Chapter 11 Large Animal Spinal Cord Disease
NEUROLOGIC EXAMINATION
In this chapter, the term large animal refers to the horse, ox, sheep, goat, pig, and the Camelidae. The neurologic examination of large animals is similar to that of small animals, with adjustments that are necessary to accommodate the size of the patient. Five components of the examination include (1) gait and posture, (2) postural reactions, (3) spinal nerves, (4) cranial nerves, and (5) sensorium. See Chapter 20 for a description of the complete examination. Only specific components that relate to spinal cord disease are reviewed here.
Postural Reactions
The postural reactions as described for small animals can be performed on small pigs, calves, foals, most goats and sheep, crias, and some adult Camelidae. The hopping, hemiwalking, and hoof replacement tests are the most reliable. If your patient is cooperative, it is possible to pick up one limb and lean into the patient to force it to hop on the opposite limb. In most adult horses and cattle, this task is difficult to do and to evaluate and is obviously stressful for you to perform. It also can be dangerous in a patient that has neurologic dysfunction. We do not routinely use this test in large patients.
Circling
Recall from the previous chapters on the UMN and GP systems that we cannot distinguish between the clinical signs caused by dysfunction of these two systems (see Fig. 10-2). Because of their close anatomic relationship, these two systems are usually affected together with spinal cord lesions, and differentiating between them is unnecessary. Our objective is to recognize a deficit of the UMN and GP systems together and determine the level of the spinal cord where these systems are affected.
Spinal Nerves
Muscle Atrophy
Muscle atrophy is a clinical sign of lack of nourishment, disuse, or denervation. Denervated muscles atrophy rapidly. Neurogenic atrophy is faster and more complete than disuse atrophy, although these types may be difficult to differentiate in some patients. A good example of neurogenic atrophy is the diffuse atrophy that accompanies equine motor neuron disease (EMND). Horse owners will complain of a rapid loss of weight despite an excellent appetite in their animal. Remember that a loss of any component of the lower motor neuron (LMN) will result in denervation atrophy. These components include the neuronal cell body in the ventral gray column as in motor neuron disease, the ventral roots, and spinal nerves as in chronic polyneuritis or any part of the peripheral nerve in its course to the muscle as in radial nerve injury.
Cranial Nerves
The cranial nerve examination is important for detecting multifocal lesions and for evaluating spinal cord lesions that affect the first three thoracic spinal cord segments and cause a sympathetic paralysis, called Horner syndrome, which was described in Chapter 7. With severe lateral funicular lesions in the cervical spinal cord segments, a UMN paralysis of the entire sympathetic innervation will occur on the ipsilateral side of the body including the head. In horses, this paralysis will be apparent by the ipsilateral whole-body sweating that will occur. A focal lesion between the T1 and L4 spinal cord segments that destroys the intermediate gray column will cause a focal area of sweating where the skin is deprived of its sympathetic innervation.
Laryngeal Adduction
Some equine clinicians test one additional reflex—the laryngeal adduction reflex—using the slap test.40 This test requires the integrity of all of the cervical and the cranial thoracic spinal cord segments, the nucleus ambiguus in the medulla, and the vagal and recurrent laryngeal nerves. The test is performed by standing beside the neck of the horse with one hand grasping the larynx. With the other hand, a brisk slap is delivered to the saddle area of the thorax. In the normal animal, this action will elicit a brief closure of the glottis by adduction of the vocal folds, which you will feel with the hand that is grasping the larynx or you can observe with an endoscope. You will feel the larynx move when this adduction occurs. The anatomic pathway involved with this response starts with the stimulation of cutaneous branches of cranial thoracic spinal nerves that synapse on long interneurons in the ipsilateral dorsal gray column. Most of the axons of these long interneurons cross to the opposite-side fasciculus proprius and course cranially to the medulla, where they synapse on GSE LMN cell bodies in the nucleus ambiguus that is contralateral to the side that is slapped. The axons of these neuronal cell bodies enter the vagus nerve and course caudally to the thorax, where they leave the vagus nerve in the recurrent laryngeal nerve to course cranially to the larynx, where they innervate the intrinsic muscles that function to close the glottis. Based on this pathway, unilateral cervical spinal cord lesions that involve the lateral funiculus may cause this slap reflex to be absent when tested on the side opposite from the lesion. Bilateral cervical spinal cord lesions may cause a bilateral loss of this reflex. Be aware that many long-necked horses have a left-side laryngeal paresis from a neuropathy in the left recurrent laryngeal nerve, which can cause this slap reflex to be absent when performed on the horse’s right side. This reflex test requires considerable practice and experience to perform well, which is something that we have not done and therefore do not believe that we can rely on this reflex to help identify cervical spinal cord lesions.
SUMMARY OF CLINICAL SIGNS AT SPECIFIC AREAS OF THE SPINAL CORD
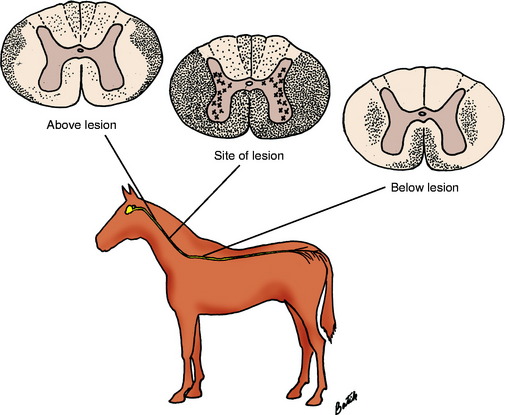
The spinal cord is divided into four regions based on the clinical signs that are exhibited when any one of these four regions are affected. See Fig. 10-1, which also applies to these large animals.
Lumbosacral: Fourth Lumbar through the Caudal Spinal Cord Segments
Thoracolumbar: Third Thoracic to the Third Lumbar Spinal Cord Segments
Craniocervical: First Cervical to the Fifth Cervical Spinal Cord Segments
Grading System for Horses
In an attempt to quantify the neurologic signs for comparative diagnostic purposes, for prognosis, and to follow the course of the disease with or without treatment, Dr. Ian Mayhew developed a grading system for cervical spinal cord disorders in horses based on the degree of neurologic deficit present in the patient.50 This system differs from the grading system used in small animals with thoracolumbar lesions, which grades the degree of function present in the patient. In small animals, the degree of spinal cord damage and resultant clinical signs are used for surgical selection, as well as for prognosis and to follow the course of the disorder.
CERVICAL VERTEBRAL MALFORMATION-MALARTICULATION
Cervical vertebral malformation-malarticulation is a very common disorder in horses that causes a compressive myelopathy. This condition was described for dogs in Chapter 10 and will be repeated here for the horse. Remember that the primary disease is the cervical vertebral column malformation and malarticulation. The compressive myelopathy is secondary because the spinal cord is in the way of and compromised by the stenosis (Fig. 11-1). Congenital and acquired forms of this disorder have been found.
Congenital Occiptoatlantoaxial Malformation
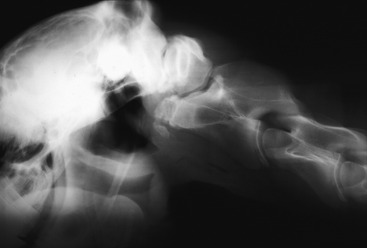
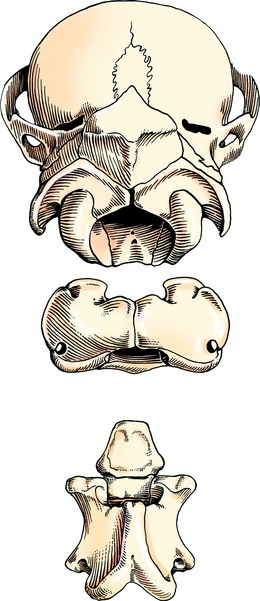
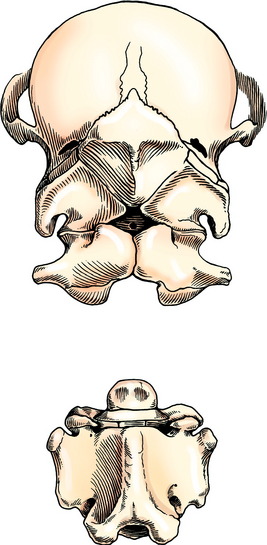
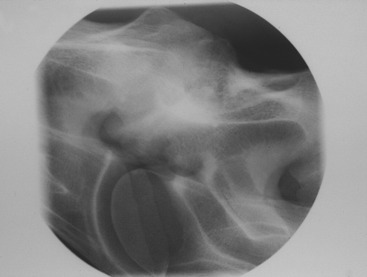
Congenital vertebral malformation is uncommon. An inherited malformation that involves the occipital and cranial cervical somites occurs in the Arabian breed and is termed occiptoatlantoaxial malformation (OAAM).57,87 This condition is presumed to be inherited as an autosomal-recessive genetic disorder. In this malformation the factors that determine the formation of the occipital bone, the atlas, and the axis have been affected such that the atlas is occipitalized and fused by fibrocartilage to the occipital bone. The caudal portion of the atlas, which should consist of articular fovea, has a more rounded appearance similar to occipital condyles. The axis has a more atlas-like appearance and has transverse processes that tend to resemble the wings of the atlas. The malformed axis usually forms a false joint in thickened connective tissue ventral to the fused atlas. This defect involves the development of the sclerotomal portions of the occipital and cranial cervical somites. Clinical signs reflect the compression that occurs to the cervical spinal cord within a reduced foramen in the atlas or occasionally, the malformation has occurred without spinal cord compression. Foals may be born recumbent and unable to stand if the spinal cord compression is severe at birth. If assisted to stand, they exhibit a severe spastic tetraparesis and ataxia and readily fall, especially if they extend their neck to nurse. Occasionally, an audible click will be heard at the site of the malformation when the head is moved. This sound most likely represents movement at the luxated atlantoaxial joint. Other foals with this malformation may not show clinical signs until a few weeks or months of age. Progression of these signs usually follows. This delay in clinical signs probably reflects the failure of the vertebral foramina of the malformed atlas or axis to remodel to accommodate the enlarging spinal cord. As a rule, the clinical signs of spastic paresis and ataxia are quite profound in all four limbs. An important diagnostic feature is the abnormal neck posture. The head and neck are more extended than normal and have a stiff appearance. Careful palpation will diagnosis this abnormality, given that no movement will be noted at the normal position of the atlantooccipital joint, and the normally palpable broad transverse processes (wings) of the atlas are reduced to small stubs of bone (Fig. 11-2). Radiographs will reveal the extent of the malformation and a myelogram is necessary to diagnose the specific location and degree of spinal cord compression (Figs. 11-3, 11-4).
We have seen a variation of this malformation in one half-Arabian foal that remained recumbent after birth in which an attempt was made to develop two atlantes and two axes.23 One atlas was reduced in size and fused to the occipital bone. The other atlas was approximately normal size and shape and articulated cranially with the occipitalized atlas. It articulated caudally with an enlarged axis that had a very long narrow cranial portion in the position of the dens and cranial part of the body. Study of the numerous growth plates indicated the formation of two fused axial bodies. The elongated cranial portion projected into the vertebral canal and compressed the spinal cord.
Acquired Vertebral Malformation-Malarticulation
Acquired vertebral malformation-malarticulation is most commonly termed the wobbler syndrome.* Realistically, this term is nonspecific and should be avoided because any cervical spinal cord lesion will make a horse wobble! So many abbreviations have been used to indicate this disorder that they just cause confusion. Therefore we will avoid using any of these abbreviations in the following discussion. In our opinion, these acquired cervical vertebral malformations and malarticulations are primarily secondary to osteochondrosis, a common developmental bone disorder in rapidly growing animals such as the horse. This disorder is multifactorial, which has all the features that are so prominent in the race horse industry: (1) selecting for the genes, which will foster rapid growth, (2) providing feed that is high in calcium and energy to encourage this rapid growth, and (3) placing these animals as soon as possible into a vigorous training program to make them winners as 2-year-old horses. Nothing could be worse for these young athletes! Two forms of this vertebral disorder are observed: (1) a vertebral foramen stenosis in the younger horse and (2) a degenerative disorder of the synovial joints that is usually in older horses, but the age is quite variable here.
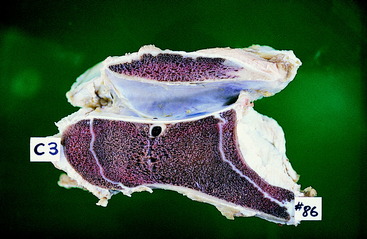
Stenosis of one or more vertebral foramina occurs in young horses from approximately 4 to 24 months of age. This abnormality is most common in male Thoroughbreds, but any breed and sex can be affected. As a rule, stenosis affects the middle cervical vertebra from C3 to C6 and is caused by failure of the bone that surrounds the vertebral foramen to resorb sufficiently to enlarge the foramen enough to accommodate the growing spinal cord (Figs. 11-5 through 11-8). Stenosis occurs at either end of the vertebral foramen or on both sides of the articulation. A prominent feature is the dorsal projection of the caudal epiphysis, which narrows the caudal opening of the vertebral foramen (Fig. 11-9). One or more vertebrae may be affected in a single horse. This lesion has been produced experimentally in Great Dane puppies fed an unrestricted diet, and it has been prevented in Thoroughbred foals by restricting their diet in its calcium and energy content, as well as volume, and restricting the foal’s exercise.28,56
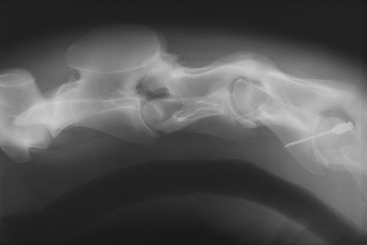
Figure 11-5 Lateral cervical radiograph of a 6-month-old Thoroughbred with a static stenosis at the C3-C4 articulation.
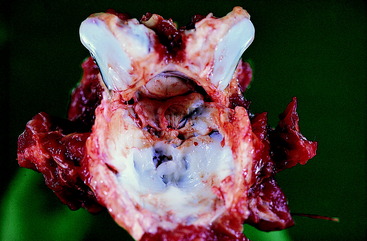
Figure 11-6 Caudal aspect of the C3 vertebra seen in the radiograph in Fig. 11-5 after disarticulation at necropsy. Note the compression of the spinal cord in the caudal orifice of the vertebral foramen.
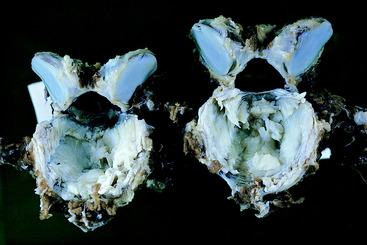
Figure 11-7 Caudal aspect of the C3 and C4 vertebrae from the horse in Fig. 11-5 after disarticulation and preservation. Note the narrow caudal orifice of the vertebral foramen of the C3 vertebra on the left compared with the normal caudal orifice of the C4 vertebral foramen on the right.

Figure 11-8 Cranial aspect of the C3 and C4 vertebrae from the horse in Fig. 11-5 after disarticulation and preservation. Note the narrow cranial orifice of the vertebral foramen of C4 on the right compared with the normal cranial orifice of the vertebral foramen of C3 on the left.
Static stenosis is indicated when myelographic evidence is found of significant spinal cord compression on both the normally extended (neutral) and the flexed radiographic views of the cervical vertebrae. Dynamic stenosis implies an instability at one or more articulations and is diagnosed when significant spinal cord compression is visible only on the radiographic view of the myelogram made when the neck is flexed. Several measurements have been described for determining whether a clinically significant stenosis is present in the cervical vertebrae. In many horses, this diagnosis can be made with lateral cervical vertebral radiographs of the standing horse that is lightly sedated with its neck in its normal resting position. Occasionally, the malarticulation is very severe and obvious without any measurements. More common are characteristics observed that suggest a stenosis, and these characteristics gain more significance in the presences of measurements that indicate a narrow vertebral canal at some point. The radiographic features that suggest a stenosis include:
The measurements of the size of the vertebral foramina are expressed as a ratio to prevent errors associated with the size of the patient and the distance between the origin of the x-rays and the patient. The minimal sagittal diameter for the vertebral foramen of a vertebra is made by measuring the height of the vertebral foramen at its most narrow point. The minimal sagittal diameter ratio is determined by dividing this measurement of the minimal sagittal diameter of the foramen by the maximal sagittal diameter of the vertebral body, which is at the cranial aspect of the vertebral body. These measurements must be made perpendicular to the ventral surface of the vertebral foramen. A study in 1994 considered a ratio below 0.52 for C4 and C5 and below 0.56 for C6 and C7 to be abnormal.61 Greater ratio values are considered normal.
This radiographic study may be sufficient to make the presumptive diagnosis of cervical vertebral compressive myelopathy (see Fig. 11-5). If this diagnosis is less certain, or if surgical therapy is a consideration, cisternal myelography is required, and the patient must be placed under general anesthesia. Myelography is necessary to distinguish between a static and a dynamic stenosis. When viewing the myelogram, be aware that, in a lateral cervical vertebral radiograph with the neck flexed, the ventral contrast column will always be thinned over the articulations. This view clearly shows the large normal range of motion that occurs between adjacent cervical vertebrae. A rule of thumb that is often used as a guide is that most horses with a significant stenosis will exhibit a 50% decrease in the height of the dorsal contrast column at the articulation when compared with the height in the middle of the vertebral foramen. A recent study, which was correlated with a necropsy diagnosis of the lesion and its location, indicated that a 70% decrease was more reliable when the neck was flexed.82 A more accurate measurement is to compare the total height of the dural tube (from the ventral surface of the ventral contrast column to the dorsal surface of the dorsal contrast column) where it is the smallest at the articulation with the same measurement of the maximal diameter of this tube in the middle of the vertebral foramen. A decrease of 20% or more at the articulation suggests a significant stenosis. In a static stenosis, evidence on a myelogram will show significant stenosis with the neck in its neutral extended position, as well as the flexed position. In a dynamic stenosis, significant stenosis is evident only on the flexed neck view. A dynamic stenosis may be prevented by surgical arthrodesis of the affected cervical vertebrae. Unfortunately at this time, magnetic resonance (MR) imaging is not routinely performed in horses because of their size.
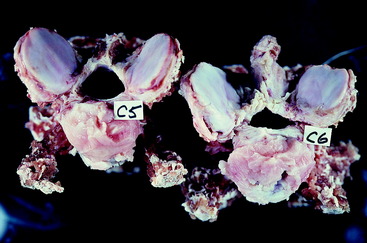
DJD of the synovial joints at the articular processes is the other form of vertebral malformation-malarticulation that can result in spinal cord compression. This osteoarthropathy is more common in older horses but has been seen in yearlings. This lesion usually occurs in the more caudocervical vertebral articulations. We believe that this degeneration is the result of chronic malarticulation at these joints that represents the effects of osteochondrosis when these patients were young or an injury followed by malarticulation. This degeneration was observed in the study of the Great Dane puppies fed an unrestricted diet. Horses that have the stenotic or degenerative lesions have an increased incidence of osteochondrosis of synovial joints in their limbs. Spinal cord compression occurs when the proliferation of the articular processes and joint capsules extends into the vertebral foramen. Occasionally, a synovial cyst develops associated with the degenerate joint capsule and contributes to the compression. Because of their dorsolateral location, these osteoarthropic lesions may show evidence of spinal cord compression on myelograms with the neck extended. If this DJD only occurs on one side, the compression and the clinical signs may be mildly asymmetric. As a rule, when a horse exhibits discomfort on neck movements, this degenerative joint lesion is usually present (Figs. 11-10 through 11-16).
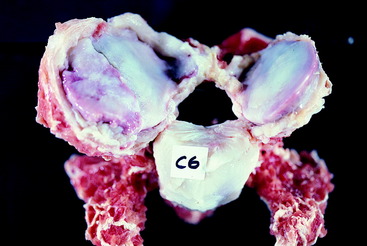
Figure 11-11 Cranial aspect of the disarticulated C6 vertebra of the horse in Fig. 11-10. Note the asymmetry of the articular processes, the disruption of the articular cartilage of the right cranial articular process and its thick joint capsule.
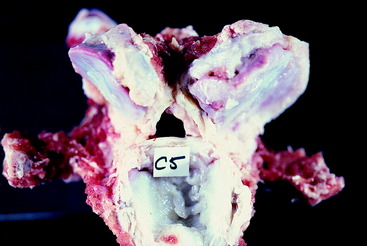
Figure 11-12 Caudal aspect of the disarticulated C5 vertebra of the horse in Fig. 11-10. This bone articulated with the bone in Fig. 11-11. Note the narrow caudal orifice of the vertebral foramen and the soft-tissue structure in this foramen on the right side, which is a synovial cyst.
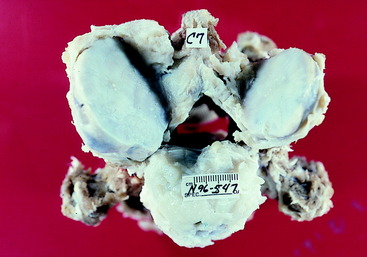
Figure 11-15 Cranial aspect of the C7 vertebra of the horse in Fig. 11-14. Note the narrowing of the right side of the cranial orifice of the vertebral foramen.
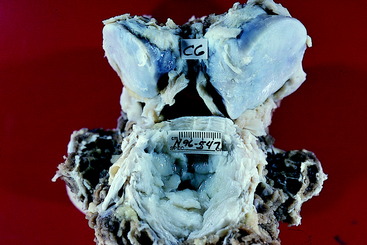
Figure 11-16 Caudal aspect of the disarticulated C6 vertebra of the horse in Fig. 11-14. Note the similar narrowing of the right side of the caudal orifice of the vertebral foramen related to the remarkably thickened right caudal articular process.
With either form of this malformation-malarticulation, the clinical signs may be sudden in onset and exhibit a variable progression, or the clinical signs may develop and progress slowly over a period of weeks to months. When they develop slowly, owners often believe their horse is lame from an orthopedic disorder. No medical therapy exists for these patients. For many years, surgical arthrodesis of the vertebral bodies has been used especially for the patients with a dynamic stenosis.83–85 Arthrodesis has also been performed on horses with DJD to stop the movement at the joints that malarticulate, which provides opportunity for resolution of some of the soft- and hard-tissue proliferation. Surgery has been most effective for horses with the stenotic form of this disorder. Most surgeons believe they can improve the clinical signs by at least one grade, and numerous mildly affected racehorses have gone back to racing. Obviously, the owner needs to be informed of the potential danger associated with riding a patient who may appear to be recovered but may well have permanently lost enough axons at the site of the compression to cause the animal to stumble when in a stressful situation. A normal neurologic examination in no way guarantees a normal spinal cord! It is one thing to repair a large-breed dog surgically with this disorder so that it can walk up and down stairs without assistance, but a human life is put in danger when a surgically repaired horse is put back into service. On the racetrack, many human lives are put at risk when one of these operated horses is loaded into the starting gait. The veterinary surgeon, as well as the owner of the patient, must accept the responsibility for the consequences of this risk.
This disorder can be prevented by slowing the rate of growth of foals by altering their diet and structuring a less-vigorous training program while these horses are young. Can you imagine a racehorse trainer recommending a restricted diet and less vigorous training program? Nonetheless, rest assured, it will work to reduce the occurrence of this disorder. A study was done on large breeding farms in Kentucky in which the foals were radiographed starting at 3 months of age.28,56 Radiographic changes were graded, and horses that were considered to be at risk for developing this cervical vertebral disorder were placed on both a restricted diet and a restricted exercise program. These management changes resulted in a significant decrease in the incidence of this bone disorder on these farms.
EQUINE DEGENERATIVE MYELOENCEPHALOPATHY
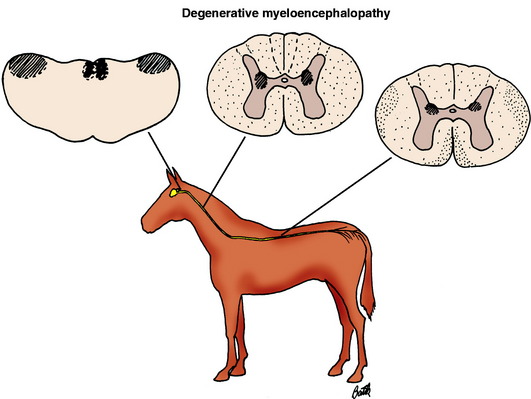
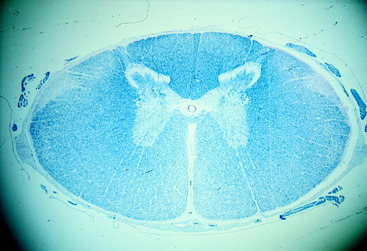
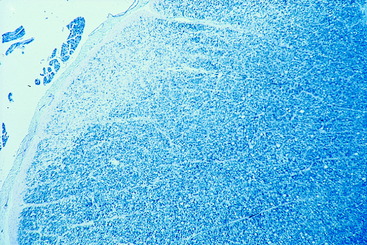
Equine degenerative myeloencephalopathy is a diagnosis that is much less common than it was 10 to 15 years ago.* The two terms used to name this disease are redundant, given that a myeloencephalopathy is a degeneration by strict definition. Nevertheless, we will keep this name to be consistent with the literature. The primary lesion is a diffuse axonopathy throughout the white matter of the spinal cord that predominates in the superficial tracts of the dorsolateral and the ventral funiculi, but all portions are affected with the least lesions in the dorsal funiculi (Fig. 11-17). This axonopathy is accompanied by a secondary demyelination and astrogliosis (Figs. 11-18 through 11-21). In addition, loss of neuronal cell bodies and spheroid development is extensive in the lateral cuneate nuclei, with variable spheroid development in the nucleus of the dorsal spinocerebellar tract (nucleus thoracicus) in the spinal cord dorsal gray column, nucleus gracilis, the medial cuneate nucleus, the olivary nuclei, reticular formation, and the vestibular nuclei in the brainstem. This brainstem nuclear lesion is the reason for including encephalopathy in the name of this disorder. In addition, an accumulation of lipopigment occurs in the endothelial cells of capillaries in the spinal cord and the pigment epithelium and outer layers of the retina, similar to older horses with motor neuron disease.22 These lesions are now thought to be the result of a deficiency in the availability of vitamin E in the diet or a disorder in its metabolism. In addition, some evidence has been found to support a possible familial basis for this disorder.48 On two large horse farms, the incidence of this disorder was significantly reduced when the pregnant mares and their foals were supplemented with vitamin E. Where serum vitamin E levels have been studied, these have only been reduced in clinically affected horses less than 1 year of age.25 Foals raised in stables and on dirt paddocks with no access to green feed are at risk for developing this disorder. Young zebras raised in captivity in a similar environment have been diagnosed with this disorder.60 As a rule, this lesion develops only in young horses. Older horses with vitamin E deficiency develop the GSE LMN degeneration of EMND. We believe that the remarkable decrease in this diagnosis parallels the decrease in the diagnosis of EMND because horse owners are more aware of the need for vitamin E supplementation, especially when a source of green feed is limited. Similar myelopathic lesions occur in nutritional deficiencies in other species of domestic animal. A good example is copper deficiency in small ruminants. Copper levels are normal in the tissues of these affected horses. The onset of clinical signs in affected horses is reported to vary from birth to 2 years. However, most affected foals are only a few months of age when these clinical signs first occur in the pelvic limbs. Those horses considered to be first affected after 1 year of age may well have had prior clinical signs that were not recognized. The clinical signs of spastic paraparesis and pelvic limb ataxia slowly progress to the thoracic limbs. If an affected horse is monitored for many months, the progression seems to cease before the affected horse becomes unable to stand. These clinical signs are identical to those caused by the malformation-malarticulation disorder. Radiographs are required to diagnose the latter. No radiographic abnormalities are noted in the cervical vertebrae of horses with degenerative myeloencephalopathy. In a prolonged case of this disorder, a fundic examination may possibly show a yellow discoloration if the lipopigment accumulation in the retina has been extensive. Vitamin E supplementation of affected horses will stop the progression of the clinical signs but will not improve them because this condition is an axonopathy, which cannot resolve, nor can these axons be effectively replaced. The disorder can be prevented by supplementation of the pregnant mare and her foal at birth.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree