CHAPTER 3 Eyes and Ocular Adnexa
Cytologic evaluation of cellular material collected from the equine eye and ocular adnexa is a valuable tool in the diagnosis and treatment of many ophthalmic diseases affecting the horse. Cytologic collection and examination are simple to perform, are inexpensive, and hold minimal risk for the patient. Also, cytologic results can be obtained more expeditiously than by histopathology. Although an important procedure used by both the general practitioner and the specialist in ophthalmology, cytology should not be considered a replacement for other diagnostic tools, including culture and biopsy. Microbial culture and histopathology have been shown to maximize the positive identification of infectious organisms and improve the interpretation of cytology alone.1,2 Cytology evaluates individual cells without regard to the architectural structure of the tissue, so histologic examination is more appropriate for the classification and prognosis of neoplastic disease.3
Collection of cytologic specimens from the eye and ocular adnexa generally adheres to the same principles as for collection of any cytologic specimen and has been reviewed elsewhere in this text (see Chapter 1). This chapter will first highlight some special considerations in the preparation of cytologic specimens from ocular tissues and then review normal ocular cytologic findings. Following these considerations, a more detailed description of the more common infectious, inflammatory, and neoplastic diseases affecting the equine globe and associated structures is given.
Cytologic Collection of Ocular Tissues
Proper sample collection is essential for accurate diagnosis. Examination or manipulation of the equine eye may be difficult due to severe ocular pain associated with the disease process. Adequate restraint—in the form of an ear or nose twitch, auriculopalpebral motor and supraorbital sensory nerve blocks, topical anesthetics, and intravenous sedation—is often required to allow proper examination and initial diagnostic testing of the affected globe.4 Diagnostic procedures performed on the eye and associated adnexa should proceed in an organized, sequential fashion to prevent initial procedures from adversely affecting subsequent test results. It is recommended that samples for aerobic and fungal culture be obtained prior to the application of fluorescein stain or topical anesthetic.5 For routine culture of samples from the eyelids, cornea, or conjunctiva, recommended media include blood agar, the basic culture medium for most aerobic bacteria, and Sabouraud’s agar for suspected fungal infection. Chocolate agar should be used in addition to blood agar if more fastidious bacteria, such as nutritionally variant streptococci, are suspected.6 Agar inoculation followed by immediate incubation is preferred to use of transport media.
Cytology may be indicated in acute conditions as well as refractory or recurrent cases of external ocular disease that manifest as exudative, purulent, or proliferative processes affecting the eyelid, membrana nictitans, conjunctiva, and/or cornea. Cytology is an essential tool used to help characterize the cellular response (neutrophilic, lymphocytic, eosinophilic, granulomatous) and to identify parasites, bacteria, or fungi. In humans, scrapings are most helpful when done in the early, active phases of disease before secondary ocular changes ensue.7
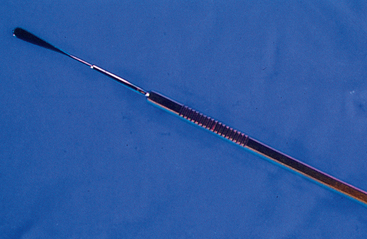
Cytologic harvesting methods include impression, fine-needle aspiration, swabbing, and brushing techniques. In ophthalmology, impression cytology has been most frequently used in the study of goblet cells and corneal and conjunctival epithelial cells.8–11 Swabbing the conjunctiva and cornea using cotton-tipped applicators does not yield a high enough cell count and therefore is not a recommended cytologic technique for the eye.12 Scraping is a common cytologic technique used for collection of ocular surface cells. Various instruments have been used in this technique, including Kimura spatulas, iris repositioning spatulas, butt of scalpel blade handles, and chemistry measuring spatulas (Fig. 3-1).3,13–14 More recently, exfoliative cytology of the conjunctiva and cornea using a brush technique has been reported.15–16 This method has been shown to yield sufficient number of cells for cytologic interpretation, and a consistent epithelial cell monolayer can be produced.
If a spatula is used for the collection of ophthalmic cytologic specimens, repeatedly flame sterilize the tip of the instrument and allow it to cool prior to touching the ocular surface. The platinum tip of the Kimura spatula permits rapid heating and cooling of this instrument, making it ideal for cytologic sampling. After cleaning mucopurulent exudate from the ocular surfaces and anesthetizing the cornea and conjunctiva with a topical ophthalmic anesthetic, scrape the sterilized spatula along the surface or margins of the lesion several times in the same direction (not back and forth) until a small droplet of material is collected on the edge or tip of the spatula (Fig. 3-2). Use gentle pressure so that the sample does not become contaminated by blood. Immediately transfer the collected material to a glass slide and gently spread or smear it as described in Chapter 1. If the material is sticky and thick, add a drop of sterile saline before spreading the sample. Ideally, spread the cells thinly so the smear is only one cell layer thick.
Corneal Scrapings
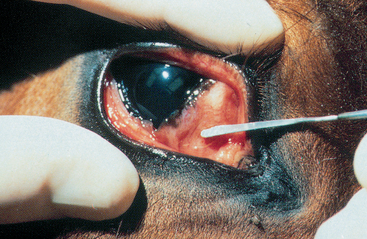
In cases of ulcerative keratitis, the corneal ulcer margin is sampled directly for both cytologic preparations and cultures. Cytologic examination of conjunctival scrapings or exudate smears alone in cases of ulcerative keratitis may be misleading because the number and types of organisms in the exudate may be different from those present in the corneal stroma. Although it is generally recommended to collect corneal samples for culture prior to application of topical anesthetic, 2 drops of 0.5% proparacaine HCl does not significantly affect the numbers and types of organisms cultured from ocular surfaces.5,17 Moistening the swab with thioglycolate broth before collecting the culture specimen increases the chance of recovering organisms from the ocular surface.18 Because various species of bacteria and fungi can be isolated from the eyelid margins and conjunctival fornices in normal horses, it is important to obtain samples from the corneal lesion and not the eyelids, conjunctiva, or ocular discharges when bacterial or mycotic keratitis is suspected.19,20
Corneal scrapings may require vigorous but careful debridement of the involved area to obtain sufficient cellular material for diagnosis. A topical anesthetic, such as 0.5% proparacaine HCl, should be applied to the cornea before a corneal scraping is collected. However, with deep ulcers or descemetocele formation, excessive pressure on the eye or eyelids must be avoided to prevent iatrogenic globe rupture. In cases of equine keratomycosis, fungi most commonly inhabit the deeper corneal layers, with an apparent affinity for Descemet’s membrane.21,22 If a corneal lesion infected with fungal hyphae has epithelialized prior to any diagnostic procedures being performed, it may be necessary to remove the superficial epithelium to obtain meaningful culture and cytology samples. Multiple, deep corneal scrapings may be required to establish the diagnosis, especially in cases with satellite lesions (Fig. 3-3).23,24s Finally, a partial keratectomy or corneal biopsy may be necessary to demonstrate fungal elements if corneal scrapings are negative.25 Impression imprints of the biopsied portion of cornea can be made for cytologic examination before the tissue is fixed in formalin for histopathologic examination. Special stains may help detect fungal elements (see discussion of special stains later in this chapter).
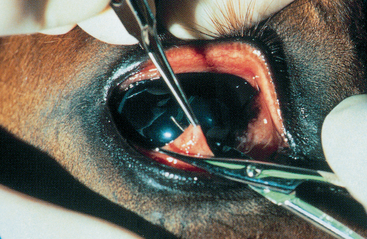
Conjunctival and Corneal Excision Biopsy
Grasp the tissue directly adjacent to the affected area with fine forceps, and use scissors or scalpel with trephine to excise a biopsy sample (Fig. 3-4). Make imprints for cytologic evaluation of the excised tissue before fixing the specimen in formalin for histologic processing. Examine wet preparations of the excised tissue for microfilariae. To make wet preparations, mince the tissue with a drop of warm physiologic saline and place it on a glass slide. Keep the specimen warm and examine it periodically over 5 to 60 minutes for microfilariae. Microfilariae may be observed under low-power magnification.26
Aqueous, Vitreous, and Subretinal Paracentesis
Severe anterior uveitis is sometimes associated with an inflammatory cell egress into the anterior chamber that may settle ventrally, resulting in hypopyon (Figs. 3-5 and 3-6). Aqueous paracentesis may be indicated in severe, nonresponsive anterior uveitis when the aqueous humor is cloudy or opaque, as in cases of infectious anterior uveitis or intraocular neoplasia. Vitreous or subretinal paracentesis should be considered in horses with marked vitreous opacification, exudative retinal separations, or suspected bacterial or fungal endophthalmitis. Blood cultures may aid diagnosis of bacterial endophthalmitis; therefore, intraocular paracentesis may not be needed in cases of Rhodococcus equi infection.27
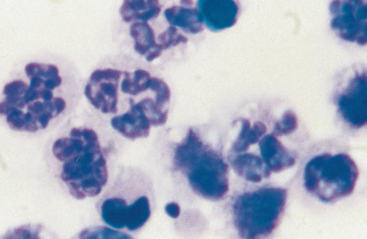
Fig. 3-6 Smear of hypopyon aspirate.
Smear from the horse shown in Fig. 3-5. The cellular response is primarily neutrophilic, though fibrin and small numbers of erythrocytes, plasma cells, lymphocytes, and pigment-containing cells may be present.
Tap the anterior chamber through a simple limbal approach (Fig. 3-7). Initially, clean the conjunctiva and cornea with a dilute (5%) aqueous povidone-iodine solution and rinse with sterile 0.9% saline. Use an eyelid speculum to retract the eyelids from the anterior surface of the eye during this procedure. Using a delicate thumb forceps, grasp the bulbar conjunctiva and stabilize near the point of entry into the globe. Attach a 22- or 25-gauge needle to a 1-ml syringe (with the plunger seal already broken) and then insert it under the bulbar conjunctiva, 3 to 4 mm from the limbus. Thread the needle under the conjunctiva toward the limbus, and enter the anterior chamber parallel to the surface of the iris. Collect a 0.5- to 1.0-ml sample of aqueous humor with slow, gentle aspiration. The region of bulbar conjunctiva that is threaded by the needle prior to insertion into the anterior chamber helps to create a small “tunnel” that will act as a seal once the needle is slowly withdrawn. Gentle pressure over the exit wound using a thumb forceps or sterile cotton-tipped applicator will also help control further aqueous egress. An alternative method of performing aqueocentesis has been described using an aqueous paracentesis pipette.28
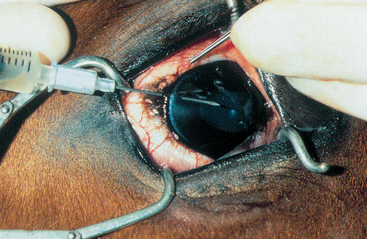
Fig. 3-7 Anterior chamber paracentesis.
A 25- or 22-gauge needle attached to a sterile 1-ml syringe is inserted into the bulbar conjunctiva, 3 to 4 mm from the limbus. The needle is threaded deep to the conjunctiva toward the limbus and enters the anterior chamber parallel to the surface of the iris.
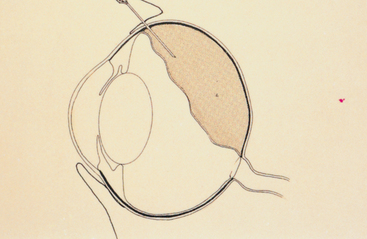
Vitreous and subretinal paracenteses are performed through a pars plana approach, taking care not to puncture the lens (Fig. 3-8). General anesthesia and ocular surface preparation are similar to those described for aqueocentesis. In addition, pharmacologically dilate the pupil to provide better visualization of the posterior segment during the sampling procedure. In vitreal and subretinal paracenteses, attach a 22- or 23-gauge hypodermic needle to a 1-ml syringe (with the plunger seal already broken) and insert approximately 7 mm behind the limbus in the dorsolateral quadrant, avoiding the inferior and medial quadrant, where the retina extends more anteriorly in most species.29 The vitreous is usually liquefied in disease, so only a small amount of negative pressure on the syringe attached to the needle is required. However, neoplastic or granulomatous cellular material may require a larger needle or greater suction. The amount of fluid aspirated from the vitreous or subretinal space usually does not exceed 0.2 ml. After sample collection, withdraw the needle and hold the conjunctival site with a forceps for a few seconds to seal the wound.
Samples collected via paracentesis can be submitted for cytology, culture, antibody titers, and other more specialized diagnostic techniques. When preparing the sample for cytology, deposit a few drops of the collected fluid onto a glass slide, spread, fix, and stain as described in Chapter 1. In poorly cellular samples, centrifugation of the sample may help concentrate the cells and give a higher yield for interpretation. Make cytologic preparations from aqueous humor as soon as possible after collection to avoid disintegration of cells in the low-protein aqueous humor.30 If special diagnostic techniques are desired, it is prudent to contact the laboratory to which the sample will be sent prior to paracentesis to ensure that the material collected is handled promptly and correctly after being obtained from the equine globe.
Orbital Aspiration
Retrobulbar fine-needle aspiration may be the least invasive way to acquire diagnostic specimens from space-occupying orbital lesions. The globe and the optic nerve must be avoided. In humans, fine-needle aspirates of the orbit have been useful for diagnosis of unresectable malignant orbital neoplasms, possibly eliminating a need for further surgical intervention.31 Despite being more efficient and economical in some cases, cytologic diagnosis obtained by orbital aspiration is not as accurate when compared to surgical biopsy.32,33
Staining Ocular Specimens
Romanowsky-type stains (Wright’s, Giemsa, modified Wright’s-Giemsa stain) are useful for identifying inflammatory and neoplastic cells, bacteria, fungi, or cytoplasmic inclusions. With Romanowsky stains, it is possible to recognize and differentiate bacterial cocci (Staphylococci, Streptococci), large rods (Bacillus), and small (some bipolar) rods (Pseudomonas, coliforms). Most fungi readily stain with Romanowsky stains. Gram stain may be used to characterize bacteria as gram positive or negative. It may be difficult to identify gram-negative organisms in the red background of exudates, including ocular discharges, stained with Gram stain. Identification of the organism from culture and antimicrobial sensitivity testing directs definitive therapy; however, immediate treatment may be instituted based on the staining properties and morphologic features of the offending organism and knowledge of common equine ocular pathogens (Table 3-1).
TABLE 3-1 Gram-Staining Reactions in Conjunctivitis and Keratitis
| Staining Characteristics | Most Probable Causative Organism |
|---|---|
| Gram-Positive | |
| Cocci, singly or in clusters | Staphylococcus spp. |
| Cocci in chains | Streptococcus spp., nutritionally variant Streptococcus spp. |
| Rods | Bacillus spp. |
| Filaments | Fungi, especially Aspergillus spp., Fusarium spp., Penicillium, Phycomycetes |
| Gram-Negative | |
| Diplobacilli | Moraxella spp. |
| Rods | Pseudomonas aeruginosa, coliforms |
| Filaments | Fungi |
Special Stains
Immunofluorescent antibody (IFA) testing is not commonly used in diagnosis of equine ocular diseases. The IFA test has been used for conjunctival antigen detection in chlamydial and rabies virus infections.34,35 Borrelia burgdorferi spirochetes have been detected in the anterior chamber of a pony using IFA techniques.36 Confirmation of equine herpesvirus, serotype 2, has been reported using fluorescent antibody staining.37 The Calcofluor white–potassium hydroxide technique is a much more sensitive test for microscopic detection of fungi than PAS or GMS staining.38 Calcofluor white (Cellufluor) is a fluorescent stain that is readily absorbed by the chitin component in fungal cell walls. Diagnostic specimens are stained for 1 minute, counterstained, and then examined under a microscope equipped with ultraviolet light. Fungi appear yellow-green against a red-orange background. The Cellufluor examination is useful for diagnosis of keratomycoses and for dermatophytoses.39–41 Not all referral laboratories can perform these tests, so the laboratory should be contacted before sample submission.
Normal Ocular Cytologic Findings
Conjunctival and Corneal Epithelial Cells
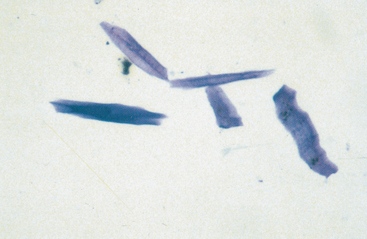
Morphologic features of normal corneal and conjunctival cells of horses are similar to those described for dogs and cats.13,14 Scrapings from healthy cornea or conjunctiva characteristically yield sheets of epithelial cells containing cells from one or more of their representative layers. Superficial epithelial cells are flattened with large amounts of pale blue cytoplasm and round to oval, central, basophilic nuclei (Fig. 3-9). Cells from the intermediate zone are more polyhedral in appearance when compared to superficial epithelial cells. Basal or parabasal cells, originating from the deepest layers of the cornea and conjunctiva, are round or cylindrical, with less abundant but darker-staining cytoplasm (Fig. 3-10). Keratinized corneal or conjunctival epithelial cells are abnormal, except for conjunctival cells adjacent to the eyelid margin, which are normally keratinized. Epithelial cells that are keratinized have dark blue to blue-green cytoplasm, with a degenerate, pyknotic, or absent nucleus. Many of these cells “scroll” when smeared, resulting in a jagged or sharply pointed and long rectangular appearance (Fig. 3-11). This artifact should not be mistaken for foreign material.
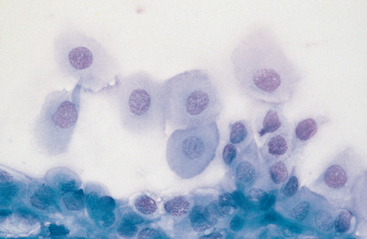
Fig. 3-9 Conjunctival smear.
Epithelial cells have pale blue cytoplasm and round to oval, central, basophilic nuclei.
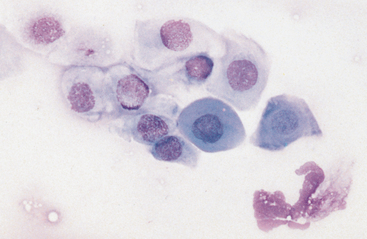
Fig. 3-10 Conjunctival smear.
Cells from the deeper (basal or parabasal cells) are round to cylindric, with less but darker-staining cytoplasm than more superficial cells.
Melanin Granules
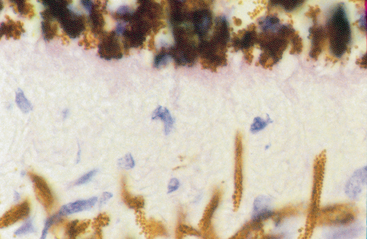
Intracytoplasmic melanin granules are common in epithelial cells collected from pigmented areas, such as the limbus, or pigmented ocular lesions. Pigment granules are not found in normal corneal epithelial cells. Melanin granules appear dark green to brown or black (Fig. 3-12) and may demonstrate slightly different shapes, depending on their location. Melanin granules in iridal cells are generally oval to round versus more lanceolate shaped in retinal pigmented epithelial cells (Fig. 3-13).29 Small amounts of free melanin granules and a few melanin-containing cells (melanocytes, melanophages) may be seen in aqueous paracentesis samples.42
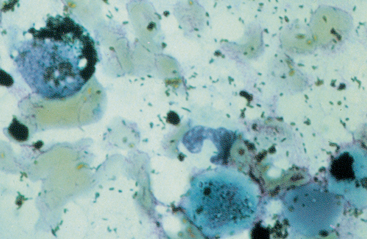
Fig. 3-12 Conjunctival smear.
Melanocytes and melanin granules appear dark green to brown or black. Numerous melanin granules are dusted throughout the background.
Microorganisms
Populations of bacterial flora normally inhabit the ocular mucosal surface and can be found on routine cytologic specimens from healthy horses. Gram-positive isolates predominate, with some gram-negative organisms found less commonly.19,43 Long-term use of topical ophthalmic antibacterials may lead to the overgrowth of pathogenic bacteria, yeast, or fungi.44
Goblet Cells and Mucus
Goblet cells are large, distended cells with a peripheral nucleus. The round to oval nucleus may be molded into a crescent shape when the goblet-cell cytoplasm becomes laden with mucus. Intracellular mucus may appear as a clear area or may stain red to blue (depending on the quantity present) with Romanowsky stains (Fig. 3-14). With periodic acid–Schiff (PAS) stain, mucus stains a dark pink (Fig. 3-15). Goblet cells tend to occur in clusters and are found in greatest concentration in the lower nasal fornix in some species.45
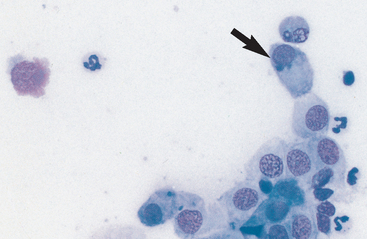
Fig. 3-14 Conjunctival smear.
A goblet cell, featured near the top of this image, has an eccentric nucleus and a clear, pale blue cytoplasm.
Erythrocytes and Inflammatory Cells
Occasional erythrocytes, lymphocytes, monocytes, plasma cells, and neutrophils are usually present in conjunctival scrapings from normal horses. If scraping has been overzealous, the number and type of these cells may be increased due to peripheral blood contamination. Inflammatory diseases usually result in much higher numbers of neutrophils, lymphocytes, monocytes, or plasma cells than occur with peripheral blood contamination alone. Therefore, if these cells are present in large numbers without accompanying erythrocytes, an inflammatory response is most likely present. The bulbar surface of the membrana nictitans normally contains some lymphoid follicles. Scrapings from this area may contain numerous, variably sized lymphocytes, similar to those found in normal lymph nodes. Therefore, correct interpretation of a cytologic specimen where inflammatory cells are present should be made with consideration to the presence or absence of clinical signs of ocular inflammation and the number and type of inflammatory cells seen. Eosinophils and basophils are not normally present in conjunctival or corneal scrapings from horses.
Aqueous Humor
Anterior chamber aspirates from normal eyes are virtually acellular.30 A few erythrocytes, mature lymphocytes, histiocytes, free pigment granules, and pigment-containing cells are occasionally present. Because the protein content of normal aqueous humor is very low, in vitro disintegration of cells may be rapid.30
Eyelids
Bacterial Lesions
Primary bacterial blepharitis is rare in horses. Eyelid abscesses are usually secondary to a foreign body reaction.46 High numbers of degenerate and nondegenerate neutrophils, with or without bacteria, are seen cytologically. Chronic blepharitis with draining tract formation secondary to a bone sequestrum of the zygomatic arch was reported in one horse.47 Moraxella sp. may cause ulcerative dermatitis with erosions of the mucocutaneous junction of the eyelids and medial canthus.48,49
Fungal Lesions
The eyelids of horses may become infected by cutaneous and systemic fungi. Dermatophytosis due to infection with Trichophyton sp. or Microsporum sp. may result in eyelid alopecia, crusting, scaling, and dermatitis (see Chapter 2). Definitive diagnosis is based on microscopic examination of hairs and surface debris, fungal culture, and skin biopsy. Because most domestic animal dermatophyte infections are of the ectothrix type, it is not usually necessary to use hair shaft–clearing techniques involving potassium hydroxide to visualize the fungi.50 Hair and keratin can be examined for fungal hyphae and conidia simply by suspending the specimens in mineral oil or saline. Arthroconidia may be seen on the hair shaft, with hyphae invading and penetrating the hair shaft. Cutaneous scrapings may also be stained with Wright’s stain. Arthroconidia appear as small (2 to 4 μ), dark blue, round to oval spores (see Chapter 2).
Phycomycosis (mycetomas) are subcutaneous infections occurring in tropical climates that may be caused by several different species.51 The eyelids may be involved when lesions occur on the head and are characterized by pruritus, granulation tissue, and necrotic draining tracts.
Histoplasma farciminosum is enzootic in Africa, Asia, and Eastern Europe. Epizootic lymphangitis, caused by H. farciminosum, may be characterized by a localized granuloma along the free margin of the eyelid.52 Conjunctival and lacrimal involvement may also occur. Diagnosis is made by seeing the double-contoured, thin-walled, budding, oval yeast cells, 2 to 3 μ in diameter (Plate 4C) on smears or aspirates from the affected tissue. Histoplasma infections are associated with a mononuclear-cell response, with monocytes and macrophages predominating. Neutrophils are generally found in fewer numbers than mononuclear cells and are nondegenerate. Reactive lymphocytes may be present. Cryptococcus mirandi may also cause ulcerative eyelid lesions (Plate 4H).46
Viral Lesions
Viral causes of equine eyelid disease include facial papillomas and horsepox virus.46 Equine papillomas (warts) resulting from a papova virus can be commonly found on the face of young horses. These warts may regress spontaneously in several months or can be treated via surgical removal, cryotherapy, or autogenous vaccination.53
Horsepox is a rare, benign disease of horses in Europe caused by an unclassified poxvirus. Horsepox has been reported once in North America in a donkey.54 The buccal form of horsepox (contagious pustular dermatitis) is characterized by multiple vesicles, umbilicated pustules, and crusts on the lips and buccal mucosa. In severe cases, the eyelids and conjunctiva may be affected. It may be possible to find large, eosinophilic, intracytoplasmic poxvirus inclusions in vacuolated keratinocytes in cutaneous scrapings and conjunctival scrapings.
Parasitic Lesions
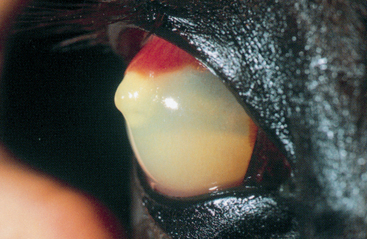
Eyelid disease may result from ectoparasitism or migrating helminth larvae. Habronemiasis is a common cause of eyelid, conjunctival, caruncular, and membrana nictitans granulomas.55,56 House flies and stable flies (Habronema muscae, H. microstoma, or Draschia megastoma) act as the biologic vectors of habronemiasis, so ocular lesions are seasonal in occurrence, predominating in warm months.46 Larvae deposited on moist ocular tissues migrate into the skin, conjunctiva, and nasolacrimal system and cause an intense inflammatory reaction. The ocular lesions are characterized by pruritic, nonhealing, raised, ulcerative, granulomatous lesions, sometimes associated with fistulous tracts (Fig. 3-16). Other clinical signs are discussed in later sections.
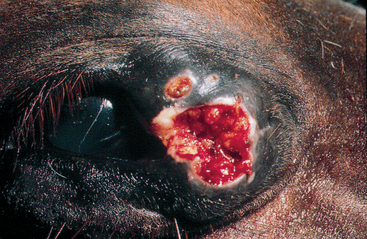
Fig. 3-16 Habronemiasis.
Habronemiasis is characterized by nonhealing, raised, ulcerated lesions containing yellow, caseous, gritty nodules.
Cytologic examination of eyelid habronemiasis is characterized by numerous eosinophils, mast cells, neutrophils, and plasma cells, often without positive larvae identification (Fig. 3-17).46 Larvae may be better identified in minced tissue preparations from a nodule. Without observation of the definitive cytologic presence of larvae, it may be difficult to distinguish eyelid habronemiasis from a mast-cell tumor, eosinophilic granuloma with collagen degeneration (nodular necrobiosis), or phycomycosis.57 Habronema and Draschia larvae may also invade other ulcerative dermatoses, such as sarcoid or squamous-cell carcinoma, which may complicate the cytologic findings, making definitive diagnosis more difficult. Therefore, histopathologic examination of any granuloma, in addition to cytologic examination, is recommended for definitive diagnosis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree