed in 0.5% and 1% solutions, often in sore throat preparations, with a maximum adult recommended dose of 300 mg. Benzocaine is an ester that is marketed in its neutral form in 14% to 20% preparations (Cetacaine, Americaine, Hurricaine) (see Fig. 29-5). Its low water solubility prevents significant penetration of the mucous membranes, thus reducing systemic toxicity if applied to intact mucosa. However, it is not a potent anesthetic and has a brief duration of action. It is more allergenic than other topical agents. Benzocaine is usually dispensed in an admixture with other therapeutic ingredients and is clinically effective only at relatively high (>14%) concentrations. Benzocaine is available as a nonprescription gel and liquid (6.3% to 20% Anbesol, for example) and is used for a variety of maladies, including ear pain, mouth pain, and teething. It is commonly used by dentists to produce mucosal anesthesia before intraoral nerve blocks (see Chapter 30). Adriani and Zepernick10 recommended this agent for lubricating catheters, airways, endotracheal tubes, and laryngoscopes and reported only one adverse reaction (methemoglobinemia) in their experience with approximately 150,000 patients. Methemoglobinemia occurs rarely after mucosal absorption of benzocaine used repeatedly for teething infants and after standard doses of benzocaine sprays used in endoscopic procedures.11 An excellent topical preparation is a combination of lidocaine, prilocaine, and tetracaine, especially useful for dental mucosa anesthesia (see Fig. 30-3). Topical gel mixtures of 2.5% lidocaine and 2.5% prilocaine (eutectic mixture of local anesthetics [EMLA]) are commonly used on intact skin but have also been used on mucous membranes. EMLA is more effective than 20% benzocaine when applied to the oral mucosa before needle injection for dental anesthesia. One study demonstrated that pain was reduced more quickly with EMLA than with benzocaine when applied to the buccal mucosa.12 As with infiltrated anesthesia, toxic reactions to topically applied anesthetics correlate with the peak blood levels achieved and not necessarily with the dose administered. Systemic absorption of a topical agent is more rapid, with a higher level being achieved than with the same dose given by infiltration (Fig. 29-6). The total dose of a topical anesthetic should be considerably less than that used for infiltration at a given site. Fractionating the total dose into three portions administered over a period of several minutes effectively reduces peak blood levels. Inadvertent suppression of the gag reflex, combined with difficulty swallowing, may lead to aspiration, an important potential adverse reaction to topical anesthesia of the nose, mouth, and pharynx. Infections from drug solutions in multidose vials for topical anesthesia of the larynx and trachea have not been substantiated. A commonly used “magic mouthwash” for the topical treatment of painful gingivostomatitis in children is often prescribed by emergency clinicians and pediatricians. There has been little scientific study of the preparation and it is not available commercially, but it has been used safely for decades. It consists of equal parts viscous lidocaine (2%), Maalox as a binder, and diphenhydramine elixir. Corticosteroids or nystatin is often added when the mixture is used to treat chemotherapy-induced mucositis.13 However, a recent review found no evidence that magic mouthwash is effective in treating this condition.14 The creamy mixture is swished around the mouth and expectorated or painted on specific lesions with a cotton swab. Packing an area with a cotton ball soaked with this mixture is one option. Repeated doses or swallowing of the elixir can produce systemic toxicity, so careful instruction should be given to limit use of the solution to every few hours. This combination would be theoretically less toxic than simply using topical lidocaine. Emergency clinicians often prescribe 2% viscous lidocaine (20 mg/mL) for patients with pharyngitis, stomatitis, dental pain, or other inflammatory or irritative lesions in the oropharynx. Although this intervention is widely used and generally safe, the common misconception that topical anesthesia is totally innocuous may result in poor patient instructions and serious consequences. Topical lidocaine is helpful for painful mouth lesions but is of little practical value for acute pharyngitis, for which systemic analgesics are usually a better option. Seizures and death from topical lidocaine have been reported when excessive repeated doses have been administered.15,16 Toxic blood levels may occur because the anesthetic effect of viscous lidocaine lasts for only 30 to 60 minutes and patients with recurrent pain may either ignore or be ignorant of the safe dosing interval of 3 hours and medicate themselves more frequently. Patients tend to increase each dose to obtain greater relief, and inflammation may increase systemic absorption. In addition, painful oral lesions may last for several days. Children are at higher risk for the rare toxicity of oral lidocaine. When compared with adults, children may exhibit increased lidocaine absorption, decreased clearance, and a longer half-life.17 Continued medication use allows lidocaine and its major metabolites monoethylglycinexylidide (MEGX) and glycinexylidide (GX) to accumulate. Both MEGX and GX are produced from the hepatic metabolism of lidocaine and are excreted in urine. They possess anesthetic and antiarrhythmic activity and have the potential for CNS toxicity. Although these metabolites are less potent than lidocaine, their elimination half-lives are considerably longer. Several investigators regard MEGX and GX to be the cause of CNS toxicity with prolonged topical use of lidocaine.16 The length of time that viscous lidocaine is retained in the mouth and whether the excess is expectorated or swallowed also affect the blood level produced. Expectorating the medication after swishing it in the mouth produces much lower blood levels than when it is swallowed. It seems logical that the most hazardous mode of administration would be to retain the solution in the mouth “until absorbed.” Clearly explain the proper way to use viscous lidocaine and inform patients not to dose themselves ad libitum. Note that a 2% solution contains 20 mg/mL, or 100 mg per standard teaspoon (5 mL). The recommended maximum adult dose is 300 mg (15 mL of a 2% solution) no more frequently than every 3 hours. When possible, instruct the patient to decrease the dose by using direct cotton swab application. When gargled or swished in the mouth, limit application time to 1 to 2 minutes, and instruct the patient to expectorate excess solution. Limit use to 2 or 3 days, especially if swallowing the solution is necessary to obtain relief. Prescribe lower doses for patients at risk for decreased clearance (see “Systemic Toxic Reactions” later in this chapter). Doses for children are prescribed at 3 mg/kg. Because infants cannot expectorate well, do not use viscous lidocaine for minor oral irritation and teething. Recommend that no food be eaten for 1 hour after application because anesthesia of the oropharynx can interfere with swallowing and cause aspiration. Special note should be made of the over-the-counter availability of benzocaine, commonly used for toothaches and teething. A gel or liquid (Anbesol) is available in 6.3% to 20% formulations. When used repeatedly in the oral cavity on irritated tissue, systemic toxicity, including methemoglobinemia, may occur. Lidocaine 4% solution can be atomized with a standard nebulizer device commonly used for delivering asthma medications and inhaled by the patient before insertion of a nasogastric tube. This method effectively anesthetizes the nasopharyngeal and oropharyngeal tissues, thereby easing the pain with tube insertion.18,19 Lidocaine Cream: Lubens and coworkers20 used 30% lidocaine cream, saturated on a gauze pad and adherent to an elastic patch, for a myriad of procedures. Despite its effectiveness, safety, and painless application, the practicality of its use in an emergency setting is limited. In 1974, Lubens and coworkers20 reported an impressive list of uses, including minor operative procedures (e.g., excision of lesions, incision and drainage of abscesses), lumbar puncture, venipuncture, and allergy testing. Lidocaine remains one of the most commonly used topical compounds. EMLA Cream, ELA-Max, and Tetracaine Base Patch: Various topical anesthetics have been suggested to decrease the pain of venipuncture or injections and to provide topical anesthesia for painful skin abrasions and lesions. These agents have been studied extensively and are safe, but they are not practical in many ED settings because of their slow onset of action and inadequate efficacy. However, their activity profiles make them more applicable than 30% lidocaine cream to emergency medicine. Tetracaine base is available as a solution, a gel, and a patch preparation. It is effective in crossing the lipid-rich barrier of the stratum corneum because it is highly lipophilic. EMLA was approved in the United States in 1992. It contains 2.5% lidocaine and 2.5% prilocaine in a unique oil-and-water emulsion yielding 5% EMLA. The mild lipophilic and hydrophilic properties of the component drugs are greatly increased when mixed together, thereby allowing absorption through intact skin. ELA-Max and ELA-Max5 are topical lidocaine anesthetic creams with a more rapid onset of action than EMLA cream. ELA-Max is a 4% concentration, and 5% ELA-Max is marketed as an anorectal cream that may benefit patients undergoing painful rectal procedures. Neither product has prilocaine, as is found in EMLA cream, and neither has U.S. Food and Drug Administration approval for pain relief before painful injections or intravenous (IV) insertion, but both have such potential.21 Tetracaine base seems to offer the advantage of being able to achieve effective anesthesia with a shorter application time and a longer duration.22 For tetracaine and EMLA preparations, onset, depth of anesthesia, duration, and blood levels vary directly with application time, use on thinner or inflamed skin, and larger doses.23 These preparations exhibit a reservoir effect.24 The drug is deposited in the stratum corneum and continues to diffuse along its concentration gradient, even after it is removed from the skin. Tetracaine base, lidocaine cream, and EMLA can be useful in the ED for providing anesthesia for many procedures: venous cannulation, venipuncture, or any needle insertion, including preinfiltration anesthesia and lumbar puncture; a variety of minor surgical procedures; and anesthetizing the tympanic membrane and external auditory canal. EMLA has also been used effectively for débridement of ulcers.25 EMLA and 5% lidocaine cream are equally effective in reducing the pain of IV insertion.26 Luhmann and colleagues27 demonstrated that 5% lidocaine cream applied for 30 minutes under an occlusive dressing was as effective as infiltrated buffered lidocaine before IV catheter insertion in children. Obviously, infiltrative administration of buffered lidocaine requires skin puncture, but its onset of anesthesia is almost immediate. When time is not an issue, topical creams may be an acceptable alternative to infiltrated anesthesia or no anesthesia at all. About a 60-minute interval after application is required for these preparations to provide optimal topical analgesia to the intact skin for such procedures as venipuncture. Early cutaneous placement of these agents (e.g., over common IV sites while the patient is being triaged) is important for practical ED use. Ethyl Chloride and Trichloromonofluoromethane and Dichlorodifluoromethane (Fluori-Methane) Sprays: These topical agents are often used for limited skin incisions (e.g., drainage of small abscesses), trigger point injections, joint aspiration, or injection of bursitis or tendonitis. These agents evaporate quickly from the skin and cool it to the point of freezing. Anesthesia is effective and immediate, but drawbacks include its short duration (only up to 1 minute), potential pain on thawing, and possibly lowered resistance to infection and delayed healing. Highly volatile ethyl chloride spray is flammable. Ethyl chloride has been studied in children to reduce the pain of venipuncture, but the results are mixed.28,29 Given their short duration of action and the time needed to perform pediatric venipuncture, these preparations have limited use for this purpose. Lidocaine Cream: This 30% cream is saturated on a gauze pad that is adherent to an elastic patch and placed over the area to be injected or incised.20 The high concentration of anesthetic and an occlusive patch are needed to achieve effective skin penetration. The duration of action varies with the application time. A 45-minute application time is needed for most procedures. To achieve a topical anesthetic duration of 30 minutes, a 2-hour application is necessary. Tetracaine Base Patch and EMLA Cream: Because both agents demonstrate a reservoir effect, anesthesia may increase or begin many minutes after removal of the drug. A precise description of application times and duration is not possible. Tetracaine base requires a minimum of 20 to 30 minutes of application time to produce several hours of anesthesia. EMLA requires an application time of 1 to 2 hours for a reported duration of 30 minutes to several hours. Occlusive dressings seem to increase penetration of EMLA whenever the cream is used. Patches are more convenient and cause no loss of effectiveness. Ethyl Chloride and Fluori-Methane Sprays: Collect all the equipment required and make all preparations needed to immediately perform the desired procedure. Invert the bottle 25 cm from the skin and spray a stream along the proposed incision site until the area turns white and hard. Make the incision or local anesthetic injection immediately or during actual spraying of the agent for optimal results because the effect is fleeting. Some clinicians use these vapocoolant sprays to decrease the pain of injection of a more traditional local anesthetic such as lidocaine. Iontophoresis: Anesthetic agents may be drawn into the skin without needles by electrical current applied through electrodes in a process called iontophoresis. Lidocaine with epinephrine administered via iontophoresis provides adequate anesthesia before venipuncture in pediatric patients and is superior to EMLA in providing cutaneous anesthesia.30–33 Iontophoresis is not widely used in emergency medicine but may be another alternative to applied anesthetic agents. Microneedle Pretreatment: Recently developed technology known as microneedle pretreatment may improve the efficacy of cutaneously applied topical anesthetics. A functional microarray of fine needles painlessly perforates the stratum corneum to facilitate the penetration of applied medications without using traditional needle injections. A recent study demonstrated that perforation of the skin of healthy volunteers with a microneedle to which dyclonine was applied resulted in decreased time to anesthesia and a greater degree of pain reduction than did application of dyclonine to nonperforated skin.34 This emerging technology may improve the practical application of anesthetics to intact skin in the ED. Jet Injection: Jet injection of anesthetic through intact skin may overcome some of the limitations imposed by the cutaneous application of anesthetics without needle infiltration. A jet injector is a device containing carbon dioxide gas that rapidly forces a plunger to expel drug through a small orifice applied over intact skin. Medication penetrates the epidermis to a depth of 5 to 8 mm in 0.2 second and causes the drug to be rapidly dispersed through the skin. Safety is improved because no needles are used to penetrate the skin. Several studies have demonstrated that jet injection of anesthetics provides more rapid anesthesia than does topical application of anesthetic agents and is preferred by patients over no anesthesia at all for painful procedures such as IV insertion and arterial blood gas sampling.35–37 General adverse reactions to anesthetic agents are discussed in the “Complications” section later in this chapter. Tetracaine base is quite safe, with a low blood concentration after proper use. In one study, cutaneous erythema developed at the site of application in approximately 25% of patients.24 This vasodilatory effect may actually be an advantage when starting IV lines or performing venipuncture. EMLA is also quite safe. Although it has a high rate of local skin reactions, they are mild and transient, with disappearance 1 to 2 hours after removal of the cream. Despite the reported successful use of EMLA cream on skin ulcers, Hansson and associates25 and Powell and coworkers38 described an increase in bacterial growth, infection, and inflammation when used in experimental wounds. Methemoglobinemia resulting from the metabolites of prilocaine may occur with EMLA.23 The risk for clinically significant methemoglobinemia seems remote when EMLA is used properly. It is contraindicated in any infant younger than 3 months and in infants between 3 and 12 months of age who are currently taking methemoglobinemia-inducing drugs (nitrites, sulfonamides, antimalarials, phenobarbital, and acetaminophen). The risk for adverse effects is increased in patients with anemia, respiratory or cardiovascular disease, and deficiencies in glucose-6-phosphate dehydrogenase or methemoglobin reductase. Prolonged inhalation of ethyl chloride spray may produce general anesthesia, coma, or cardiorespiratory arrest. Ethyl chloride is also flammable, thus precluding its use with electrocautery. Topically applied anesthetics are often covered with an occlusive dressing to contain the medication. Covering the medication with an occlusive dressing may dramatically increase drug absorption and the amount of metabolites. Oni and associates demonstrated that 4% lidocaine cream applied to volunteers with an occlusive dressing resulted in three times the level of serum lidocaine and double the level of MEGX when the same dose was applied to volunteers without occlusion.39 Use caution when occluding topical anesthetics over intact skin because markedly elevated serum level of drug may be reached. In 1980, Pryor and colleagues40 reported their experience with a topical anesthetic solution (tetracaine-adrenaline-cocaine [TAC]) for wound repair. The original formula, used in most subsequent studies, consists of a solution of 0.5% tetracaine, 1 : 2,000 epinephrine (adrenaline), and 11.8% cocaine. Traditionally, anesthesia is produced by firmly applying a solution-saturated gauze pad or cotton ball directly to the laceration for 10 minutes. The resulting loss of cutaneous sensation is centered about the area of application. Gel formulations of topical wound anesthesia (TWA) with alternative mixtures of agents promise to improve the ease and safety of topical anesthetic solutions for wound repair in the ED. Advantages of TWA include painless application, no distortion of wound margins, good hemostasis, and good patient and parental acceptance in the pediatric age group. Use of TWA is generally restricted to young children with wounds less than 5 cm in length in whom the delay in application of an anesthetic is acceptable and proper application can be ensured (Fig. 29-7). TWA containing vasoconstricting agents is not generally used in structures without a collateral blood supply (e.g., digits, tip of the nose, pinna of the ear, penis), although there is no evidence that this is unsafe. When some wound preparation is desired before anesthesia, remove large debris and clotted blood to allow the appropriate application of TWA and then finish wound preparation once the wound is properly anesthetized. TWA appears to be less effective on the trunk and extremities than on the face and scalp and less effective than lidocaine infiltration in these areas. The 10- to 20-minute onset time may be unacceptable in a busy ED.41 Two other often mentioned drawbacks may be more theoretical than real. Vasoconstrictor-induced higher infection rates (see “Complications” later in this chapter) have not been clearly demonstrated. The argument that the necessary 10-minute application period is time-consuming and takes valuable nursing time is partially offset by using the child’s caretaker or adhesive paper tape alone to hold the solution in place. It is not necessary for anyone to “hold” the medicine in place when gel is used. Some EDs still stock topical anesthetic gels that contain cocaine. Cocaine use is complex because of cost and federal regulations requiring storage in a locked cabinet and maintenance of separate written records of its use. In view of the evidence that mixtures without cocaine are efficacious, avoid the potential complications related to cocaine, and do not have the potential for abuse that cocaine does, the practicality of cocaine-containing TWA has been questioned.42
Local and Topical Anesthesia
Background
Pharmacology and Physiology
Nerve Structure and Impulse Transmission
Mechanism of Action
The Physiologic and Cellular Basis for Neuronal Blockade
Activity Profile during Neuronal Blockade
Onset of Action
Potency
Duration
Topical Anesthesia
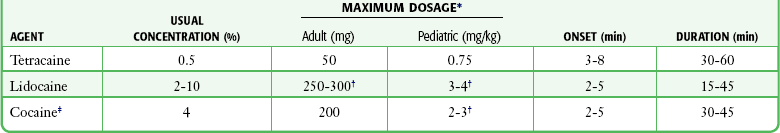
Mucous Membranes
Technique and Precautions
Intact Skin
Technique
Complications
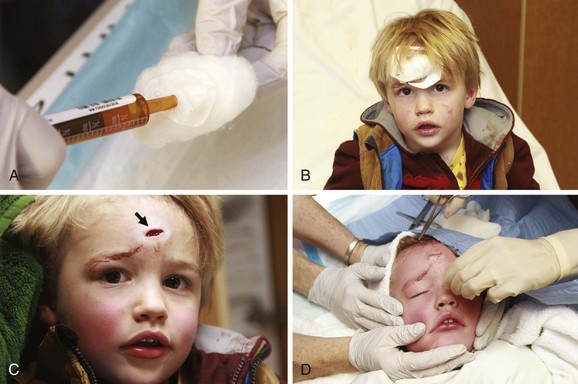
Lacerations
Indications and Contraindications
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Local and Topical Anesthesia