CHAPTER 40 Female Reproduction
Normal Reproduction
Seasonality
In the northern hemisphere, increasing daylight length in January and February promotes the onset of estrous activity. Peak estrous activity is usually seen in the northern hemisphere from February to April. Regular estrous activity will continue until as late as October or November, depending on the geographic distance from the equator (and therefore the length of daylight). Most cats housed indoors in North America will experience winter anestrus because of the short length of daylight. The effect of seasonality diminishes or disappears near the equator.12
Puberty
The first estrus typically occurs in queens between 5 and 9 months of age, but age at onset may be highly variable (3.5 to 18 months).37 The time of the first estrus is influenced by a number of factors: breed (shorthair breeds reach puberty earlier than longhair breeds), season (which determines the length of daylight), and the queen’s body condition. Persian and related breeds may not have their first estrus until 18 months of age or older and may not be sexually mature until 2 to 3 years of age. The average body weight at puberty is 5 to 7 lb (2.3 to 3.2 kg) or 80% of adult body weight.25 Shorthair breeds, such as the Siamese and Burmese, are more precocious and may reach puberty at a lower body weight.
The Feline Estrous Cycle
The feline estrous cycle may be divided into proestrus, estrus, interestrus, anestrus, and luteal (diestrus) phases. See Box 40-1 for feline reproduction data. Proestrus is considerably more difficult to detect in the queen than in the bitch. This part of the estrous cycle may last only one day or so, and the signs may be subtle; so, it is often not detected. In proestrus, many queens rub their head and neck against convenient objects and display affectionate behavior. Occasionally, queens in proestrus have a slight mucoid vulvar discharge and pollakiuria. During proestrus, tom cats may be attracted to the queen, but the queen will not be receptive to breeding.
BOX 40-1
Feline Reproduction Data
Length of estrus: Average 5.8 ± 3.3 days
Length of interestrus: Average 7 days, range 2-19 days
Length of pseudopregnancy: 40-50 days
Length of gestation: 66.9 ± 2.9 days (research colony); 65.1 ± 2.2 days (pedigreed)
Pregnancy rate: 73.9% (research colony)
Queening rate: 65.2% (research colony)
Kittens per litter: Average 3.7, range 1-5 (research colony); 4.6 ± 1.7 (pedigreed)
Number of litters/year: Average 2-2.5; range 1-3
Age at puberty—male: 7-18 months
Age at puberty—female: 4-18 months
Data from Feldman E, Nelson R: Feline reproduction. In Feldman E, Nelson R, editors: Canine and feline endocrinology and reproduction, ed 3, St Louis, 2004, Saunders, p 1016; Root MV, Johnston SD, Olson PN: Estrous length, pregnancy rate, gestation and parturition lengths, litter size, and juvenile mortality in the domestic cat, J Am Anim Hosp Assoc 31:429, 1995; Sparkes AH, Rogers K, Henley WE, et al: A questionnaire-based study of gestation, parturition and neonatal mortality in pedigree breeding cats in the UK, J Feline Med Surg 8:145, 2006; Verstegen J: Physiology and endocrinology of reproduction in female cats. In Simpson G, England G, Harvey M, editors: Manual of small animal reproduction and neonatology, Cheltenham, UK, 1998, British Small Animal Veterinary Association, p 11.
Estrus is defined as behavioral receptivity to mating. This stage may last from as little as 2 days to as long as 19 days, with the average duration being 5.8 ± 3.3 days.90 Mating may shorten the length of estrus, although conflicting evidence exists. A queen in estrus will crouch with the front legs pressed to the ground, the back in a position of lordosis, and the tail turned to one side to present the vulva (Figure 40-1). The queen may roll or thrash about on the floor. Queens in estrus often call or vocalize to attract the attention of males. They may be restless, have a poor appetite, and show increased affection to their caretakers. It is not uncommon for inexperienced owners to interpret estrus behavior as a sign of injury or illness.
Occasionally, queens have prolonged estrus (lasting more than 7 days). In some cases, this may be due to the maturation of overlapping waves of follicles with prolonged high estradiol levels.25 This type of prolonged estrus is most commonly seen in Siamese and related breeds. Other queens with prolonged behavioral estrus, however, have normal distinct patterns of follicular growth.25 Why these queens show prolonged estrus rather than distinct estrus periods is not understood.
Prolonged estrus can also be associated with cystic ovarian follicles. Functional cystic follicles can produce persistent increases in plasma estradiol levels (>20 pg/mL [>73.4 pmol/L]).25 Cystic ovarian structures may be identified with abdominal ultrasonography. Another infrequent variation is the split heat, most often associated with young queens. The proestrus signs occur but then subside, only to be followed a few days later by a normal proestrus and estrus. This phenomenon tends to disappear with maturity.
Anestrus is the absence of cycling activity that may occur naturally in periods of short daylight. In the northern hemisphere, this is between October and December. The effect of season on duration of anestrus diminishes with proximity to the equator.12 Individual variation is common. During this time, progesterone and estrogen are at baseline concentrations (progesterone <1 ng/mL [<3.2 nmol/L], estrogen 8 to 12 pg/mL [29.4 to 44.0 pmol/L]).53
The luteal (diestrus) phase of the queen’s estrous cycle is the period after ovulation when the dominant hormone is progesterone. Unlike the bitch, the queen does not experience a pre-ovulatory rise in progesterone. After ovulation, fertilization of oocytes occurs in the oviducts, and the embryos enter a uterine horn 4 to 5 days after ovulation.101 The embryos then space out along the uterine horns and may even migrate from one horn to another before implantation. Some embryos may be lost in this process. Implantation occurs about 12 to 13 days after breeding, and the implantation rate is estimated to be about 84%.106 The feline placenta is endotheliochorial in structure and zonary in shape. Pregnancy length varies from 62 to 74 days in queens, with the average length being 65 to 67 days.90,97
Estrus may resume about 10 days after the end of the luteal phase, but nursing queens often experience a lactational anestrus that can last for up to 8 weeks after weaning. Most queens will return to estrus about 4 weeks after weaning their kittens if it is still the breeding season. However, it is entirely possible for a queen to return to estrus while still nursing. Very often the first estrus after a pregnancy is shorter and less fertile. Estrus behavior during gestation has also been reported in the queen, although serum estradiol is not increased and no luteinizing hormone (LH) surge occurs, even if the queen allows copulation.37,107 Superfetation—kittens of different gestational ages in one litter—has never been proven to occur in the cat. The presence of poorly developed fetuses along with kittens of normal gestational age in a litter is most likely a problem of arrested development.
Hormonal Events of Estrus and Pregnancy
Although little data about follicle stimulating hormone (FSH) concentrations or activity in the queen exist, it is believed to be similar to that in other species. FSH, produced by the pituitary gland, initiates the development of ovarian follicles. Three to seven follicles develop and start producing estradiol-17β. As the follicular activity peaks, plasma estradiol levels increase and also vary widely but usually are greater than 20 pg/mL (greater than 73.4 pmol/L).32 Estradiol levels stay high for 3 or 4 days during estrus and then abruptly fall. The high estradiol levels produce two important effects: overt estrous behavior and priming of the gonadotropin surge necessary to cause ovulation. Estradiol concentrations rise again about day 58 of gestation and then decline just before parturition.55
Ovulation requires the release of luteinizing hormone from the anterior pituitary gland. During intromission, the penis probably causes distention of the posterior vagina115 and induces release of gonadotropin-releasing hormone (GnRH) from the medioventral hypothalamus resulting from neuroendocrine reflexes. Sufficient stimulus, either copulatory or noncopulatory, is required to provoke the release of GnRH. A surge of LH occurs within minutes of breeding. With multiple copulations, the LH surge is higher in amplitude and lasts longer than when only one breeding occurs, thus increasing the chances that ovulation will occur.
Several days of estradiol priming are required before LH release sufficient to cause ovulation occurs. This is typically reached by the third or fourth day of estrus. There also appears to be a stimulus threshold individual to each queen that must be exceeded in order for adequate LH release to occur. Unlike the rabbit, in which a single mating is sufficient to induce ovulation, queens vary considerably in the number of copulations required to induce sufficient LH release and ovulation. Most queens will ovulate after four or more copulations.25
Ovulation occurs 48 hours or more following the LH surge.95 All oocytes are released at the same time. The remaining granulosa cells of the ovarian follicles undergo luteinization and begin to produce progesterone almost immediately. Progesterone concentrations rise within 24 hours, and may reach highs of 60 to 90 ng/mL (190.8 to 286.2 nmol/L) by 15 to 25 days post-ovulation.113 Peak progesterone concentrations are highly variable from queen to queen. Throughout pregnancy, progesterone is maintained at high concentrations until the last few days of gestation, when the level falls to about 2 ng/mL (6.4 nmol/L) and to less than 1 ng/mL (3.2 nmol/L) immediately following parturition.113 A minimum progesterone concentration of 1 ng/mL (3.2 nmol/L) appears to be necessary to sustain pregnancy in the queen.109 Although progesterone declines at term, baseline concentrations are not required for onset of parturition in the queen, as in the bitch.55 Progesterone test kits designed for ovulation timing in the bitch have been validated for use in the queen.6
As for other induced ovulators, recent research suggests the corpus luteum (CL) may be the primary source of progesterone throughout pregnancy in the cat.113 Conflicting evidence from earlier research showed maintenance of pregnancy despite ovariectomy at 45 to 50 days of gestation and demonstrated the ability of the feline placenta to synthesize progesterone.
Two other hormones are important in feline pregnancy. Relaxin is produced primarily by the placenta in carnivores, and facilitates delivery by softening the connective tissue of the pelvis, softening the cervix and relaxing uterine musculature. Relaxin concentrations increase as early as day 20 of gestation and are the basis of a commercially available pregnancy test (see below).107 Prolactin is produced by the anterior pituitary and has various effects, including regulation of lactation. Prolactin concentrations increase from about day 35 of gestation, plateau at about day 50, and then increase abruptly just before parturition.107 Prolactin appears to be necessary for maintenance of pregnancy by supporting the CL, as the suppression of prolactin with a dopamine agonist results in abortion.
Pseudopregnancy may result if a mating is infertile. High progesterone concentrations are maintained by a centrally mediated blockage of GnRH secretion during both pregnancy and pseudopregnancy.109 This prevents the queen from returning to estrus until the luteal phase is ended. During pseudopregnancy, progesterone concentrations start to decline by about day 25 to 30 and are less than 1 to 2 ng/mL (<3.2 to 6.4 nmol/L) by day 40 to 50.113 The feline CL may be preprogrammed to atrophy after 25 to 30 days unless luteotrophic factors are present. These luteotrophic factors may originate from the fetoplacental unit and/or from the pituitary. The two most likely luteotrophic factors in the queen are relaxin and prolactin.109
Spontaneous Ovulation
A study of 44 female cats with uterine disease classified them on the basis of ovarian status (active or cystic follicles versus luteal phase ovaries).65 Of the 44 queens, 35 had no recent exposure to male cats. However, 20 of these 35 queens had luteal phase ovaries, established by histologic examination. In another study, 20 domestic shorthair queens ranging from 2.5 to 11 years old were evaluated.66 These cats were housed individually, but they could see and hear other cats, including males. Seven of the 20 queens had evidence for spontaneous ovulation, and some queens experienced it repeatedly in the study period. Spontaneous ovulation was most prevalent in older queens (although the study group had a preponderance of older queens, mean age 7.4 years), and it may be that these queens have altered hormonal function.
A study designed to approximate conditions in multicat homes and catteries group housed 15 female cats.41 The queens were all young and nulliparous. After 3 months, a male cat was housed in the same room but caged separately so that there was no physical contact with the queens. Of the 15 queens, 87% showed evidence of at least one instance of ovulation and pseudopregnancy without mating during the 4.5 months of the study. As well, 67% of the queens had evidence of spontaneous ovulation during the 3 months before the male cat entered the room.
Fertility and Breeding Management
The ancient Egyptian goddess of fertility, Bastet, was portrayed as a cat for good reason. Queens are most fertile between the ages of about 18 months to 8 years, although examples of successful production of kittens in aged queens have been reported.37 Queens more than 8 years of age tend to have more irregular estrous cycles, smaller litters, and more spontaneous abortions and kittens with congenital defects.
The average litter size ranges from 3.7 to 4.6 kittens, but there is wide variability, especially among pedigreed cat breeds.90,97,106 Given a breeding life of about 10 years and no human interference with breeding, a queen can easily bear up to 100 kittens in a lifetime.37 Queens are not monogamous and may accept several toms during an estrous cycle, allowing some litters to have multiple sires (superfecundity). Multiple paternity litters may occur more than 70% of the time in free-roaming cats in population-dense urban environments compared with less than 22% of the time in sparsely populated rural environments.91 Also, queens may use partner selection to control inbreeding. One study of eight queens in a feral colony concluded that queens avoid breeding with closely related toms but not distant relatives.48
Breeders of pedigreed cats attempt to exert control over reproduction and plan pairings based on many factors, such as the qualities (e.g., health, color, conformation) desired in the offspring. Breeders also control the timing of litters based on the health of the queen, demand for kittens, show schedules, and lifestyle factors. Breeders should be educated about maintaining proper breeding records for queens (Box 40-2) as part of a sound cattery management plan.
BOX 40-2 Reproductive Data Collection for Breeding Queens
3 Length of each estrous cycle
4 Details of behavior during proestrus and estrus
5 Details of behavior during breeding
6 Dates of breedings and number of times bred
7 Outcome of each breeding—pregnancy or date of return to estrus
8 Details of each pregnancy—length of gestation, problems with labor or delivery
9 Details of each litter—litter size, sexes, birth weights, stillbirths, congenital defects if present, illnesses, necropsy findings, and so forth
10 Documentation of problems such as vulvar discharges, pyometra, abortions, mastitis, metritis, and so forth
Queens selected for a breeding program should also meet certain health criteria. Breeding queens should be healthy and up to date with vaccinations. They should be free of common problems, such as upper respiratory tract infection, diarrhea, skin disease, and so forth. Ideally, all cats in a cattery should be tested negative for feline leukemia virus (FeLV) and feline immunodeficiency virus (FIV) and any incoming cats should be tested and confirmed free of infection before joining the cattery population. Before breeding, queens should be free of internal and external parasites. In addition, testing for inherited diseases (e.g., polycystic kidney disease, hip dysplasia, and hypertrophic cardiomyopathy) may be desirable for certain breeds and should be accomplished, where possible, before a queen or tom has reproduced. Queens of breeds with a high prevalence of blood type B should be blood typed before breeding to prevent neonatal isoerythrolysis (see Chapter 41).
The queen in estrus will signal willingness to breed by displaying interest in the tom cat, and vocalizing or purring. The queen will assume a lordosis position low to the ground with the tail to one side (see Figure 40-1). The tom cat mounts the queen, grasps the skin on the back of the queen’s neck and positions the queen for breeding. Intromission and ejaculation occur in a matter of seconds. Immediately after a successful breeding, the queen will vocalize (the “coital cry”) and leap away from and often swat at the tom cat. The tom cat should have an avenue of escape; the breeding area should be roomy or should have usable vertical space. For the next several minutes the queen will roll and thrash on the ground, stretching and licking at the perineum. Most pairs will mate several times in a day. On average, the male cat makes 2 to 6 times more attempts at breeding than the female accepts.
One efficient breeding protocol involves breeding the queen 3 times daily (at 4-hour intervals) on the second and third days of estrus. It has been shown to induce ovulation in greater than 90% of queens.101 Another successful breeding scheme allows the pair of cats to breed ad libitum for short periods during the first 3 days of estrus. However, ovulation and pregnancy rates quoted in the literature are derived from random-bred or colony-bred cats and may not always be achieved with pedigreed cats. This is especially true of the Persian and related breeds that appear to have reduced fertility. Simply housing queen and tom cat together for the duration of the queen’s estrus can also result in pregnancy, but it may deplete sperm reserves in tom cats that are frequently used for breeding.
Control of Estrus and Reproduction
Contraception for the cat must be safe, reliable, convenient, and affordable. Surgical methods (ovariohysterectomy and orchidectomy, as well as ovariectomy and, less commonly, vasectomy) are well described, although not without risk.47 However, there are various reasons why surgery may not be available, affordable, or appropriate for every cat. Even if a cat is not intended for breeding, owners may have negative attitudes toward surgical sterilization. For these reasons, safe and effective methods of nonsurgical contraception are necessary. A list of drugs for control of estrus and reproduction in the queen can be found in Table 40-1.
The simplest method of estrus control is to induce ovulation, which delays return to estrus by causing a luteal phase (pseudopregnancy) that lasts, on average, about 40 to 50 days. Mechanical stimulation of the vagina using an instrument, such as a glass rod or cotton tip swab, will induce ovulation in a queen in estrus. A teaser tom cat (a vasectomized male or a castrated male with intact libido) can also be used to induce ovulation in queens in estrus. Pharmacologic options for induction of ovulation during estrus include human chorionic gonadotropin (hCG) (250 IU/cat, IM) and gonadotropin releasing hormone (Cystorelin, Merial [Duluth, Ga.]; 25 µg/cat, IM).57 Induction of ovulation will not shorten the length of that estrus period, however. Repeated induction of pseudopregnancy may predispose queens to cystic endometrial hyperplasia-pyometra complex.
Available pharmacologic methods for longer term control of estrus include progestins, androgens and gonadotropin-releasing hormone analogues.63 Progestins are the oldest class of drugs used to control reproduction in cats. Megestrol acetate (Ovaban, Intervet/Schering-Plough Animal Health [Summit, NJ] and others) is effective for suppressing estrus in queens when given orally, starting in anestrus (2.5 to 5 mg/cat once daily for 5 days, then once weekly). Medroxyprogesterone acetate (Depo-Provera, Pfizer [New York, NY], and other brands) is a long-acting injectable progestin that is also effective at suppressing estrus when given every 6 to 12 months (25 to 100 mg/cat IM). If the queen is intended for breeding, it should be planned for the second estrus after cessation of therapy.
Proligestone (Delvosteron and Covinan, Intervet/Schering-Plough Animal Health) is a long-acting injectable progestin with weaker progestational activity than the other available drugs.10 It is licensed in Europe for temporary and permanent suppression of estrus in the queen. At the licensed dose of 100 mg/cat given subcutaneously, the effect on estrus suppression lasts about 6.5 months.63 Although it appears to be safer than other progestins, there are reports of adverse effects, such as hair loss and calcinosis circumscripta at the injection site.80
Another progestin, chlormadinone acetate has been reported as safe and effective for prevention of estrus in queens when given by SC or IM injection, orally or by SC implantation. The drug is not widely available. One study reported that long-term oral dosing for up to 4.6 years at 2 mg/cat once weekly was not associated with adverse effects other than weight gain.103 When treatment was continued for longer periods of time, mammary and uterine disorders similar to those seen with other progestins were noted.
Adverse effects of progestins are well known and include diabetes mellitus, uterine disease and infertility, adrenocortical suppression, mammary hyperplasia, and mammary neoplasia.10,61,63,67 Progestins are not approved for use in the queen in all countries (the most notable are the United States and Canada) because of the potentially serious adverse effects. Prolonged use should be avoided, and consideration should be given to alternate forms of contraception for valuable breeding queens.
Mibolerone is an androgen that has been used successfully for contraception in the bitch and queen. However, the dose necessary to suppress estrus in the queen is near the drug’s toxic dose, and so, its use cannot be recommended, nor is the drug licensed for use in the cat. Adverse effects include hepatotoxicity, skin thickening, and clitoral hypertrophy.63
Melatonin is a hormone produced by the pineal gland with secretion controlled by photoperiod. Higher concentrations are produced during times of shorter photoperiod and will suppress ovarian activity. Exogenous melatonin (30 mg/cat, PO, once daily in the evening) was found to be effective at estrus suppression after 30 days of treatment.34 The effect was reversible, with normal ovarian activity returning 21 to 40 days after melatonin was withdrawn.
Although daily oral administration of a drug may be impractical, subcutaneous melatonin implants have also been investigated in the queen. In one study, SC implants of 12 mg and 60 mg melatonin were evaluated.38 Queens were monitored for 6 months, and then ovariohysterectomies were performed. No changes in body weight or hematology and serum biochemistries were noted. Estrus was suppressed in two of four cats given 12 mg melatonin and in three of four cats given 60 mg melatonin. The mean time from implantation to estrus suppression was 20 days, and the mean duration of estrus suppression was 75 days. However, histopathology of the ovaries and uterus of all eight treated queens revealed pathologic changes consistent with cystic endometrial hyperplasia.
In another study, a single SC implant containing 18 mg melatonin effectively and reversibly suppressed estrus in nine treated queens for 2 to 4 months without adverse effects.30 Although ovariohysterectomy and histopathology were not performed on these queens, six of eight treated queens that were bred after return to estrus had normal pregnancies. The implants used in these two studies were manufactured by different companies, so that differences in formulation may account for some of the variability in study outcomes.
Gonadotropin-releasing hormone is the master reproductive hormone, controlling release of LH and FSH from the pituitary gland. Sustained exposure to GnRH analogues causes downregulation of GnRH receptors and decreased release of LH and FSH, thereby suppressing fertility. GnRH analogues are under investigation primarily for control of reproduction in male and female dogs, while few studies have been published for cats. In one placebo-controlled study, a 6-mg deslorelin (Suprelorin; Peptech Animal Health, North Ryde, Australia) implant was administered subcutaneously to 10 queens.76 A placebo implant was used for 10 control queens. All cats were followed for 14 months, with daily observation for estrus and frequent monitoring of fecal estradiol levels. The deslorelin implant was effective in inducing reversible suppression of estrus, but the duration of suppression varied widely from cat to cat (range, 5 to 14 months or longer). No adverse effects on the health of treated cats were noted.
Nonsurgical pregnancy termination is not often requested for the queen, but several pharmacologic options are described. The traditional mismating treatment has been estrogen (diethylstilbestrol 2 mg/cat, IM or estradiol cypionate 0.25 mg/cat, IM) given 2 to 3 days after breeding. The mechanism of action is thought to be interference with tubal transport of fertilized eggs. However, estrogens will prolong estrus in the queen and are associated with potentially serious adverse effects, such as pyometra and aplastic anemia, and so cannot be recommended. A single oral dose of megestrol acetate (2 mg/cat) is reported to prevent implantation of fertilized eggs.25
There are several protocols for induction of midpregnancy abortion in the queen. Prostaglandin F2alpha (Lutalyse, Pfizer Animal Health) may be given daily for 5 days after day 33 of pregnancy (2 mg/cat, IM).57 After day 40 of pregnancy, the drug may be given twice, 24 hours apart (0.5 or 1.0 mg/kg, SC).57 Abortion will occur within 1 week. At higher doses, side effects of prostaglandin are well known and include nausea, vomiting, diarrhea, and restlessness. The effects are seen within 10 to 15 minutes of administration and last 1 to 3 hours. Administration of prostaglandin appears to be luteolytic during pregnancy (but not pseudopregnancy), causing a rapid decline in plasma progesterone.
The prolactin inhibitor cabergoline (Galastop; Ceva Animal Health, St. Louis, Mo.) has been studied in Europe for induction of abortion in the queen. Cabergoline (1.65 µg/kg, divided twice daily, SC, for 5 days) given on day 30 of gestation induced abortion in four of five queens 7 to 10 days later.112 The mechanism of action is luteolysis by inhibition of prolactin release, a luteotrophic hormone in the queen. In a feral cat colony, oral cabergoline (5 to 15 µgkg/day, to effect [typically 5 days]) was successful in inducing abortion when started after day 36 of pregnancy.49 Cabergoline may also be used with a prostaglandin F2alpha analogue. In one study, cabergoline (5 µg/kg/day, PO, to effect) was combined with cloprostenol (Estrumate, Intervet/Schering-Plough Animal Health; 5 µg/kg, SC, every 2 days, to effect) in five queens after 30 days of pregnancy. All queens aborted in 8 to 10 days with no adverse effects, and no compromise of subsequent fertility.82 In another study, cabergoline (15 µg/kg/day, PO, to effect) combined with alfaprostol (10 µg/kg, SC, every 2 days, to effect) was effective in inducing abortion when started on days 25 to 42 of pregnancy.23 The average duration of treatment was 5.6 days (range, 3 to 8 days). Vomiting was reported in 5.5% of treated queens. If treatment is given late in pregnancy, premature birth is induced with early death of kittens.
Aglepristone (Alizin; Virbac, Carros, France) is licensed for pregnancy termination in the bitch in many countries and is also used in the queen. It has the advantage of infrequent administration when compared with other pharmacologic methods. The mechanism of action is by blocking progesterone receptors, which leads to placental detachment.29 One large study evaluated the efficacy of aglepristone (15 mg/kg, SC, repeated 24 hours later) after 33 days of pregnancy in 61 queens.27 Termination of pregnancy was achieved in 88.5% of the queens, with 50% of pregnancies terminated within 3 days. Mild depression and anorexia were seen in less than 10% of queens. Termination of pregnancy occurred despite high plasma progesterone concentrations. Another study achieved 87% success in 23 queens when aglepristone (10 mg/kg, SC) was administered on days 25 and 26 of pregnancy.28 Pregnancy termination occurred within 5 to 9 days.
The majority of the methods described above for control of estrus and reproduction are not suitable for large scale use. Overpopulation of cats in North America and other countries around the world leads to euthanasia and suffering. There is an urgent need for development of safe, effective, inexpensive contraceptives that would be easy to administer to large groups of cats, especially free-roaming or feral cats. One such approach may be immunocontraception—the use of the immune system to block fertility. This concept is under investigation for various reproductive tissues and hormones and may result in a vaccine-type product in the future.88
Clinical Problems
Ovarian Remnant Syndrome
Ovarian remnant syndrome (ORS) is the presence of functional ovarian tissue with signs of estrus after ovariohysterectomy (OHE) or ovariectomy. Neoplasia in ovarian remnants, such as granulosa cell tumor, is a rare cause of ORS. Signs of estrus may occur weeks to many years after surgery74 and include lordosis, vocalizing, rolling on the ground, and receptivity to intact males. Age at time of surgery and breed of cat do not appear to influence risk of ORS, although one report did not find any cases in queens spayed before 4 months of age.74 The most common causes of ORS are failure to remove all or part of an ovary at surgery, accessory ovarian tissue74 and revascularization of ovarian tissue inadvertently dropped into the abdomen during OHE.19
Diagnosis is most commonly made by observing signs of estrus in a spayed cat and concurrent vaginal cytology consistent with estrus (cornified epithelial cells, absence of red or white blood cells, clear background). Documentation of serum estradiol levels greater than 20 pg/mL (greater than 73.4 pmol/L) while signs of estrus are occurring is also consistent with ORS, although the diagnosis cannot be ruled out if estradiol levels are low. Caution must be exercised with interpretation of baseline estradiol and progesterone concentrations; however, as there is considerable fluctuation with time. The diagnosis may also be established by inducing ovulation of mature ovarian follicles during estrus with GnRH (Cystorelin; Merial; 25 µg, IM) and documentation of elevated serum progesterone (>2 ng/mL [6.4 nmol/L]) 1 to 3 weeks later.51
A protocol has also been described to detect ovarian activity when a queen is not in estrus by administering the GnRH analogue buserelin (Receptal; Intervet/Schering-Plough Animal Health; 0.4 µg/kg, IM) with measurement of serum estradiol 2 hours later.5 Serum estradiol concentrations greater than 3 pg/mL (>11 pmol/L) were consistent with presence of ovarian tissue. No adverse effects from administration of buserelin were noted.
Mammary Hyperplasia
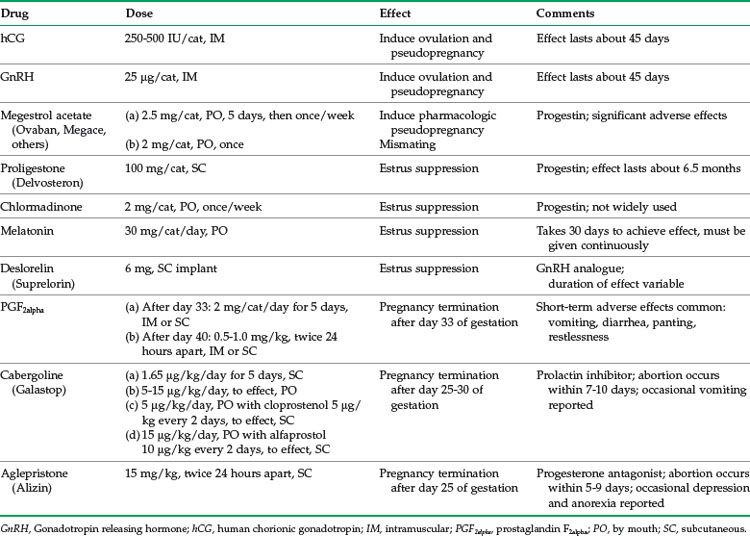
Approximately 80% of feline mammary masses are neoplastic, most commonly adenocarcinomas. The remaining 20% are benign and are predominately mammary hyperplasia (also called fibroepithelial hyperplasia and mammary fibroadenomatous hyperplasia). Mammary hyperplasia (MH) is most commonly seen in young cycling queens but may also be seen in pregnant queens and in male or female cats treated with progestins (e.g., megestrol acetate, medroxyprogesterone acetate).67,69 Typically, most or all of the glands are affected. The hyperplasia can be severe, leading to tissue necrosis, ulceration, and infection. It is often mistaken for neoplasia on gross appearance (Figure 40-2). Histologically, the lesions consist of benign, unencapsulated, fibroglandular proliferation. Progesterone receptors have been commonly found in MH samples, while estrogen receptors have been found in only 50% of cases.71 The etiology is suspected to be an exaggerated response to natural progesterone or synthetic progestins, but the disease is also rarely reported in sterilized male or female cats with no history of progestin therapy. In spayed queens, ovarian remnant syndrome should be ruled out.
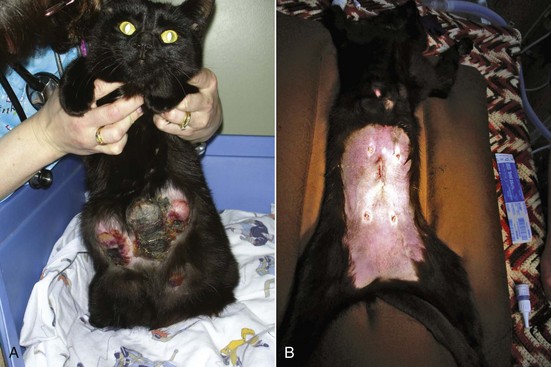
The diagnosis is made by clinical signs, patient signalment and history. Biopsy of affected tissue and histopathology will confirm the diagnosis of MH. However, surgical biopsy of markedly swollen mammary glands may create incisions that are difficult to heal due to wound tension. Treatment varies with the underlying cause. Intact queens should be spayed, and a flank approach is most appropriate (Figure 40-3). If the cat is being treated with progestins, treatment should be stopped. The drug of choice for treatment of MH is the progesterone receptor blocker aglepristone (Alizine, Virbac; 10 to 15 mg/kg/day, SC, days 1, 2, and 7).33,78 One long-term study monitored 14 queens with MH for 12 months following treatment with aglepristone.58 Remission of clinical signs occurred in an average of 4 weeks. Cats that had been treated with long-acting medroxyprogesterone acetate required treatment for 5 weeks. Six of the queens were subsequently bred, and four delivered normal litters. Aglepristone may not be available or licensed for cats in every country. For cats that have not received exogenous progestins, other choices include dopamine agonists that reduce prolactin levels, such as cabergoline (5 µg/kg/day, PO, 5 to 7 days)70a or bromocriptine (0.25 mg/cat/day, PO, 5 to 7 days).25,70a In most countries, these drugs are not licensed in the cat and must be obtained from a compounding pharmacist. Infections should be treated with broad-spectrum antibiotics. Occasionally, MH will resolve spontaneously, but it typically takes several weeks to several months to resolve even with treatment.
Determining Reproductive Status
A rapid, semiquantitative colorimetric assay is available that shows a positive result when serum LH level is greater than 1 ng/mL (Witness-LH; Synbiotics Corporation, Kansas City, Mo.). The test was developed for canine ovulation timing and has been validated in the queen. Test sensitivity for determination of reproductive status was determined to be 100% and specificity to be 92%.92 A single negative test is highly likely to indicate a sexually intact queen. A single positive test suggests a spayed queen, although false positives may occur if an episodic LH surge is sampled or the queen is in estrus. The manufacturer recommends that positive tests be confirmed with a second sample taken 2 hours later. Anecdotally, equivocal test results have been reported in some spayed cats.
Congenital Anomalies
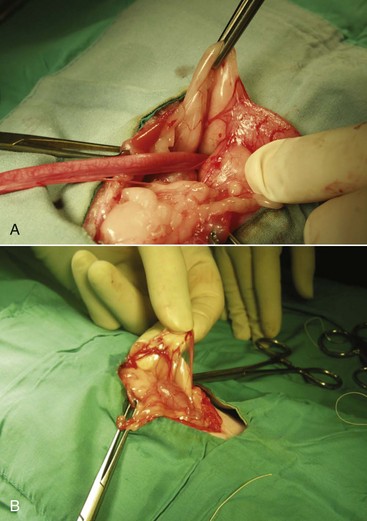
Congenital anomalies of the queen’s reproductive tract are not common and are poorly described in the literature. Segmental aplasia/hypoplasia/agenesis of the uterine horn, often called uterus unicornis, may be encountered occasionally and can present difficulties for veterinarians when found incidentally during ovariohysterectomy. Anecdotally, this condition appears to be more common in Ragdoll cats than other breeds or nonpedigreed cats. The abnormality may also be discovered during investigation of infertility in breeding queens.73 One uterine horn may be missing or reduced to a thread-like remnant, and the ipsilateral kidney is often absent.7,31,52,70 However, both ovaries are typically present and the surgeon must ensure the ipsilateral ovary is found and removed during ovariohysterectomy (Figure 40-4). Failure to remove the ipsilateral ovary is likely to result in ovarian remnant syndrome and necessitate an exploratory laparotomy at a later date.When one normal uterine horn and ovary are present, the queen may have normal estrous cycles and may even become pregnant. However, segmental aplasia may cause failure to conceive associated with fluid accumulation in the uterine lumen, depending on the location of the occlusion.70,73
Normal Gestation and Parturition
Pregnancy Diagnosis
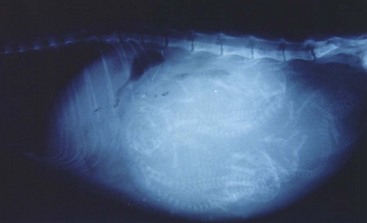
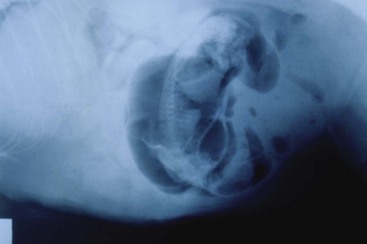
Radiography may be used to detect pregnancy once fetal bones begin to mineralize, typically by day 36 to day 45 of gestation.72 Until this time, only uterine enlargement may be detected, which could be consistent with either pregnancy or uterine disease, such as pyometra. Radiography is useful for determining the number of fetuses by counting the number of skulls present (Figure 40-5). Although fetal death is detected earlier by ultrasonography, radiographic changes include a hyperextended or hyperflexed position, collapse of the skull bones, and intrafetal or intrauterine gas (Figure 40-6).
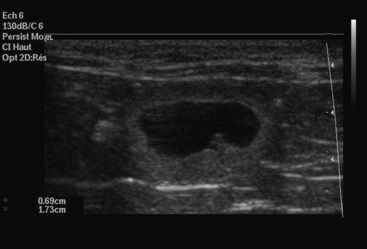
Ultrasonography is a more sensitive test for pregnancy than radiography. The gestational sac, a spherical anechoic structure slightly compressed at the pole, can be detected at 11 to 14 days and the embryo at 15 to 17 days postbreeding.17 From day 30, it is possible to identify fetal organs. Details on the time of ultrasound appearance of various fetal and extrafetal structures in the cat have been published.118,120
A benefit of ultrasonography is the ability to determine fetal viability by detecting a beating heart (as early as 16 days) and fetal movement (as early as 32 days). Fetal heart rate in the cat averages about 228 beats per minute (range 193 to 263 beats per minute).114 Unlike the dog, fetal heart rate remains stable during gestation in the cat. Sex determination is even possible, at about days 38 to 43 postbreeding.119 Early fetal death is also identifiable, because examinations performed on consecutive days will show that the gestational sacs decrease in size (Figure 40-7). However, ultrasonography may not be as good as radiography for determining the number of fetuses present. Ultrasonography views each fetus individually, and movement of the queen or the uterus makes identification of individual fetuses confusing.
In a study designed to evaluate the commercially available relaxin test kit, 11 queens were mated and monitored for pregnancy.93 All queens were confirmed pregnant and delivered kittens. In addition, 13 pregnant queens undergoing ovariohysterectomy were also tested. A group of 23 nonpregnant cats were tested as controls. The kit was able to detect pregnancy between days 20 and 25 of gestation. All pregnant queens tested negative within 5 days postpartum. In the control group, two cats tested false positive and both of these queens had large ovarian cysts. This suggests another possible source of relaxin production in some queens. The test was estimated to have 100% sensitivity and 91% specificity in cats after day 25 of gestation, with a positive predictive value of 93%.
Prediction of Due Date
The mean length of pregnancy in the queen is 65 to 67 days,90,97 but it can be highly variable. It is influenced by breed (the longest gestations are in the Siamese and Oriental breeds) and litter size (larger litters are associated with shorter gestations).97 Normal pregnancies lasting less than 54 days or more than 74 days are rare and are often associated with high neonatal mortality. During a breeding life, most queens will establish a fairly predictable pattern for length of gestation. If the breeding date is unknown, it is helpful to have an alternate method of estimating the queen’s due date, especially if the queen may require assistance during labor and delivery. Due dates can be calculated using measurements obtained from radiography and ultrasonography.
There is a predictable sequence of bone mineralization in the feline, similar to that in the canine, but beginning about 1 week earlier in gestation. Prediction of the date of parturition within 3 days was possible for 75% of 32 cats (and within 7 days in all cats) using a schedule for bone mineralization developed in one study (Table 40-2).44 Not all structures were reliable for prediction of parturition, however. Mineralization of the humerus and femur occur over the narrowest range, while the ulna, fibula, and pelvic bones have more variable mineralization times. The fibula, calcaneus, and phalanges may not become visibly mineralized before parturition.
TABLE 40-2 Number of Days Prior to Parturition for First Radiographic Detection of Fetal Skeletal Mineralization of Various Bones and Teeth in 17 Pregnant Cats
| First Day of Visible Mineralization | ||
|---|---|---|
| Structure | Mean ± SD | Range |
| General mineralization | 26 ± 1 | 25-29 |
| Vertebral column | 24 ± 1 | 22-27 |
| Skull | 22 ± 1 | 21-27 |
| Ribs | 22 ± 2 | 20-25 |
| Scapula | 20 ± 2 | 17-24 |
| Humerus | 20 ± 1 | 20-24 |
| Femur | 21 ± 1 | 19-23 |
| Radius | 19 ± 2 | 15-22 |
| Tibia | 19 ± 1 | 15-21 |
| Ulna | 17 ± 2 | 5-21 |
| Pelvis | 19 ± 1 | 8-20 |
| Fibula | 13 ± 3 | 0-17 |
| Tail | 15 ± 2 | 8-16 |
| Metacarpals and metatarsals | 8 ± 3 | 3-14 |
| Phalanges | 6 ± 3 | 0-11 |
| Calcaneus | 6 ± 3 | 0-10 |
| Teeth | 2 ± 1 | 1-6 |
SD, Standard deviation.
Reprinted with permission from the Journal of the American Veterinary Medical Association: Haney D, Levy J, Newell S et al: Use of fetal skeletal mineralization for prediction of parturition date in cats, J Am Vet Med Assoc 223:1614, 2003.
Prediction of gestational age and date of parturition are also readily accomplished using fetal ultrasound measurements of head or body diameter (Box 40-3 and Figure 40-8). Using ultrasound measurements, the due date can be estimated ±2 days about 75% of the time. Ideally, the measurements should be taken between 23 and 28 days postbreeding.
BOX 40-3
Prediction of Gestational Age in the Cat (Within 1 to 2 Days) from Ultrasonographic Measurements
Gestational age (GA) in days = 25 × HD + 3 or 11 × BD + 21
Days before parturition = 61 − GA
Modified from Beck K, Baldwin C, Bosu W: Ultrasound prediction of parturition in queens, Vet Radiol Ultrasound 31:32, 1990.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree