Flaviviridae
Abstract
This chapter describes the properties of flaviviruses, pestiviruses, hepaciviruses, and pegiviruses, and features of the diseases they cause in animals.
Keywords
Japanese encephalitis virus; Flaviviridae; Flavivirus; Hepacivirus; Pestivirus
The family Flaviviridae includes four genera (Flavivirus, Pestivirus, Hepacivirus, and Pegivirus), the members of which, although similar in their genomic organization, replication strategy and physicochemical properties, are genetically distinct and biologically different (Table 29.1). The family is named after yellow fever virus (flavus being the Latin word for yellow), which is the prototype virus of the genus Flavivirus that was discovered in the course of investigating epidemic yellow fever. The genus Flavivirus contains over 90 viruses; several of which are of veterinary importance, including Japanese encephalitis, West Nile, louping ill, and Wesselsbron viruses. Some 30 members of this genus are arthropod-borne human pathogens, the causative agents of diseases varying from fevers with rash to life-threatening hemorrhagic fevers to encephalitis to hepatic necrosis. Mosquito-transmitted flaviviruses such as the four dengue viruses, yellow fever virus, West Nile virus, Japanese encephalitis virus, and Zika virus, together with several tick-borne encephalitic flaviviruses, rank among the most important arthropod-transmitted human viral pathogens. New arthropod-borne flaviviruses continue to be identified and characterized.
Table 29.1
| Virus | Host of Concern (Reservoir Host) | Arthropod Host (Mode of Transmission) | Disease in Domestic Animals (or Humans) | Geographic Distribution |
| Genus Flavivirus | ||||
| (i) Mosquito-borne flaviviruses | ||||
| Dengue viruses 1, 2, 3, and 4 | Humans (humans and monkeys) | Mosquitoes: Aedes aegypti, other Aedes spp. | (Fever and rash, arthralgia, myalgia, hemorrhagic fever) | Africa, tropical areas of Asia, Oceania, Australia, and the Americas (Central and South America) |
| Japanese encephalitis virus | Swine, humans, horses (birds) | Mosquitoes: Culex tritaeniorhynchus, other Culex spp. | Abortion, neonatal disease (encephalitis) | Asia |
| Murray Valley encephalitis virus | Humans (birds) | Mosquitoes: Culex annulirostris | (Encephalitis) | Australia, New Guinea |
| St. Louis encephalitis virus | Humans (birds) | Mosquitoes: Culex tarsalis, Cx. pipiens | (Encephalitis) | United States, Canada, Central and South America |
| Tembusu virus | Chickens and ducks | Mosquitoes: Culex spp. | Egg-drop-syndrome | Malaysia, Indonesia, Thailand and China |
| Turkey meningoencephalitis virus | Turkey | Mosquitoes | Neuroparalytic disease | Israel and South Africa |
| Usutu virus | Birds and humans (birds) | Mosquitoes: Culex spp. and Aedes spp. | Encephalitis, myocardial necrosis, hepato- and spleenomegaly | Africa and Europe |
| Wesselsbron virus | Sheep | Mosquitoes: Aedes spp. | Generalized infection, abortion | Africa |
| West Nile virus | Humans, horses, birds (birds) | Mosquitoes: Culex spp. (rarely ticks) | Fever, generalized disease, and encephalomyelitis | Africa, Middle East, North America, Central America, South America, India |
| Yellow fever virus | Humans (humans and monkeys) | Mosquitoes: Aedes aegypti, Aedes albopictus and Haemagogus spp. | (Yellow fever) | Africa, Central America and South America |
| (ii) Tick-borne encephalitis viruses | ||||
| Kyasanur Forest disease virus | Humans (monkeys, rodents) | Ticks: Haemaphysalis spp. | (Hemorrhagic fever, encephalitis) | India (Mysore) |
| Louping ill virus | Sheep, horses, humans | Ticks: Ixodes ricinus | Encephalitis | Europe |
| Omsk hemorrhagic fever virus | Humans (muskrats) | Ticks: Dermacentor spp. | (Hemorrhagic fever, gastrointestinal disease) | Central Siberia, Confederation of Independent States |
| Powassan virus | Small mammals, humans, possibly horses | Ticks: Ixodes spp. | Encephalitis | Canada, United States, Russia |
| Tick-borne encephalitis virus | Humans (rodents, birds, ruminants) | Ticks: Ixodes spp. and via ingestion of raw milk | Encephalitis | Europe, Russia, Asia |
| Genus Pestivirus | ||||
| Bovine viral diarrhea virus | Cattle, calves | (Contact, congenital) | Mostly inapparent Congenital disease: generalized persistent infection, mucosal disease | Worldwide |
| Border disease virus | Sheep | Congenital disease | Worldwide | |
| Classical swine fever virus | Swine | (Contact) | Systemic disease Congenital disease | Worldwide, but eradicated in some countries |
| Genus Hepacivirusa | ||||
| Nonprimate hepaciviruses | Horses, dogs, etc. | Uncertain | Hepatitis possibly in horses | Apparently worldwide |
| Persistent infection | ||||
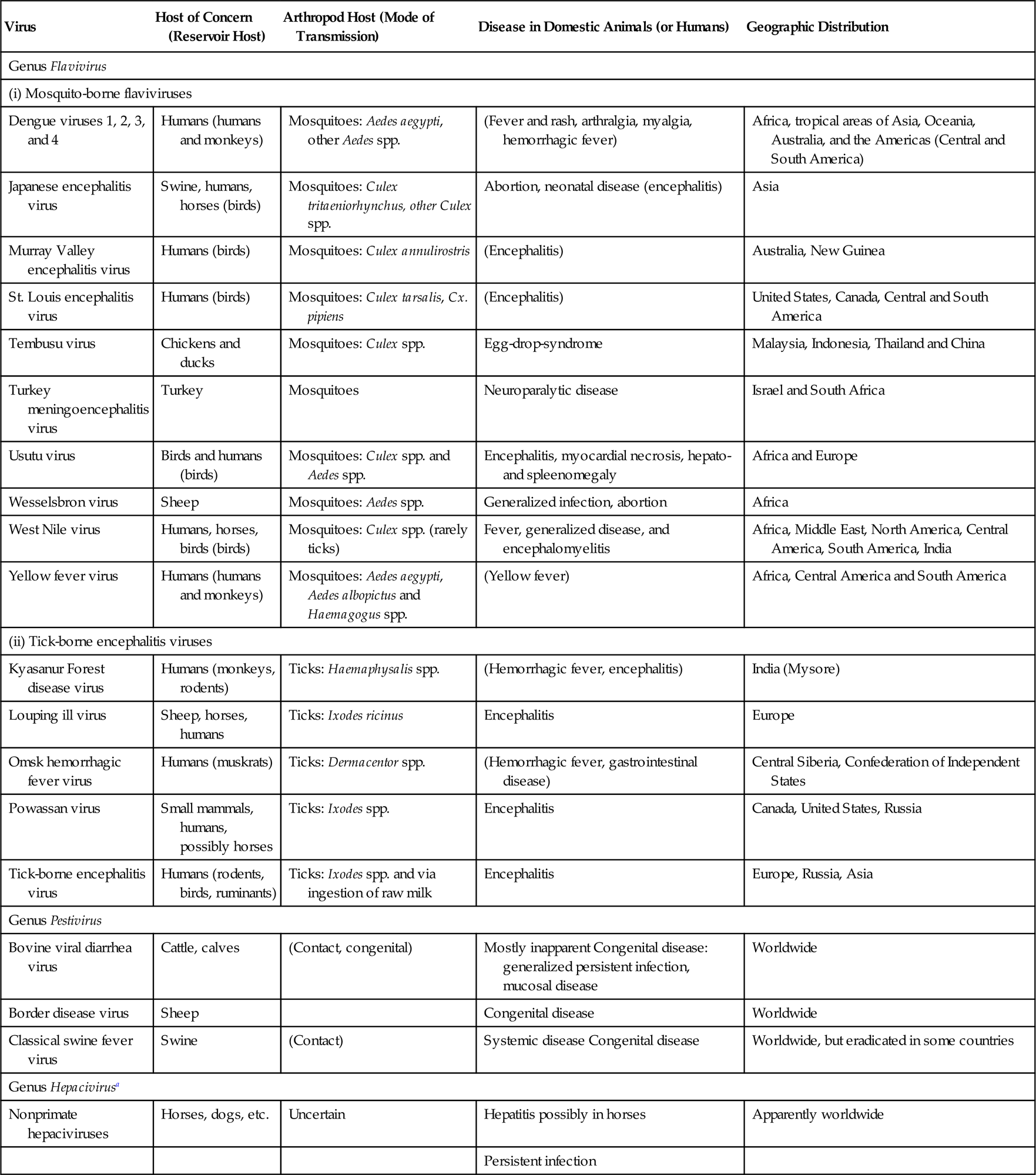
aThe genus Hepacivirus contains hepatitis C virus, an important cause of human hepatitis.
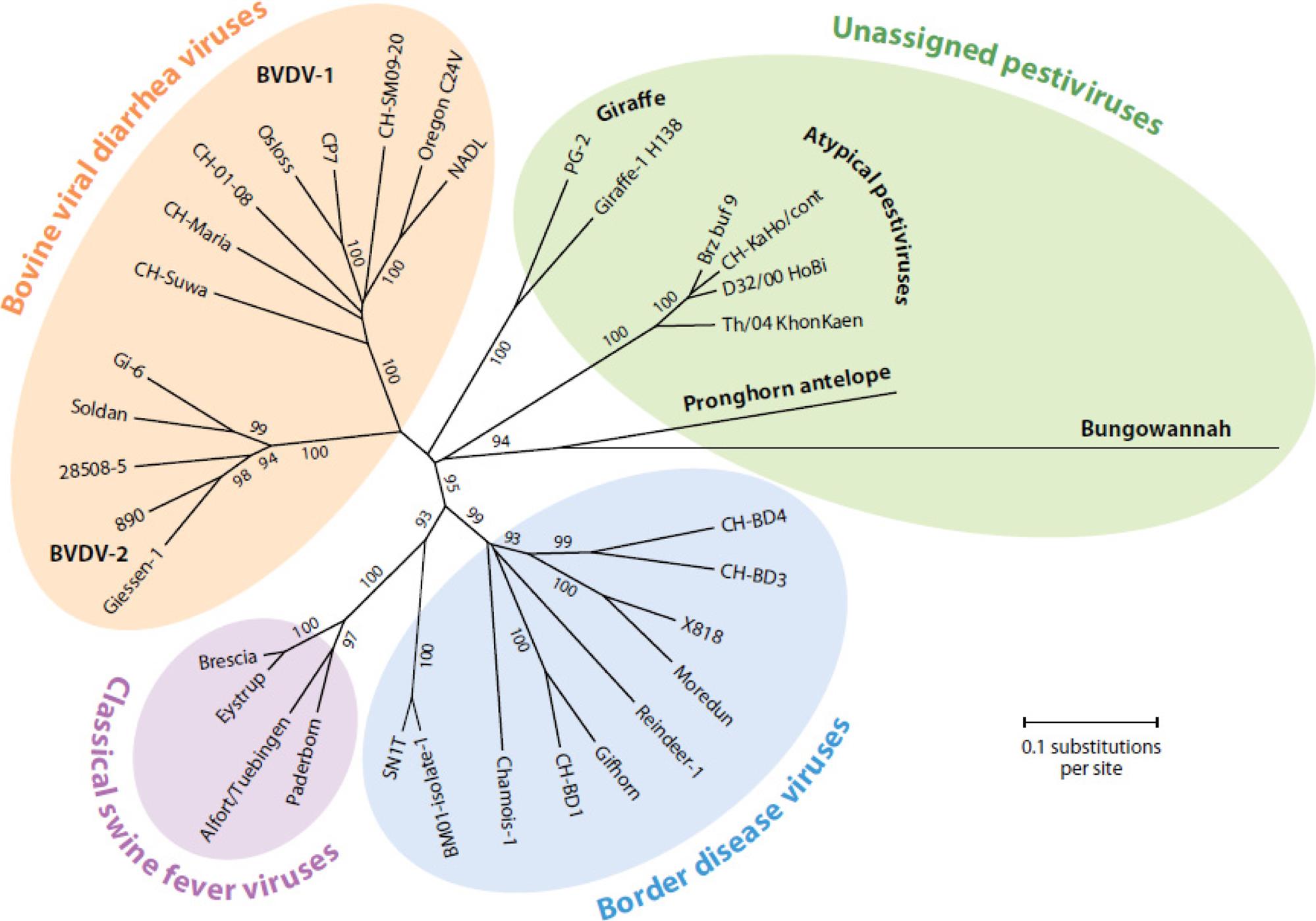
The genus Pestivirus contains important veterinary pathogens, including bovine viral diarrhea virus, border disease virus of sheep, and classical swine fever virus (Fig. 29.1). Member viruses of the genus Pestivirus occur worldwide as economically important veterinary pathogens. The host range of pestiviruses includes all even-toed ungulates (Order Artiodactyla). Classical swine fever (also known as hog cholera) was first recognized in Ohio in 1833; it has been conjectured that the virus might have emerged at that time by species jumping—that is, by a host-range mutation of another pestivirus. Early in the 20th century, as intensive swine production expanded, classical swine fever became perhaps the most important disease of swine in developed countries. Subsequent eradication programs were so successful that today reintroductions of the virus, including those from wild boar, represent the main threat to domestic herds. Bovine viral diarrhea was first described in New York State in 1946 as an apparently new disease of cattle. Mucosal disease, another clinical entity caused by the same virus, but with markedly different severity and herd incidence pattern, was described in 1953. Border disease originally was described in 1959 in sheep in the border region between Wales and England, and the infection still is common in intensive sheep production areas worldwide.
The genus Hepacivirus contains both human (hepatitis C virus) and nonprimate (eg, equine, canine) viruses. A fourth genus Pegivirus was created recently to include previously unassigned members of the family Flaviviridae, and includes a number of viruses that are widespread among mammals [humans, nonhuman primates (both New World and Old World monkeys), bats, mice, and horses]. Hepatitis C virus was discovered in 1989 by a tour de force of modern molecular biology. Although the virus still has not been successfully grown in either cell culture or laboratory animals other than the chimpanzee, it has been completely sequenced and its diagnosis made routine using reagents produced with recombinant DNA technology. This success now serves as a model for the detection, characterization, and diagnosis of other uncultivable viruses. Similarly, the availability of modern sequencing technologies has lead to the recent identification of a number of new hepaciviruses and pegiviruses in animals, although the pathogenic significance of many of these viruses remains to be determined.
Properties of MEMBER VIRUSES of the Family Flaviviridae
Classification
The family Flaviviridae includes four genera (Table 29.1), one of which is designated Flavivirus. In an effort to avoid confusion in this chapter, the term “flavivirus” will be applied only to members of the genus Flavivirus unless otherwise specified, and not to member viruses of the three other genera in the family (which will be designated individually as pestiviruses, hepaciviruses, or pegiviruses).
All members of the genus Flavivirus are closely related and share significant amino acid sequence identity, which results in serological cross-reactivity. Historically, members of this genus were assigned to it on the basis of these antigenic cross-reactions. Since most of the viruses are transmitted by arthropods (vector-borne), they were once referred to as Group B arboviruses. These vector-borne flaviviruses are divided into two major groups based on the vector they utilize for transmission: the mosquito-borne flaviviruses (including yellow fever virus, dengue virus complex, and Japanese encephalitis complex viruses) and the tick-borne encephalitis (TBE) group of flaviviruses (Table 29.1). A third group of flaviviruses are either not vector-borne or have an as yet unidentified arthropod vector [eg, Apoi virus (rodent virus), Israel turkey meningoencephalitis virus, and Rio Bravo (bat virus)], and a fourth group of flaviviruses have no known natural animal host. Most of the approximately 90 known flaviviruses are transmitted by mosquitoes, but at least 14 are transmitted by ticks, generally Ixodid species. Individual flaviviruses are adapted to either a tick or mosquito vector, and interchange of vectors does not occur. Furthermore, the mosquito-borne flaviviruses segregate phylogenetically into a lineage vectored primarily by mosquitoes belonging to the genus Culex and virus lineages vectored by mosquitoes belonging to the genus Aedes. However, in each of these lineages the restriction on the species of mosquito vector is not firm and mosquitoes belonging to other genera may also vector many of these viruses (eg, yellow fever virus in Africa is vectored by Aedes mosquitoes whereas in the Americas it is vectored by Hemagogous mosquitoes). The mosquito- and tick-borne flaviviruses are maintained in nature within arthropod–vertebrate–arthropod cycles, whereas the nonarthropod-borne viruses presumably are transmitted directly among their bat or rodent hosts.
Genomic sequence analyses confirm that the principal pestiviruses, ie, bovine virus diarrhea virus (with two different genotypes designated 1 and 2), Border disease virus, and classical swine fever virus, are closely related (Fig. 29.1). Pestiviruses also have an overlapping host spectrum, but with species-specific preferences: swine fever virus can be transmitted to cattle; bovine viral diarrhea virus can infect swine, sheep, goats, New World camelids, and a variety of other wild and domestic ungulates, including deer, antelope, yak, and buffalo; border disease virus infection rarely has been documented in cattle. Variant pestiviruses have been identified in giraffe, swine (Bungowannah virus—Australia; atypical porcine pestivirus—US), bats, and cattle (“Hobi-like” atypical pestiviruses).
The genus Hepacivirus contains human hepatitis C virus, the cause of hepatitis C, and similar viruses of horses and other nonprimate species. Other human viruses (so-called “GB viruses”) in the family that were initially included in the genus Hepacivirus, but which are not associated with severe liver disease are now reclassified in the genus Pegivirus, along with related viruses from animals (eg, horses, nonhuman primates, bats, and rodents).
Virion Properties
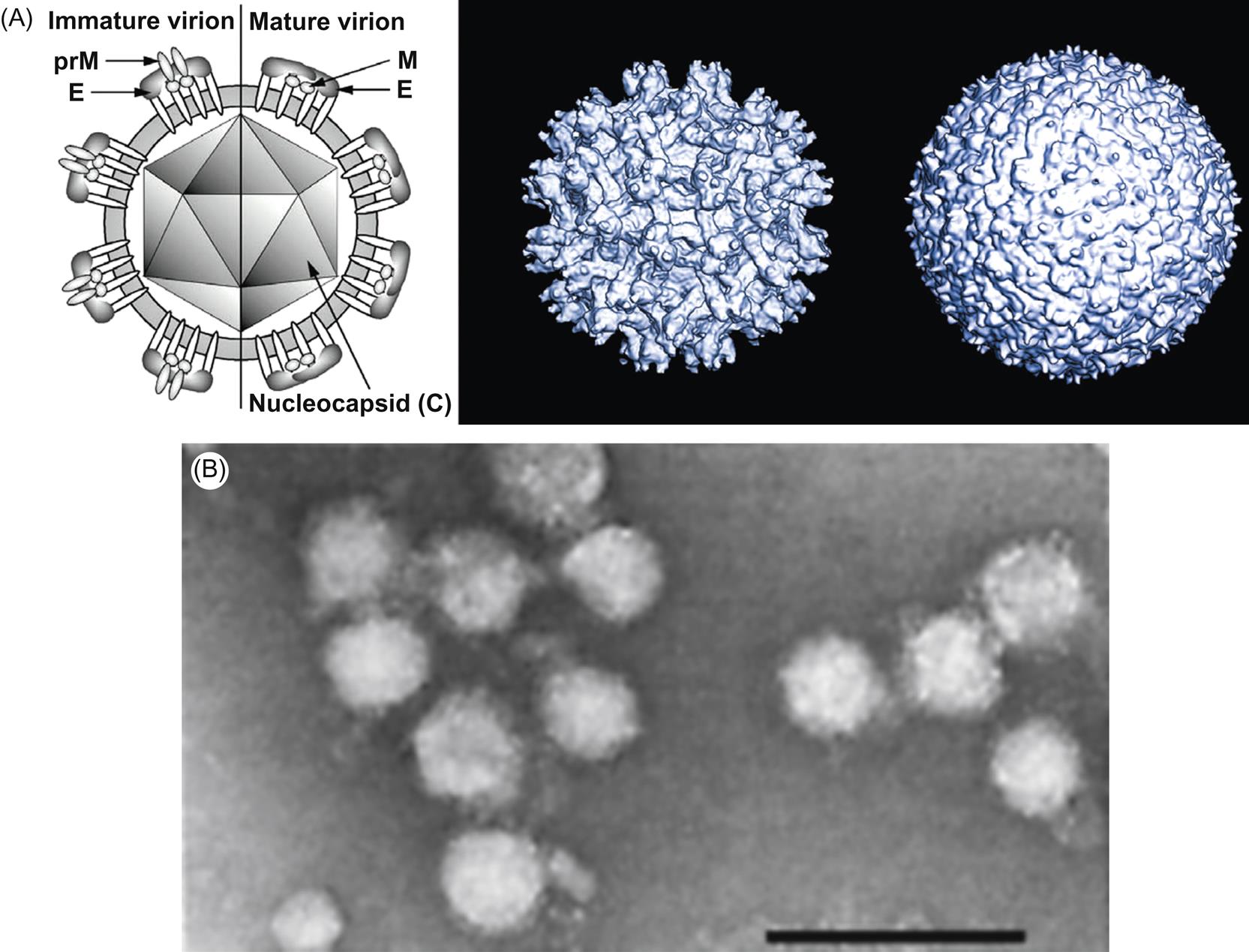
Virions of viruses in the family Flaviviridae, regardless of genus, are spherical, 50 nm (flaviviruses, hepaciviruses) or 40–60 nm (pestiviruses) in diameter, and consist of a tightly adherent lipid envelope that may display indistinct glycoprotein spikes, surrounding a spherical nucleocapsid with icosahedral symmetry (Fig. 29.2; Table 29.2). The genome consists of a single molecule of linear, positive-sense, single-stranded RNA of approximately 11, 12.3, 9.6–10.4 kb, and 11.2 kb for flavi-, pesti-, hepaci-, and pegiviruses, respectively. Flaviviruses have a 5′-terminal cap structure, whereas pestiviruses, hepaciviruses, and pegiviruses do not. Instead of the 5′ cap, the hepaci-, pesti-, and pegiviruses have an internal ribosomal entry site in the 5′ nontranslated region. Member viruses of the family Flaviviridae do not have a poly-A tail at the 3′-end of the genome.
Table 29.2
Properties of Members of the Family Flaviviridae
Genera: Flavivirus, mostly arthropod-borne viruses; Pestivirus, nonarthropod-borne, includes several veterinary pathogens; Hepacivirus, human hepatitis C virus and several related and newly identified viruses of animals of uncertain pathogenic significance; Pegivirus, including a group of newly identified human and veterinary viruses of uncertain pathogenic significance
Virions are spherical, 40–60 nm in diameter, and consist of a tightly adherent lipid envelope covered with indistinct spikes (peplomers) surrounding a spherical nucleocapsid
Genome is a single molecule of linear, positive-sense, single-stranded RNA, approximately 11 kb (flaviviruses), 12.3 kb (pestiviruses), or approximately 9.6 kb (hepaciviruses and pegiviruses) in size; 5′ end capped in flaviviruses, but not pestiviruses or hepaciviruses
Genomic RNA is infectious
Cytoplasmic replication; a single polyprotein is translated from genomic RNA; it is cleaved co- and posttranslationally to yield eight or nine nonstructural proteins and three or four structural proteins
Maturation occurs on intracytoplasmic membranes without evidence of budding
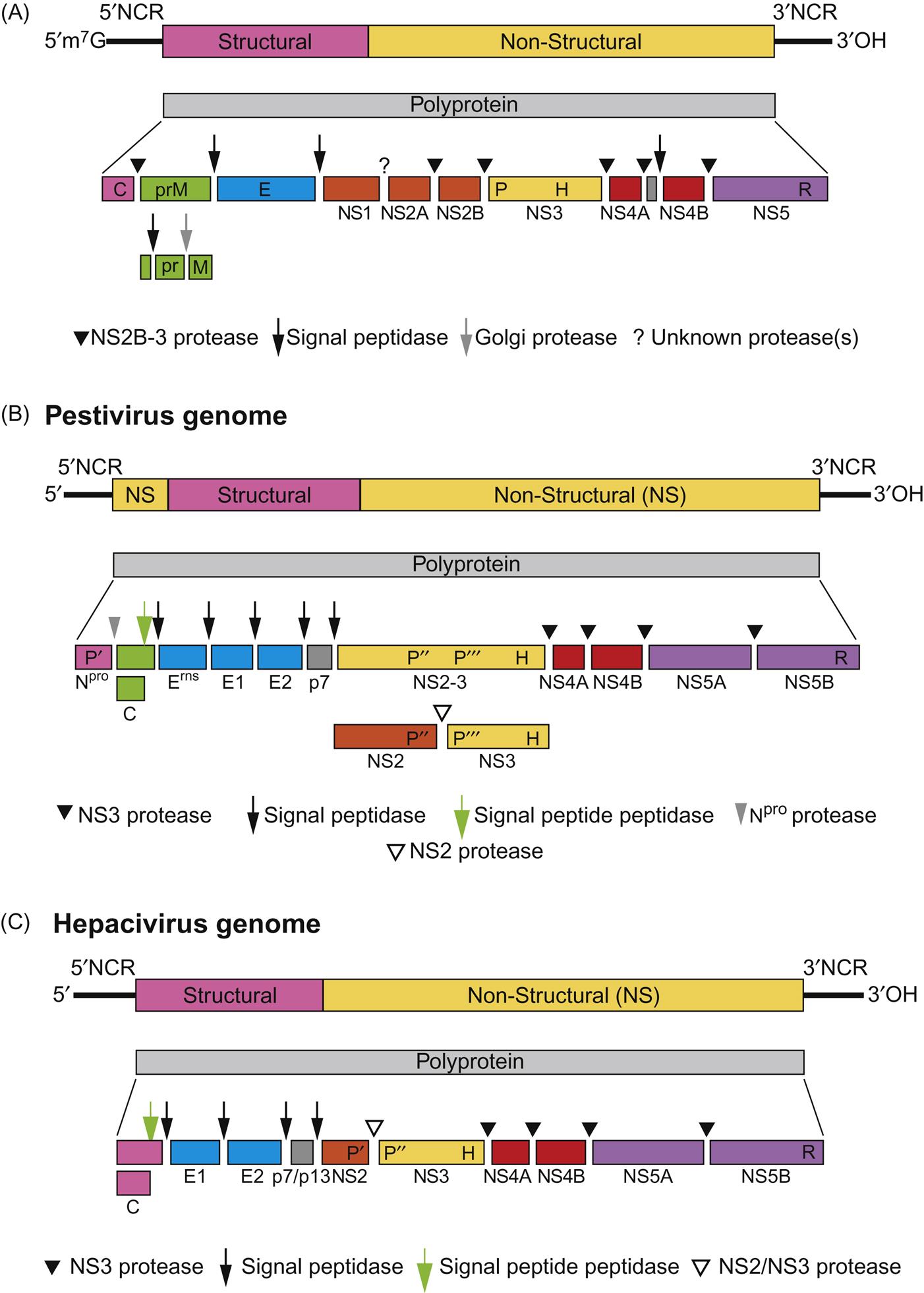
The genome of all members of the family contains one long open reading frame encoding 10 or more proteins that are created by co- and posttranslational processing and cleavage of a single, large polyprotein (Fig. 29.3). Virions contain three (genera Flavivirus, Hepacivirus, and Pegivirus) or four (genus Pestivirus) structural proteins that are encoded in the 5′ end of the genome; the nonstructural proteins are encoded in the 3′ end. Structural proteins of flaviviruses include: C, the nucleocapsid protein; prM, a precursor glycoprotein that is cleaved during virus maturation to yield M, the transmembrane protein; E, the major spike glycoprotein, which also is the major target for neutralizing antibodies. Pestiviruses have four structural proteins, including C (nucleocapsid protein) and the Erns, E1, and E2 envelope glycoproteins. There are seven or eight virus-encoded nonstructural proteins, including NS5, the RNA-dependent RNA polymerase, and NS3, which has several functions, including helicase and protease activities, in addition to contributing to the RNA polymerase complex. NS2B and NS3 are largely responsible for cleavage of the polyprotein, and host-cell proteases are responsible for the remainder of this processing. Flaviviruses encode only the NS3 protease, and the hepaci- and pestiviruses encode a second protease in the NS2 region that cleaves between NS2 and NS3. Pestiviruses also encode a unique nonstructural protein, Npro, which autocatalytically releases itself from the polyprotein; this protein is not essential for virus replication in cell culture, but modulates the interferon response in infected cells (see Chapter 4: Antiviral Immunity and Virus Vaccines).
The viruses in the family Flaviviridae are all inactivated easily by heat and by common disinfectants, and the lipid envelope is susceptible to organic solvents. However, the stability of swine fever virus in meat products and offal for weeks or even months has contributed importantly to its spread and reintroduction into previously virus-free areas.
Virus Replication
Many members of the genus Flavivirus can be grown in cell culture: Vero (African green monkey kidney), BHK-21 (baby hamster kidney), and mosquito (C6/36) and primary chick and duck embryo fibroblasts are commonly used for isolation and propagation of these viruses. Many flaviviruses infect and kill newborn and, in some cases, adult mice; indeed, most of the flaviviruses were first isolated in newborn mice. Members of the genus Pestivirus generally replicate well in primary and continuous cell cultures derived from the principal host species—for example, bovine viral diarrhea virus in bovine embryonic fibroblast or kidney cells. Border disease virus is best isolated in ovine cells, and classical swine fever virus propagates in porcine lymphoid or kidney cells. Pestiviruses isolated from naturally infected animals are predominantly noncytopathic in cell culture. To date, virtually all pegiviruses and hepaciviruses have not yet been propagated in cell cultures.
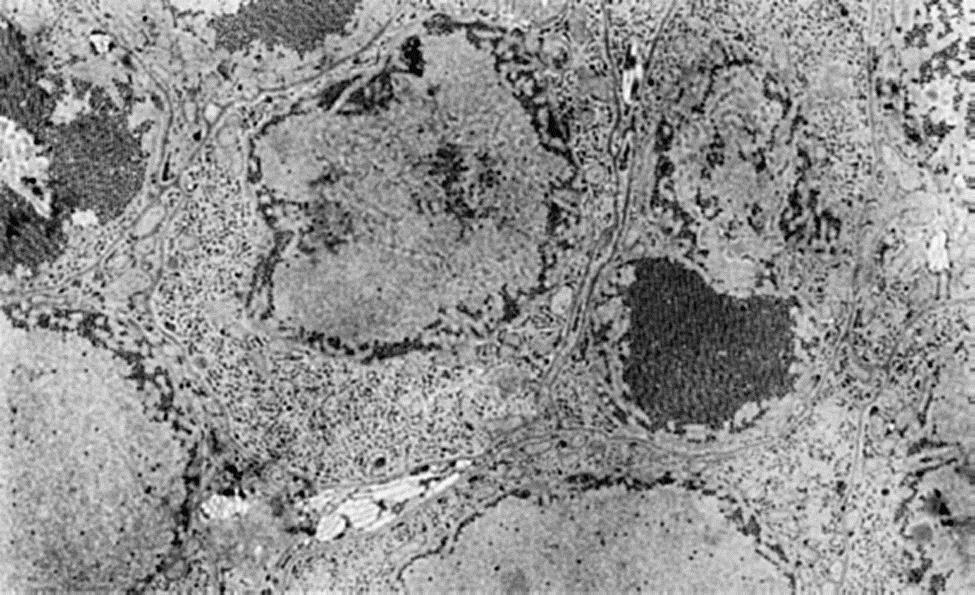
Cellular attachment of all members of the family Flaviridae, regardless of genus, appears to be mediated by ligands on the virus’ E glycoprotein(s) that interact with the cell surface molecules. Several cell surface molecules have been shown to interact with flavivirus particles, but few definitive cellular receptors have been unambiguously identified and it appears that flaviruses can utilize multiple receptors that are specific for different cell types and different host species. Virus entry into cells occurs via receptor-mediated endocytosis and replication takes place in the cytoplasm. Infection and virus replication commonly is accompanied by a characteristic proliferation of perinuclear membranes.
Virus replication involves the synthesis of complementary negative-sense RNA, which then serves as a template for positive-sense (genome-sense) RNA synthesis. The only viral mRNA is the genomic RNA—translation yields a single polyprotein that is cleaved and processed to form the various structural and nonstructural virus proteins (Fig. 29.3). For mosquito-transmitted flaviviruses, virion assembly, including acquisition of the glycoprotein-containing lipid envelope, occurs on membranes of the endoplasmic reticulum and plasma membrane in mosquito cells (Fig. 29.4), but preformed capsids and budding are not seen. Instead, fully formed virions appear within the cisternae of the endoplasmic reticulum and are transported in cytoplasmic vesicles through the secretory pathway before they are released by exocytosis or cell lysis. West Nile virus is an exception to this general rule as there is prominent budding of preassembled nucleocapsid at the plasma membrane, although some intracellular assembly of virions also occurs.
Members of the Genus Flavivirus: Mosquito-Borne Flaviviruses
All members of the genus Flavivirus are serologically related. The mosquito-transmitted flaviviruses are subdivided, on the basis of neutralization assays, into at least seven serocomplexes that collectively include approximately 40 individual flaviviruses, several of which are important pathogens of humans and animals. Viruses in the Japanese encephalitis virus serocomplex infect a variety of animal species and humans, with birds being most important to the enzootic transmission cycles and Culex mosquitoes serving as the main vectors. Member viruses of this serocomplex include: (1) Japanese encephalitis virus (Japanese encephalitis) in Asia; (2) West Nile virus (West Nile fever/encephalitis) in Africa, North America, Europe, and Asia, and Kunjin virus (Kunjin virus encephalitis) in Australia; (3) St. Louis encephalitis virus (St. Louis encephalitis) in North and South America; (4) Murray Valley encephalitis virus (Murray Valley encephalitis) in Australia and New Guinea; (5) Rocio virus (Rocio encephalitis) in South America; and (6) Usutu virus in Africa and Europe. The flaviviruses that cause human yellow fever and dengue fever form two other serocomplexes with primates as the major animal hosts and Aedes mosquitoes as the main vectors, specifically: (1) yellow fever virus (yellow fever) in Central Africa and South America; and (2) dengue virus serotypes 1–4 (dengue fever, dengue hemorrhagic fever, dengue shock syndrome) in Asia, Africa, and Central and South America.
Japanese ENCEPHALITIS Virus
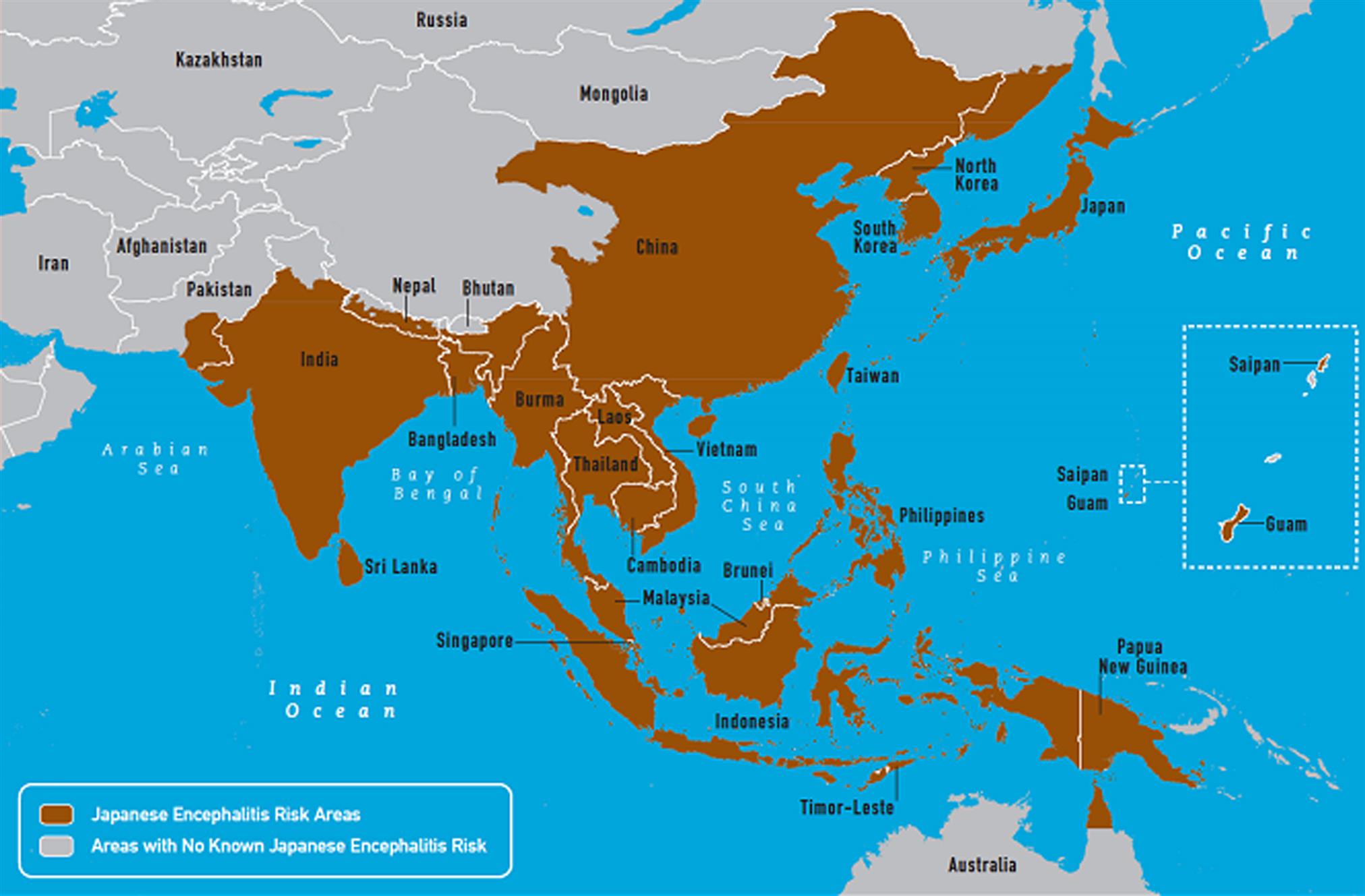
Japanese encephalitis virus is an important mosquito-borne human pathogen in much of Asia including Southeast Asia, China, Japan, the Korean peninsula and adjacent areas of the Russian Federation, Sri Lanka, and portions of the Indian subcontinent with recent incursions into the Pacific islands and northern Australia (Fig. 29.5). Although there is a single serotype of Japanese encephalitis virus, at least five distinct genotypes (I–V) are recognized that differ in their regional and temporal distribution. Human disease is devastating. Although there are many inapparent infections, the case-fatality rate amongst symptomatic cases is 10–40%, and 40–70% of survivors have permanent neurologic deficits. The primary mosquito vector, Culex tritaeniorhynchus, develops in freshwater marshes and irrigated rice fields, and blood-feeds mostly on large mammals including, swine, and occasionally humans (Fig. 29.6). Japanese encephalitis virus also can be transmitted by Culex and Aedes mosquitoes. A variety of animals, including swine, horses, dogs, bats, and reptiles, are infected naturally with Japanese encephalitis virus. Horses may develop fatal encephalitis similar to that caused by West Nile virus infection, whereas infection of adult swine is generally inapparent, with the notable exception of the occurrence of stillbirths and abortion that may occur after infection of pregnant sows. Piglets may develop neuroinvasive disease.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree