Chapter 34 Laminitis
 Pathophysiology of Laminitis
Pathophysiology of Laminitis
Histopathology of Acute Laminitis
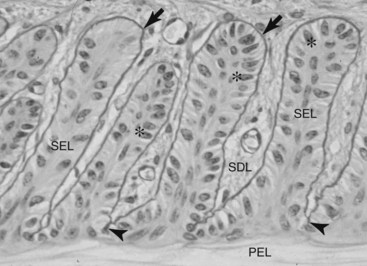
Normal lamellar anatomical characteristics, assessed before allocating a laminitis grade to a section of lamellar hoof tissue, are as follows (Figure 34-1):
Grade 1 Histopathology
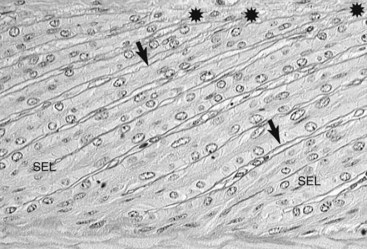
The earliest change attributable to laminitis is loss of shape and normal arrangement of the lamellar basal and parabasal cells. The basal cell nuclei become rounded instead of oval and take an abnormal position in the cytoplasm of the cell. The secondary epidermal lamellae become stretched, long, and thin, with tapering instead of club-shaped tips. These changes were present at 12 hours in serial lamellar biopsies taken after oligofructose dosing.18 First noticeable at the tips of the secondary epidermal lamellae, teat-shaped bubbles of loose basement membrane form. PAS staining shows this best (Figure 34-2).
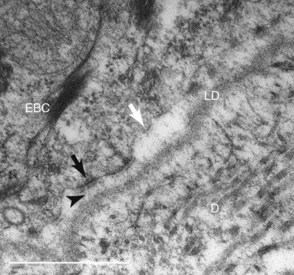
Examination of laminitis tissues with the electron microscope confirms lysis and separation of the lamellar basement membrane.19 Importantly, the greater magnification shows widespread loss of basal cell adhesion plaques (hemidesmosomes) and contraction of the basal cell cytoskeleton away from the inner cell surface. Electron microscopy shows why the basement membrane separates from the feet of the basal cells. The filaments that anchor hemidesmosomes to the lamina densa of the basement membrane no longer bridge the dermal/epidermal interface (Figure 34-3).13
Grade 2 Histopathology
Because the basement membrane is no longer completely tethered to the basal cells, it slips farther away with each cycle of weight bearing by the horse. Portions of the lamellar basement membrane are lysed initially between the bases of the secondary epidermal lamellae (see Figure 34-2). The basement membrane retracts from the tips of secondary epidermal lamellae, taking the dermal connective tissue with it. The basement membrane–free epidermal cells appear not to be undergoing necrosis, at least initially, and clump together to form amorphous, basement membrane–free masses on either side of the lamellar axis.1
Grade 3 Histopathology
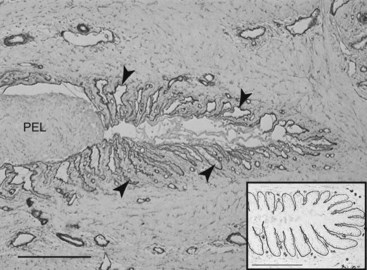
In laminitis the worst-case scenario is a rapid and near-total basement membrane separation from all the epidermal lamellae of the hoof toe, quarters, heel, and bars. Sheets of basement membrane peel away to form aggregations of loose, isolated basement membrane in the connective tissue adjoining the lamellae. The epidermal lamellar cells are left as isolated columns with little viable connection to the dermal connective tissue still attached to the DP. The hoof lamellar tips slide away from the basement membrane connective tissue attachments, at first microscopically, but as the degree of separation increases, the distance between hoof and DP becomes measurable in millimeters (Figure 34-4) and can be detected radiologically.20 This manifests clinically as a sinker. Because the basement membrane is the key structure bridging the epidermis of the hoof to the connective tissue of the DP, wholesale loss and disorganization of the lamellar basement membrane follow and inexorably lead to the pathology of hoof and bone that characterizes the chronic stage of laminitis.
 Diagnosis of Laminitis
Diagnosis of Laminitis
Radiographic examination is critical for establishing a treatment protocol and prognosis. Although rotation of the DP often can be managed successfully, sinking warrants an extremely guarded prognosis. Lateromedial images help to determine whether the condition is acute or an exacerbation of a more chronic problem. Abnormal thickness of the dorsal hoof wall, with or without modeling of the toe of the DP, implies previous disease. Lateromedial images are also important to establish the baseline position of the DP within the hoof capsule. Dorsopalmar images may be useful for assessing mediolateral balance in horses with chronic, unstable laminitis and to delect radiolucent lines indicative of lamellar separation (Figure 34-6, B). If the foot is grossly misshapen, trimming it first is preferable; otherwise, a false impression of severe rotation of the DP, which merely reflects the abnormal hoof wall growth and the development of a lamellar wedge, may occur.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree