(1)
Arezzo, Italy
3.1 Introduction
In this chapter, cytopathological findings of the main non-neoplastic skin diseases are discussed. Unlike skin neoplasias, non-neoplastic skin diseases, which in this book are also named inflammatory diseases, are characterised by a wide clinical polymorphism.
Although the following classification is not useful for diagnostic purposes, skin lesions are usually classified as primary, which represent the damage of the disease on the skin, and secondary, the evolution of lesions over time or the outcome of self-trauma. Skin lesions that are observed on a dermatological patient are numerous and morphologically very varied, and although the macroscopic appearance makes them easily recognisable, cytological findings allow us to interpret them. Unfortunately, their nature may not always be able to be established through cytology; in these cases, other diagnostic techniques or a histopathological examination are necessary. In this chapter, only the skin lesions, in which a cytological investigation may be diagnostic or useful for diagnosis, are discussed. The author believes that to better interpret the cutaneous inflammatory cytology, it is mandatory to be able to distinguish clinical aspects of the skin lesions from where the cells have been sampled and, for this reason, he prefers to discuss the cytological findings starting from clinical signs. This more practical approach allows veterinarians, even those less confident with dermatology, to interpret cytological results in an easier and more rational way, and to gain from them all the information possible for their interpretation.
3.2 Papules
As mentioned in the chapter on sampling techniques, it is very unlikely to obtain a diagnosis through cytological examination from papules, as they usually result in poor cellular specimens.
Papules with an intact surface are too small for FNB and cells cannot be collected by the imprint technique; therefore, only crusted papules permit the collection of a few cells, which, even if they are not diagnostic, may guide the clinician in the choice of further diagnostic tests.
3.2.1 Papular Diseases in Dogs
3.2.1.1 Pyoderma
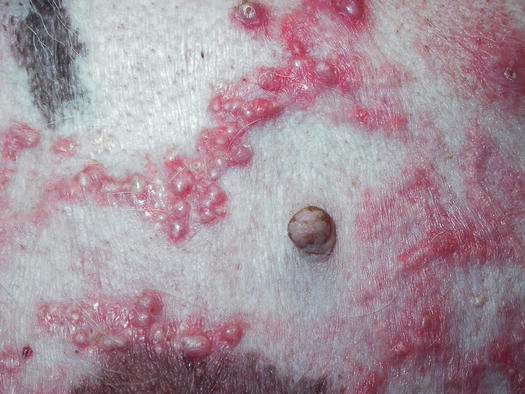
In dogs, papules are frequently observed during superficial staphylococcal pyoderma (Ihrke 1996; Miller et al. 2013). In this disease, papular lesions are very often associated with pustules and epidermal collarettes, which help the clinician to suspect the disease (Fig. 3.1). When present, pustules must always be preferred to papules for sampling cells, but when papules are the only lesions available, it is possible try to collect cells via the imprint technique.
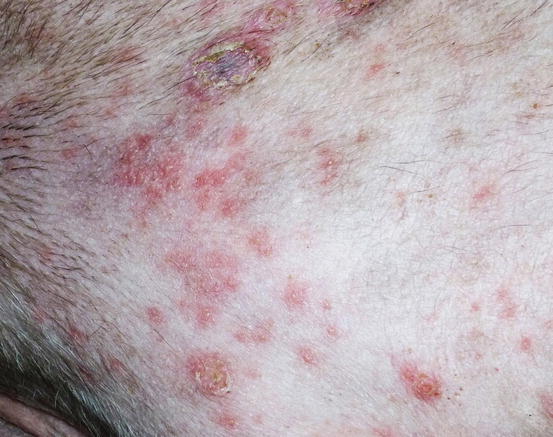

Fig. 3.1
Papules, pustules and epidermal collarettes on the abdomen of a dog with superficial pyoderma
Cytological Findings
Slides from papular bacterial infection are sparsely cellular, usually haemocontaminated and have few moderately karyolytic neutrophils. In more exudative papules or in crusted papules, it is very rare to detect cocci phagocytosed by neutrophils. Usually, the superficial crusts that cover the papules entrap a large number of cocci, which are spread on the background of the slide; in these cases, grouped bacteria must not be interpreted as pathogenic, as they are simply indicative of environmental contamination (Fig. 3.2).
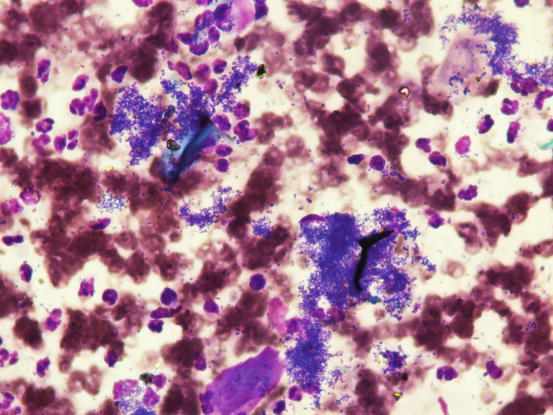

Fig. 3.2
Blood, rare neutrophils and multiple aggregates of bacteria indicative of environmental contamination
3.2.1.2 Scabies and Fleabite Allergic Dermatitis
Papules are very common in many canine parasitic and hypersensitivity diseases. Scabies and fleabite allergic dermatitis are characterised by papules and small crusted papules. In these diseases, it is not possible to obtain diagnosis through cytology, but the detection of many eosinophils, together with the history and the distribution of the lesions on the body, can help the clinician to suspect the disease.
3.2.2 Papular Diseases in Cats
Papular lesions in cats are observed in numerous diseases and are grouped under the umbrella of miliary dermatitis, so called because the papular lesions are the same size as millet seeds. Miliary dermatitis represents a very common and aspecific pattern of feline skin reaction, which occurs in many diseases with different causes, but they all share the presence of an eosinophilic infiltrate into the dermis. It is deduced, as in all these diseases, that the cytological specimens are similar.
As for dogs, the cytology of crusted papules does not permit a diagnosis, but may be helpful in confirming a clinical suspicion or aid the clinician in the selection of further tests to perform.
3.2.2.1 Hypersensitivity Diseases
Among the most common causes of miliary dermatitis in cats are fleabite dermatitis and food or environmental allergy (no-food and no-flea hypersensitivity) (Hobi et al. 2011; Favrot et al. 2011; Miller et al. 2013; Ravens et al. 2014).
Skin lesions are identical and for this reason do not allow clinical differentiation between them (Fig. 3.3).
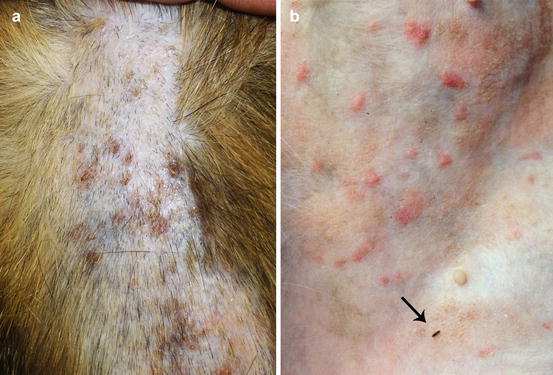

Fig. 3.3
(a) Crusted papules on the back of a cat with miliary dermatitis; (b) erythematous papules in a cat with fleabite dermatitis: note the adult flea (arrow)
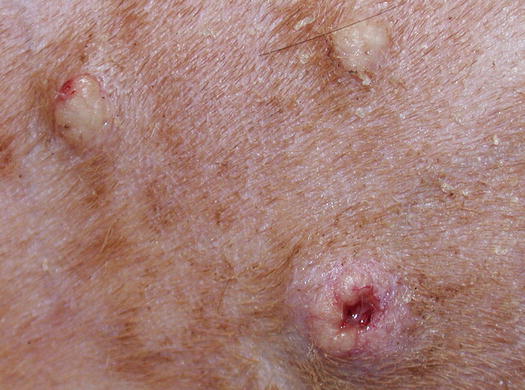
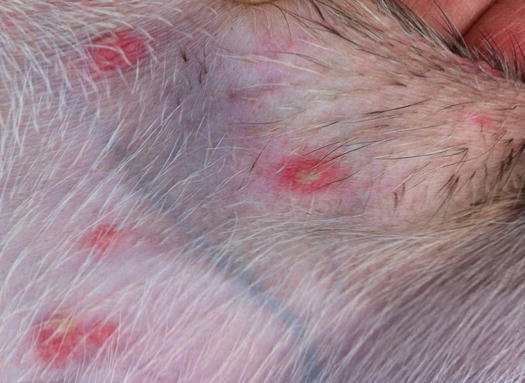
Another disease characterised by papules, and less frequently by papular–nodular lesions, is mosquito bite hypersensitivity (Gross et al. 2005). The cause of this disease is an immune-mediated type I hypersensitivity reaction against antigens of mosquitos. Clinically, it is characterised by crusted papules on the head and especially on the outer surface of the pinna, the bridge of the nose and on the nose (Figs. 3.4 and 3.5) (Nagata and Ishida 1997). Less frequently, lips, eyelids and paws are affected. It seems that black-coated domestic short-haired (DSH) cats are most predisposed to becoming hypersensitive, although the cause of this preference has never been investigated or demonstrated. Skin lesions are very pruritic and tend to ulcerate owing to self-trauma.
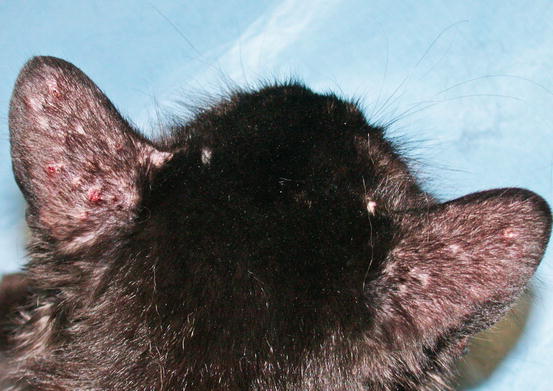
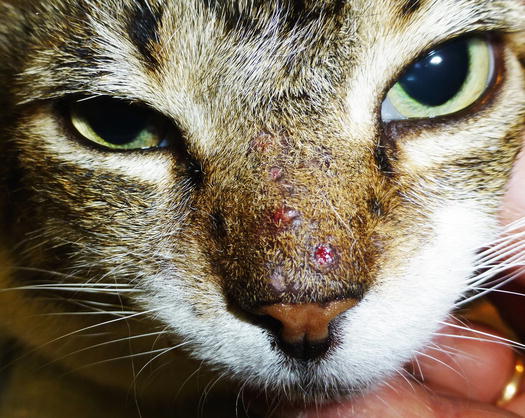

Fig. 3.4
Multiple ulcerated and crusted papules on the pinnas of a black -coated DSH cat with mosquito bite hypersensitivity

Fig. 3.5
Crusted papules on the nose of a cat with mosquito bite hypersensitivity
Cytological Findings
In all the hypersensitivity diseases characterised by miliary dermatitis, cytological specimens show a variable number of eosinophils. In some cases, in small crusted papules, the slides will be composed only of blood, with a basophilic proteic background, few eosinophils intermingled with neutrophils and rarely histiocytes can also be observed (Figs. 3.6 and 3.7). In larger ulcerated papules and in more successful samples, it is possible to obtain many more cellular specimens, rich in eosinophils and with a variable number of macrophages (Fig. 3.8).
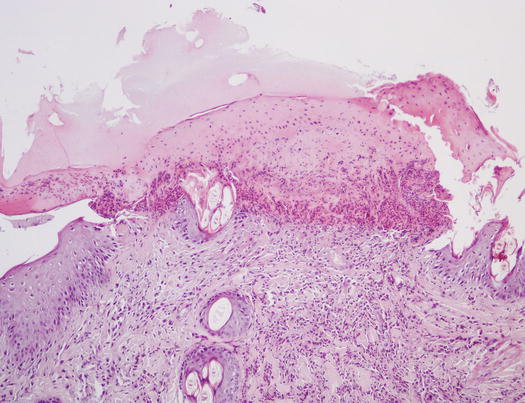
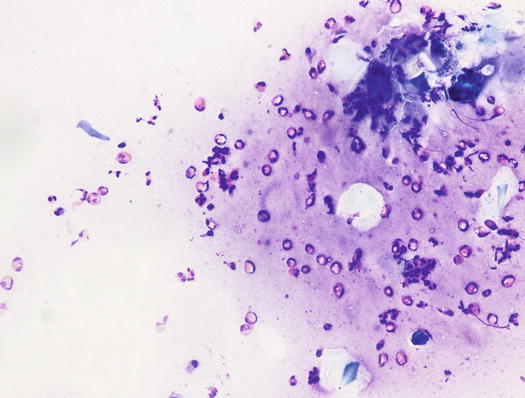
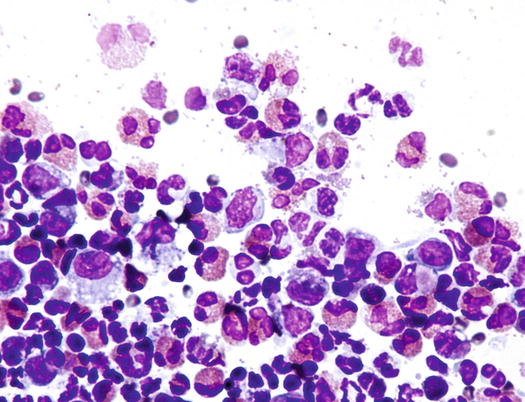

Fig. 3.6
Histopathology of a crusted papule: sero-neutrophilic crusts covering a small erythematous papule

Fig. 3.7
Cytology of miliary dermatitis: many eosinophils and few corneocytes on a proteinaceous background

Fig. 3.8
Cytology of miliary dermatitis: many eosinophils intermingled with neutrophils and macrophages
Rarely, in some cases, it is possible to observe a few basophils, distinguishable from eosinophils by their larger size, and especially for characteristic intracytoplasmic basophilic granules, which stain pale blue (Fig. 3.9).
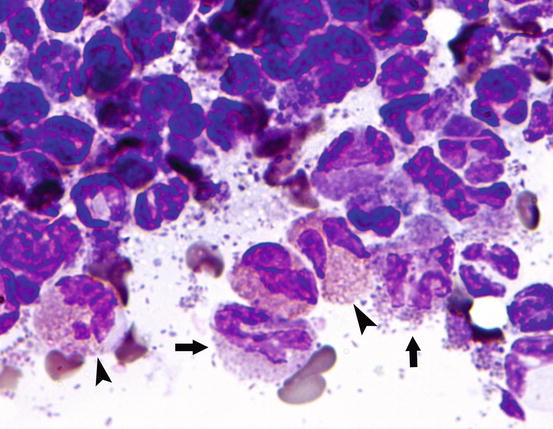

Fig. 3.9
Cytology of miliary dermatitis: basophils (arrow) and eosinophils (head of arrow)
3.2.2.2 Benign Papular Mastocytic Hyperplasia
In human medicine, urticaria pigmentosa is a form of cutaneous mastocytosis. An idiopathic papular disease, named benign papular mastocytic hyperplasia, and also defined as urticaria pigmentosa-like disease, has been reported in cats (Vitale et al. 1996; Noli et al. 2004). This disease has been reported in Sphynx and Devon rex cats as a clinical form of mastocytosis exclusively localised to the skin. The affected cats develop, in the early stages, many crusted papules (miliary dermatitis), which spread all over the body and with a linear configuration that is more evident on the ventral region of the abdomen (Fig. 3.10). Skin lesions often tend to coalesce, resulting in little plaques with thickened skin (Fig. 3.11). The cause of this disease is not known and as it is observed almost exclusively in these feline breeds, it is assumed that there may be a genetic cause. The healing of the lesions following immunomodulatory therapy and the non-specificity of the histopathological pattern make it impossible to differentiate from feline hypersensitivity diseases, which must always be ruled out. In rare cases, miliary dermatitis may be secondary to dermatophytic hypersensitivity. Some Devon rex cats affected by dermatophytosis and with clinical and histopathological lesions similar to urticaria pigmentosa-like disease, have been reported (Colombo et al. 2012).
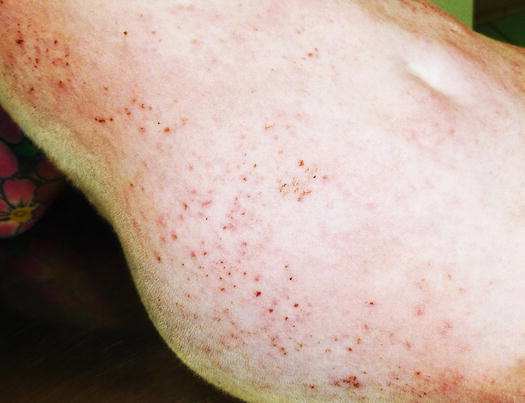
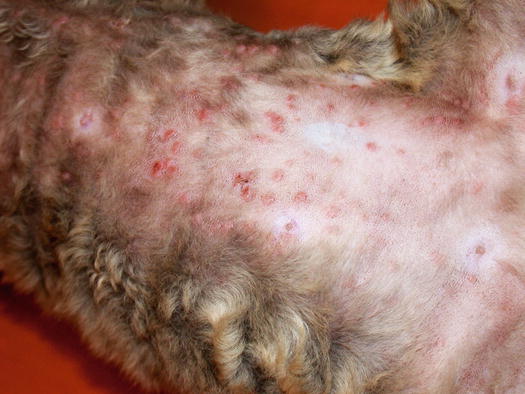

Fig. 3.10
Miliary dermatitis on the abdomen of a Sphynx cat affected by benign papular mastocytic hyperplasia

Fig. 3.11
Multiple papules and small plaques on the abdomen of a Devon rex with urticaria pigmentosa-like disease
Cytological Findings
Histologically, the lesions are characterised by slight superficial perivascular dermatitis with a significant number of mast cells and few eosinophils (Fig. 3.12). Cytology obtained using the imprint technique, after removal of the crust that covers the top of crusted papules, is haemodiluted and usually characterised by neutrophils. Rarely, in more successful slides, specimens can show some well-differentiated mast cells and rare eosinophils, which do not authorise the diagnosis of benign papular mastocytic hyperplasia, but reinforce the suspicion of the disease, which must be confirmed with histopathology (Fig. 3.13).
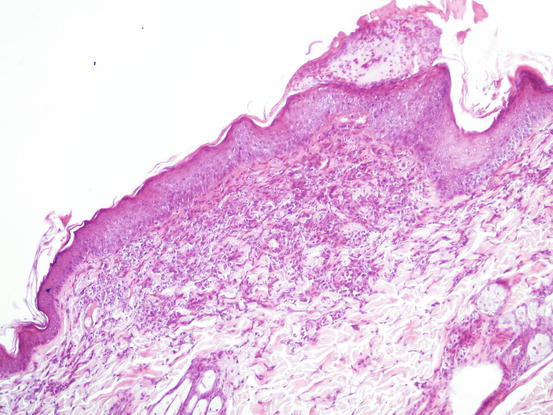
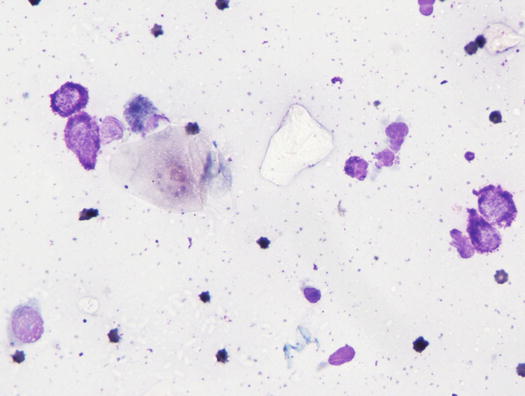

Fig. 3.12
Histopathology of benign papular mastocytic hyperplasia: superficial dermal infiltration of mast cells and eosinophils with mild hyperplasia of the epidermidis

Fig. 3.13
Cytology of benign papular mastocytic hyperplasia: many well differentiated mast cells collected from papules
3.3 Nodular Papules
Nodular papules provide more diagnostic specimens compared with papules, because their size permits more cells to be collected.
3.3.1 Deep Pyoderma (Furunculosis)
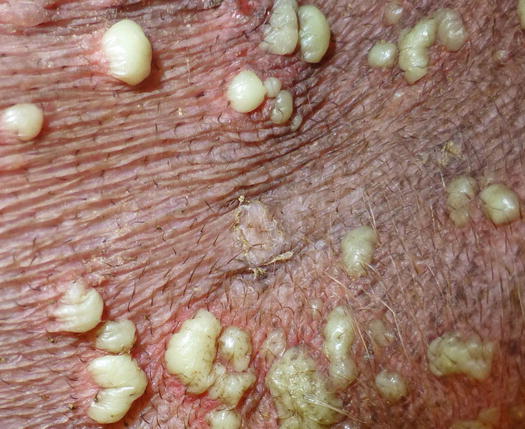
Histologically, rupture of the hair follicles following bacterial staphylococcal folliculitis creates small dermal pyogranulomas focused on corneocytes or on hair shaft fragments, both from follicular lumen (Figs. 3.14, 3.15, and 3.16) (Gross et al. 2005). Furunculosis in dogs with deep pyoderma is clinically characterised by papular–nodular lesions that are mostly detectable on the chin, on the limbs and among the interdigital areas, especially in shorthaired breeds (Figs. 3.17, 3.18, and 3.19) (Miller et al. 2013). Many nodular papules fistulise on the skin surface, where a purulent exudate is discharged; therefore, a large amount of cells can be collected for cytopathological examination via FNB of intact lesions or via an imprint on the exudate released from draining tracts.
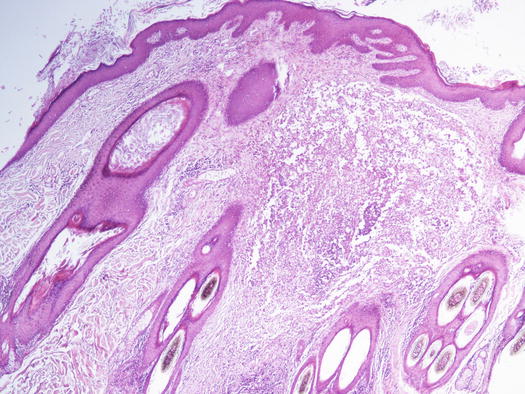
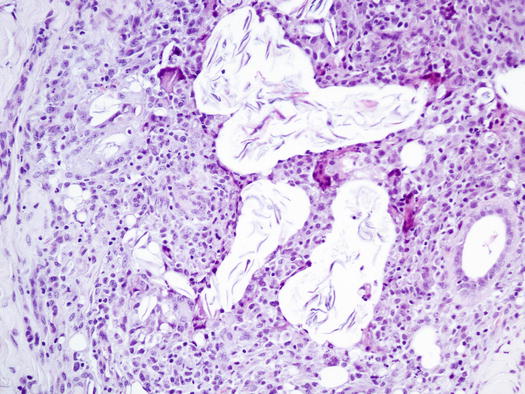
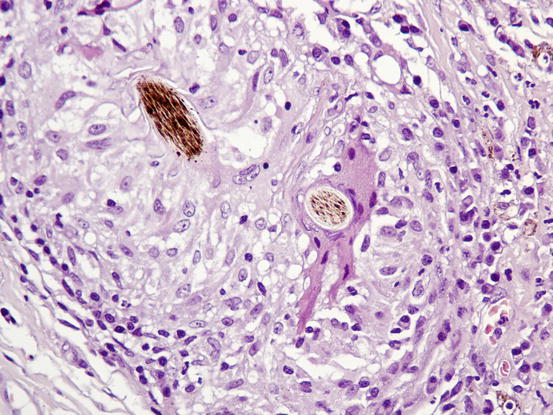
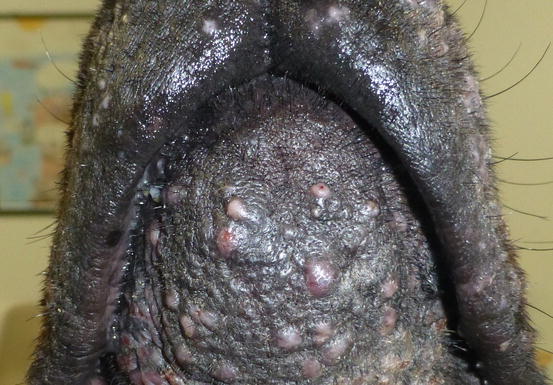
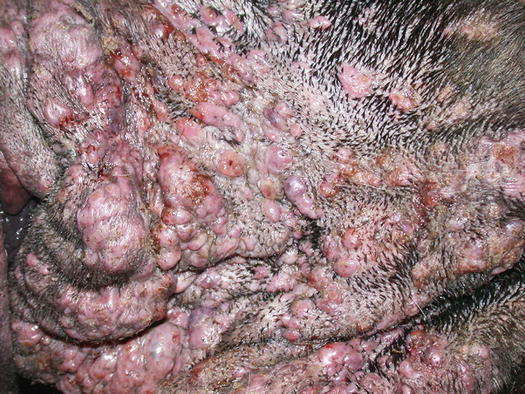
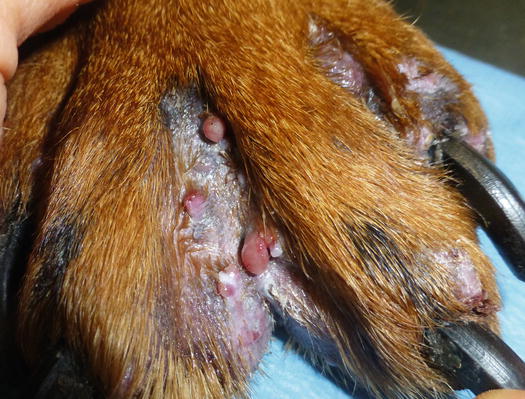

Fig. 3.14
Histopathology of deep pyoderma: large nodular pyogranulomatous inflammation replacing normal pilosebaceous units in a dog with deep pyoderma

Fig. 3.15
Histopathology of deep pyoderma: granulomatous inflammation around lamellar corneocytes

Fig. 3.16
Histopathology of deep pyoderma: epithelioid macrophages and a large giant cell; the latter is phagocytosing a hair shaft fragment

Fig. 3.17
Multiple nodular papules on the muzzle of a Doberman affected by deep pyoderma

Fig. 3.18
Many single and confluent nodular papules on the muzzle and lips of a Neapolitan mastiff with deep pyoderma

Fig. 3.19
Papular–nodular lesions in the interdigital spaces of a Doberman with pyoderma
Cytological Findings
Samples obtained by FNB are certainly of better quality and are usually composed of many inflammatory cells, mainly represented by numerous karyolytic and segmented neutrophils and macrophages. The latter are of variable size and morphology, with large and often vacuolated cytoplasm, and show phagocytosis of leukocytes, cellular debris, amorphous material and, sometimes, of melanin pigment (melanophages) in animals with pigmented skin or with post-inflammatory hyperpigmentation (Fig. 3.20). In dogs with furunculosis, keratin is spread in the dermis as lamellar corneocytes or as hair shaft fragments; because keratin is strongly antigenic and irritating, it attracts many inflammatory cells, but as keratinocytes are very large, normal macrophages are not able to eliminate them. For this reason, macrophages tend to transform into so-called epithelioid macrophages, larger cells with wide cytoplasm that resemble epithelial cells (hence the term “epithelioid”) and also give origin to histiocytic multinucleate giant cells (Gross et al. 2005).
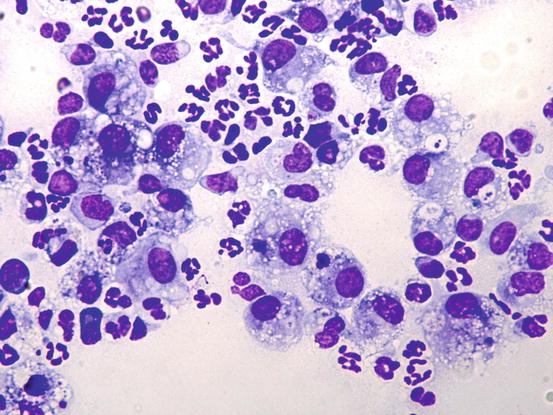

Fig. 3.20
Cytology of deep pyoderma: segmented neutrophils and many vacuolated macrophages showing leukophagocytosis
On cytological specimens, both cells are variably numerous and often arranged around single or rafts of corneocytes (Figs. 3.21, 3.22, and 3.23).
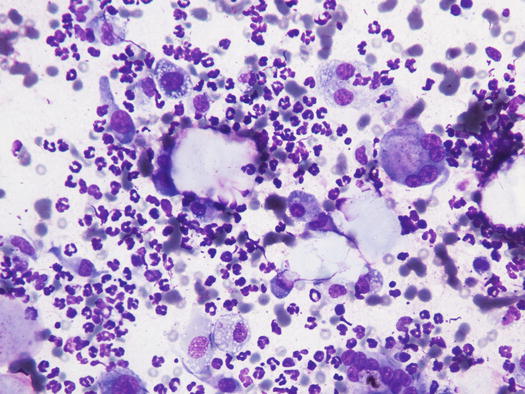
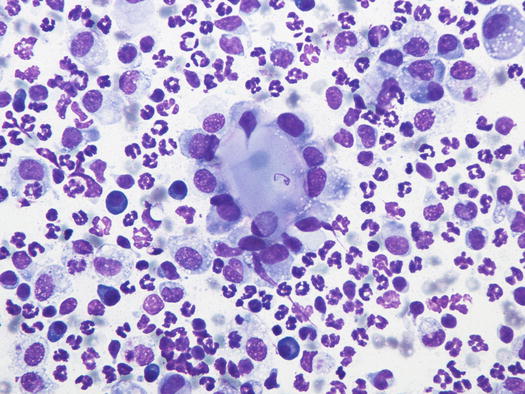
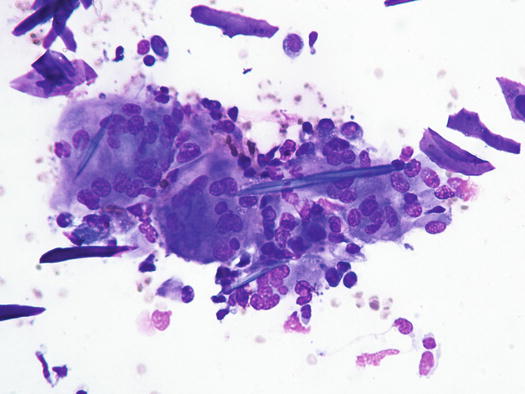

Fig. 3.21
Cytology of deep pyoderma: neutrophils, macrophages and multinucleated giant cells attacking corneocytes

Fig. 3.22
Cytology of deep pyoderma: epithelioid macrophages surrounding a group of corneocytes

Fig. 3.23
Cytology of deep pyoderma: many giant cells are phagocytosing keratin fragments
As the keratin acts as a true foreign body, the secondary impressive pyogranulomatous reaction observed in furunculosis is the reason why, in deep pyoderma, bacteria are not easily observed (Fig. 3.24). This cytological aspect is very important in canine skin cytology, because the failure to see bacteria in samples from deep pyogranulomatous lesions does not authorise us to rule out a pyoderma. In chronic lesions, cytological samples enriched with lymphocytes and plasma cells indicate continuous antigenic stimulation.
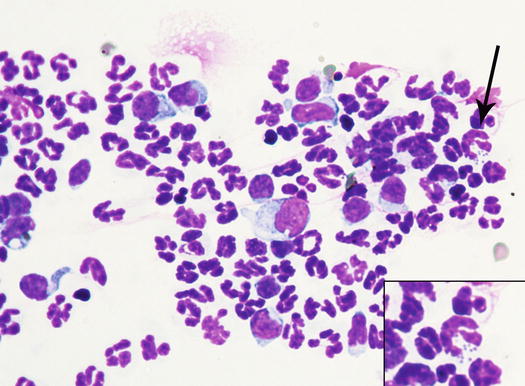

Fig. 3.24
Cytology of deep pyoderma: few macrophages and many karyolytic neutrophils with intracytoplasmic cocci (arrow inset)
Similar clinical, cytological and histopathological findings are observed in dogs with furunculosis due to demodicosis (Figs. 3.25 and 3.26) (Gross et al. 2005; Mueller 2004; Mueller et al. 2012; Miller et al. 2013). The pathogenetic mechanism underlying the furunculosis is the same as deep pyoderma and although cytological examination is not the best technique for diagnosing demodicosis, it is sometimes possible, from FNB performed for papular–nodular lesions, to detect Demodex canis mites or their eggs, immersed in pyogranulomatous inflammation (Figs. 3.27, 3.28, and 3.29).
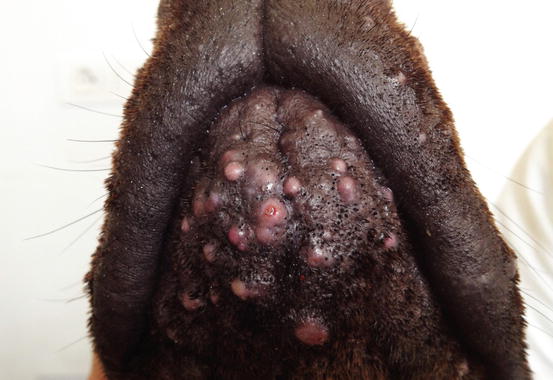
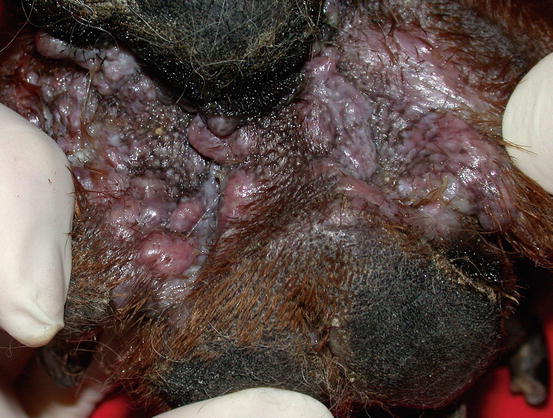
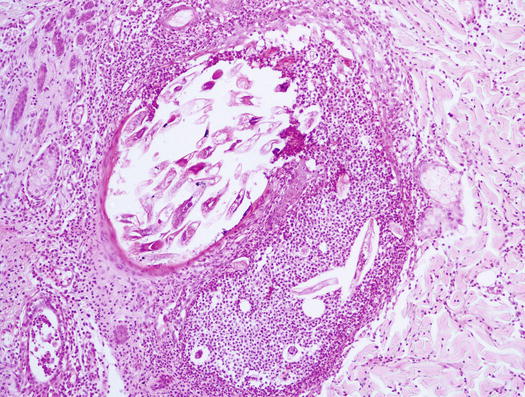
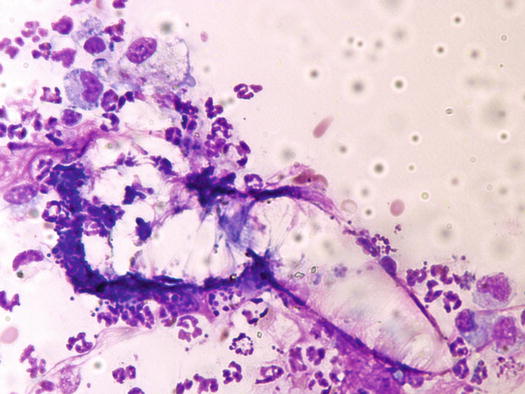
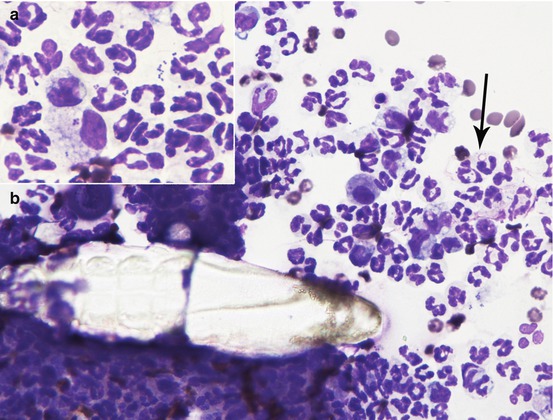

Fig. 3.25
Papular–nodular lesions with follicular plugs and comedones on the muzzle of a Dobermann with demodicosis

Fig. 3.26
Multiple confluent nodular papules on the interdigital area of a dog with demodicosis

Fig. 3.27
Histopathology of demodicosis: many Demodex canis immersed in a pyogranulomatous exudate due to the rupture of an infested follicle

Fig. 3.28
Cytology of demodicosis: an adult Demodex surrounded by neutrophils and macrophages

Fig. 3.29
Cytology of demodicosis: a Demodex canis mite surrounded by macrophages and neutrophils, the latters with many intracytoplasmic cocci (arrow);cocci are well recognizable into the cytoplasm of neutrophils and into a macrophage (inset)
3.3.2 Facial Eosinophilic Furunculosis
Eosinophilic furunculosis is a disease of dogs, characterised by the acute onset of papular–nodular lesions, mostly located on the face and less frequently on other parts of the body such as the pinna, neck, abdomen and limbs. Early lesions are represented by nodular papules, mainly spread over the bridge of the nose and muzzle (Fig. 3.30) (Curtis et al. 1995; Gross 1993; Guaguère et al. 1996). In a few hours, nodular papules tend to grow and merge with each other, giving rise to plaques and nodules, which tend to ulcerate and become infected (Fig. 3.31). Eosinophilic furunculosis is usually very pruritic and this is the reason why self-trauma can rapidly change the initial papular–nodular appearance (Fig. 3.32).
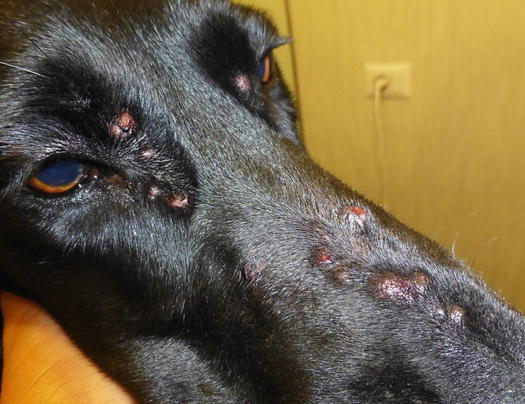
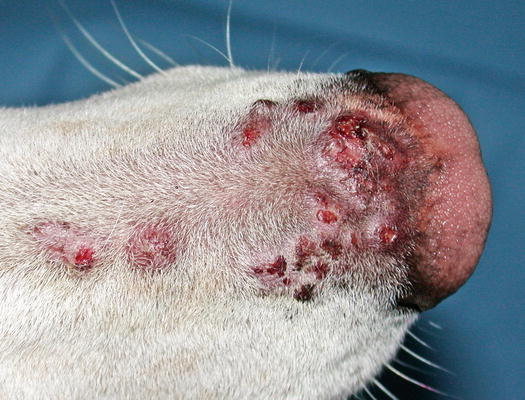
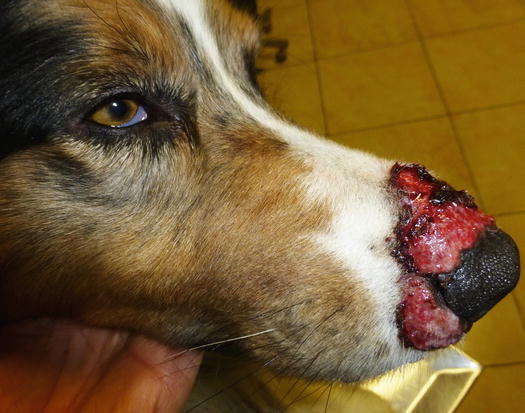

Fig. 3.30
Many small erythematous nodular papules on the bridge of the nose and eyelids of a dog with eosinophilic furunculosis

Fig. 3.31
Single and confluent papular nodular lesions on the nose of a dog with eosinophilic furunculosis

Fig. 3.32
Ulcerative lesions secondary to self-trauma in an Australian shepherd with eosinophilic furunculosis
Cytological Findings
Although histologically the target of the inflammatory process are the follicles that cause furunculosis, the number of giant cells observed in cytological specimens is very low compared with what is usually see in furunculosis from deep pyoderma (Fig. 3.33). The specimens of early and non-infected lesions are therefore characterised by a high number of eosinophils, macrophages and a variable number of neutrophils (Gross et al. 2005). Epithelioid macrophages and multinucleated cells are present in variable amounts (Figs. 3.34 and 3.35).
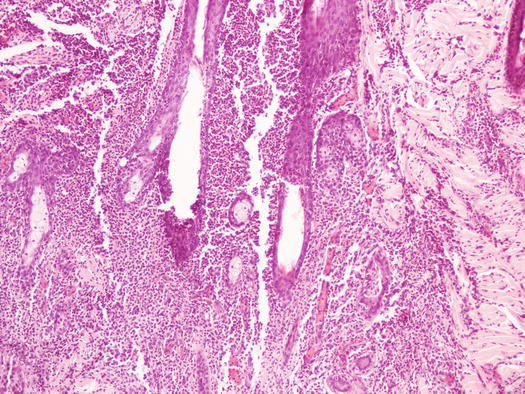
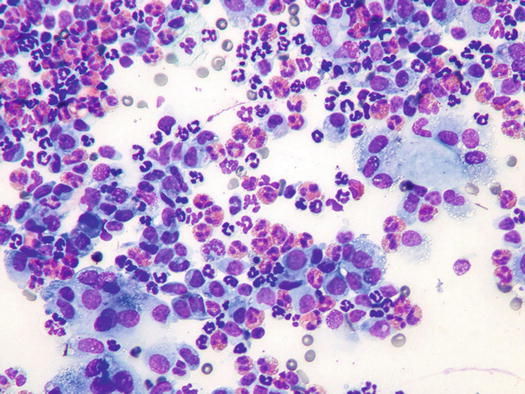
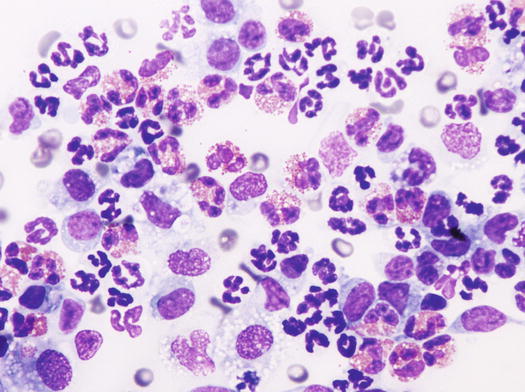

Fig. 3.33
Histopathology of eosinophilic furunculosis: note that the follicles are the target of the eosinophilic inflammation

Fig. 3.34
Cytology of eosinophilic furunculosis: many eosinophils and macrophages. Note that the corneocytes are attacked by epithelioid macrophages

Fig. 3.35
Cytology of eosinophilic furunculosis: neutrophils, macrophages and many eosinophils
Karyolytic neutrophils with cocci, intermingled with eosinophils, are only observed in secondarily infected lesions. A low number of lymphocytes and plasma cells completes the cytological findings.
3.3.3 Papular–Nodular Canine Leishmaniasis
Canine leishmaniasis is a protozoan disease characterised by a wide clinical polymorphism, depending on the type of immune response developed by affected dogs. In the clinical classification of canine leishmaniasis, a form characterised only by papular–nodular lesions, mainly located on hairless areas of the face, the bridge of the nose, eyelids, the inner surface of the pinna, and less frequently on the hairless areas of the abdomen and thighs, has been reported (Ordeix et al. 2005; Bottero et al. 2006; Solano-Gallego et al. 2009, 2011; Lombardo et al. 2014). Lesions are alopecic, slightly erythematous and mostly characterised by an umbilicated shape with a central depression (Figs. 3.36, 3.37, and 3.38). Usually, these types of lesions are observed in young dogs with no other dermatological signs and systemic symptoms, with normal blood values and negative or very low anti-leishmanial antibody titres. Some authors speculate that such lesions might represent a local reaction to phlebotomus bites in immunocompetent animals (Ordeix et al. 2005).
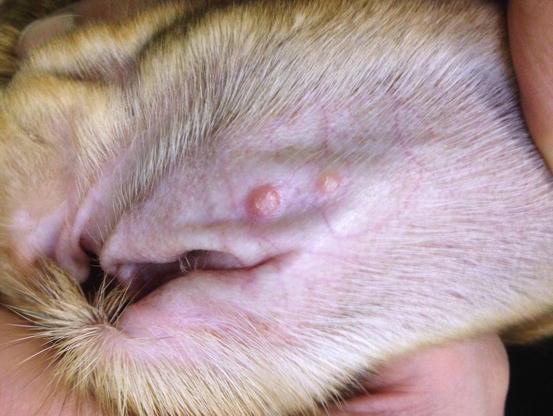
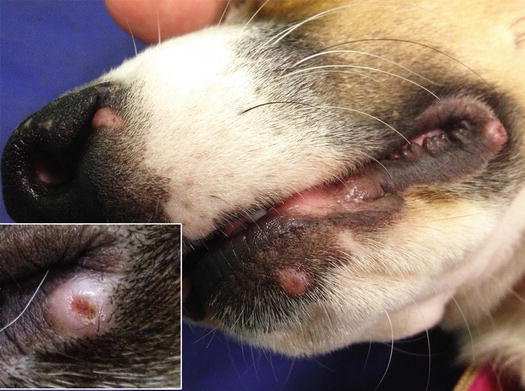
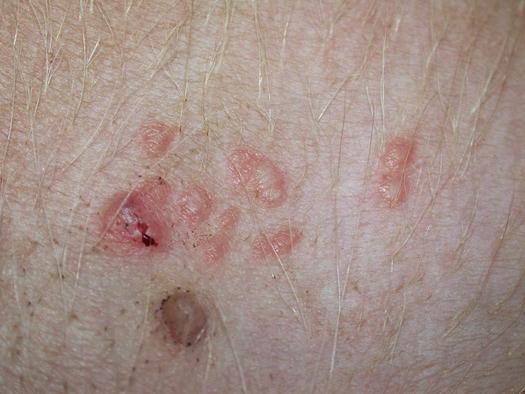

Fig. 3.36
Two nodular papules on the inner surface of the pinna of a leishmaniotic dog

Fig. 3.37
Multiple papular–nodular lesions on the nose and lips of a young dog with canine leishmaniosis. Inset: typical umbilicated shape of a nodular papule

Fig. 3.38
Multiple nodular papules on the abdomen of a Dogue de Bordeaux with papular leishmaniasis
Cytological Findings
The Leishmania infantum amastigotes are recognisable by their characteristic oval shape, measuring approximately 1–2 × 2–5 μm, containing a rod-shaped hyperchromatic kinetoplast, from which the flagellum originates in the promastigote stage. Morphology of Leishmania spp. is easy to recognise when the kinetoplast is perpendicular to the nucleus, giving a T-shaped appearance (Fig. 3.39). Histopathological examinations of skin biopsies reveal a nodular or diffuse lympho-histio-plasmacellular dermatitis (Fig. 3.40) (Gross et al. 2005). Mixed inflammatory cells characterise slides, together with a variable number of amastigotes in the cytoplasm of macrophages (Fig. 3.41). In literature, there are discrepancies about the number of amastigotes that can be detected in cytological specimens from this lesions; some articles report a low number of parasites (Ordeix et al. 2005), whereas in other case reports, the number of amastigotes detected was consistently high (Bottero et al. 2006; Noli and Cornegliani 2006). Anecdotally, according to some of these authors, the number of amastigotes may be related to the chronicity of the lesions. Cytological specimens are mainly composed of macrophages and many lymphocytes and plasma cells, whereas the number of amastigotes can be so low that specimens must be carefully observed to detect few protozoa (Fig. 3.42). Sometimes, as reported, immunohistochemical staining is needed to confirm the presence of the parasites (Ordeix et al. 2005).
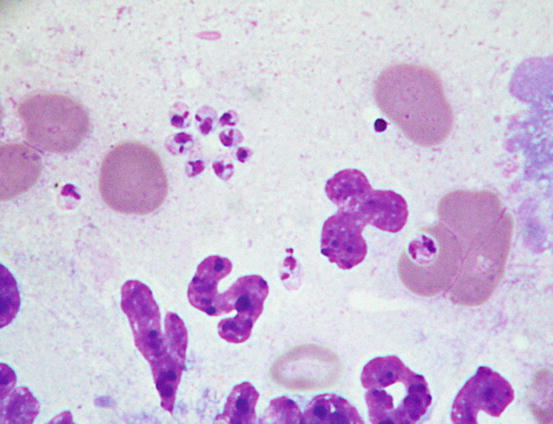
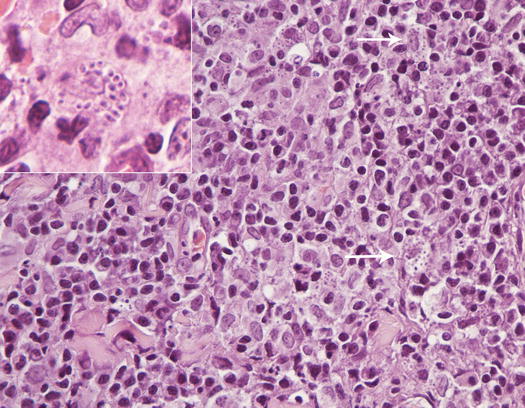
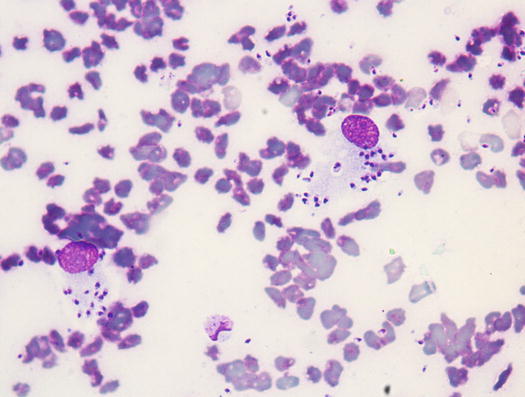
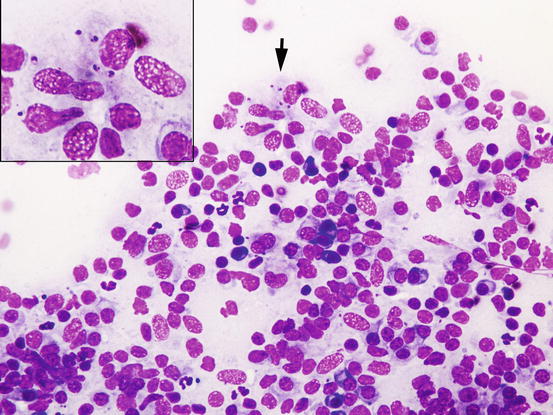

Fig. 3.39
Cytology of leishmaniasis: amastigotes of Leishmania spp. in which kinetoplast is easily recognisable as a rod-shaped structure

Fig. 3.40
Histopathology of leishmaniasis: diffuse dermatitis composed mostly of macrophages, lymphocytes and plasma cells. Many macrophages are filled with matigotes (arrow, inset)

Fig. 3.41
Cytology of leishmaniasis: many amastigotes, both free and in the cytoplasm of macrophages

Fig. 3.42
Cytology leishmaniasis: many macrophages and plasma cells; few amastigotes are evident in the cytoplasm of macrophages (arrow, inset). Note that neutrophils are completely absent
3.3.4 Sterile Granuloma and Pyogranuloma Syndrome
Although sterile granuloma and pyogranuloma syndrome (SGPS) is commonly included under nodular dermatitis, because in the early phases the lesions appear as nodular papules, it is discussed in this chapter. SGPS is rare in dogs and extremely rare in cats. In dogs, lesions are mainly located on the face, eyelids, nose, bridge of the nose and pinna, and with regard to clinical aspect, size and location, they are very similar to those observed in papular–nodular leishmaniasis from which it must be always differentiated. In some dogs, the lesions may coalesce to form plaques or primarily arise as true nodules, which can be observed in different areas of the body (Figs. 3.43, 3.44, 3.45, and 3.46) (Scott et al. 1990; Panich et al. 1991; Miller et al. 2013).

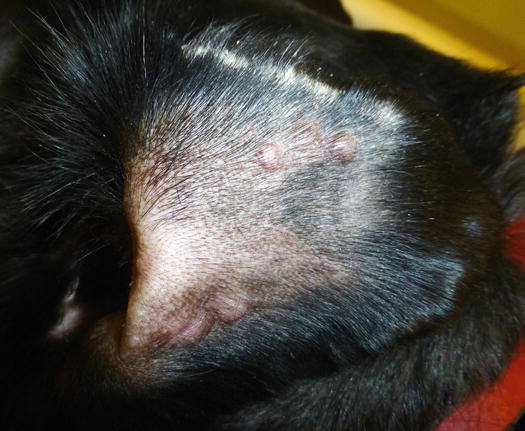
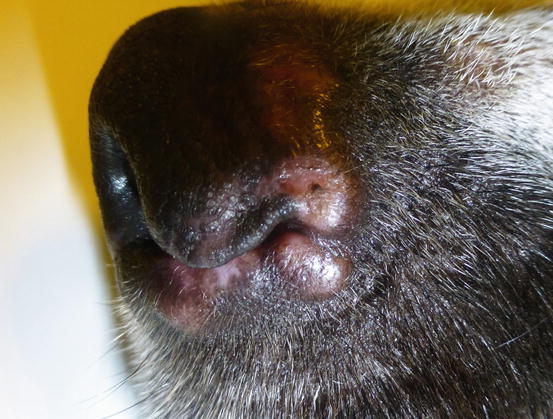
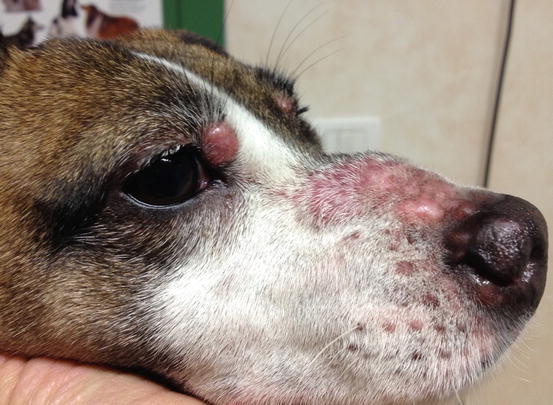

Fig. 3.43
Papular nodules on the eyelids of a dog with sterile granuloma and pyogranuloma syndrome (SGPS)

Fig. 3.44
Papular nodules on the inner surface of the pinna

Fig. 3.45
Papular nodules on the mucocutaneous junction of the left nostril

Fig. 3.46
Confluent papular nodules, which form plaques on the bridge of the nose of a Jack Russell terrier with SGPS
The SGPS is a sterile idiopathic disease and, although some authors have suggested that an infectious antigen can cause an immunity dysfunction that results in an aberrant histiocytic immune response, its real pathogenesis is still unknown. The histological features and the good response to immunomodulatory drugs suggest the sterile nature of the lesions (Scott et al. 1990; Gross et al. 2005; Miller et al. 2013). In a study of 46 dogs with a histological diagnosis of SGPS, 21 were positive for Leishmania according to polymer chain reaction (PCR) and immunohistochemical staining (Cornegliani et al. 2005). The meaning of this positivity needs to be clarified because the detection of DNA fragments of amastigotes in the skin does not necessarily imply that they play a role in the development of the lesions.
Regarding SGPS, the term sterile should only be attributed to cases where granulomatous/pyogranulomatous lesions with typical clinical and histopathological findings and those in which any microorganisms have been excluded by special stains, PCR, immunohistochemistry and bacterial culture.
Further studies are needed to determine the true sterile nature of the lesions included in the SGPS group or if the possibility of a triggering stimulus operated by infectious agents really exists.
Cytological Findings
A typical histopathological feature of SGPS is perifollicular granulomas with a characteristic elongated or sausage-like shape (Figs. 3.47 and 3.48).
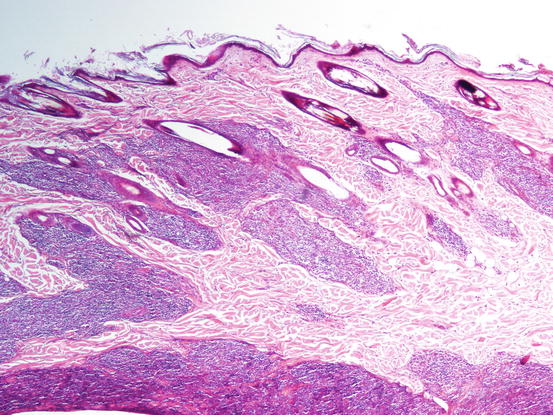
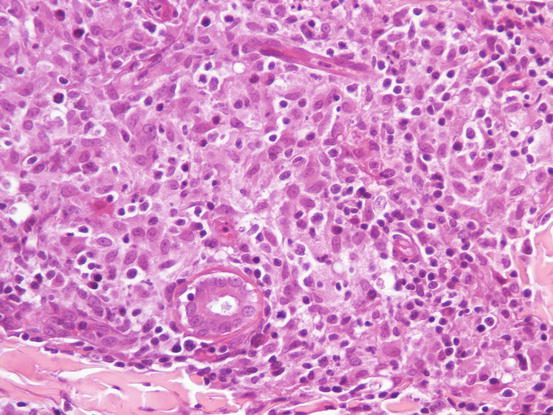

Fig. 3.47
Histopathology of SGPS: multiple elongated perifollicular nodules

Fig. 3.48
Histopathology of SGPS: at high magnifications it is possible recognise many histiocytes and lymphocytes
In the early phase of the disease, as granulomas are small and scattered between follicles, it is not always easy to collect a large number of cells; indeed, in these cases, it is very difficult to centre the small granulomas with the needle. However, when granulomas merge and transform from nodular to diffuse dermatitis, it is easier to obtain more cellular samples. When this situation occurs, the typical histological aspect of SGPS is lost. Despite the term pyogranuloma, the number of neutrophils is usually poor and, when present, they are clearly segmented and located in the centre of the granulomas. Slides are therefore usually characterized by a high number of poorly vacuolated macrophages and epithelioid macrophages, the latter with oval to kidney-shaped nuclei and moderate to large, slightly grey–blue cytoplasm. Many histiocytes are represented by bare nuclei, associated with a variable number of lymphocytes and a few plasma cells. The main clinical–cytological differential is with the papular–nodular form of canine leishmaniasis. In specimens in which amastigotes are not detected, it is in fact impossible to cytologically differentiate SGPS from canine leishmaniasis. The main cytological difference between these two diseases is probably the higher number of plasma cells observed in the latter (Figs. 3.49 and 3.50).
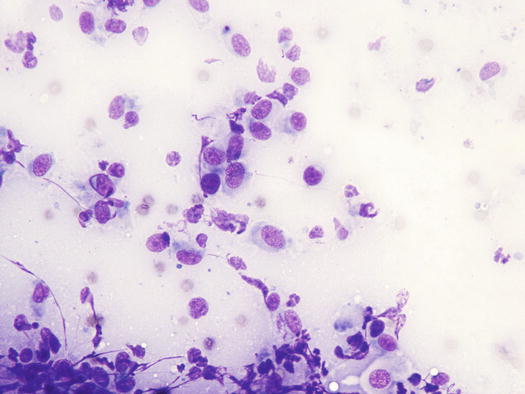
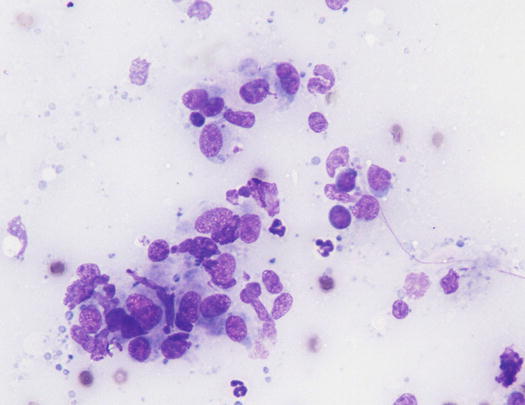

Fig. 3.49
Cytology of SGPS: histiocytes and rare lymphocytes

Fig. 3.50
Cytology of SGPS: histiocytes and rare lymphocytes. Note the presence of lympho-glandular bodies indicative of the rupture of many lymphocytes
3.3.5 Calcinosis Cutis
Skin tissue mineralisation can occur through different pathogenetic mechanisms: dystrophic, metastatic, idiopathic and iatrogenic. Calcinosis cutis is a skin disease characterised by widespread mineralisation of the dermis (collagen fibres) and rarely of the epidermidis. The mineral salts deposited in the skin, are insoluble inorganic compounds mainly composed of a combination of calcium, phosphate and carbonate salts. Because the former are the most common, the term calcinosis is commonly used to describe the presence of mineral salts, even if their composition has not been investigated (Gross et al. 2005; Doerr et al. 2013).
In dogs, the most frequent cause of skin calcium salt deposits is spontaneous or iatrogenic hypercortisolism (Frazier et al. 1998). The reason why, in this disease, calcium salts are deposited in the dermis is not clear and the definition of dystrophic calcinosis used by many authors to describe calcium salt deposits in the course of hypercortisolism could be not correct at the time; the term dystrophic should in fact be associated with cases where structural damage to the tissue, which may lead to intra- or extracellular deposits of minerals, has been identified.
Clinical lesions are vary greatly, ranging from small papular–nodular lesions, often alopecic, erythematous and confluent, to form different sized plaques, often linear or serpiginous in shape (Figs. 3.51, 3.52, and 3.53) (White et al. 2013; Miller et al. 2013). Papular and nodular lesions are better visible in the hairless areas of the axilla, groin and abdomen. As the host tries to eliminate calcium salts through the epidermidis, lesions are often ulcerated and covered with crusts and pus as a result of a secondary bacterial infection (Fig. 3.54).
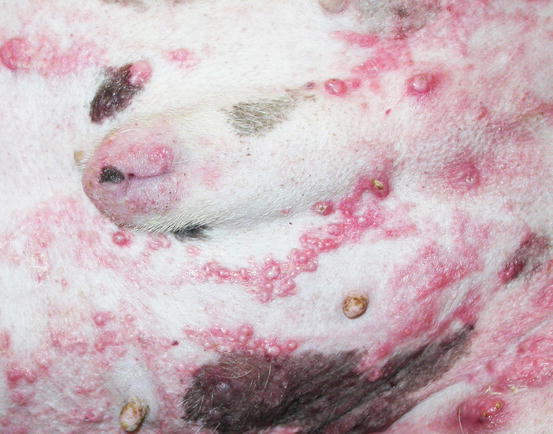
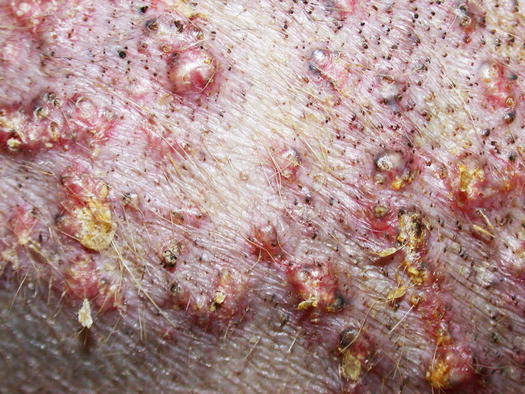
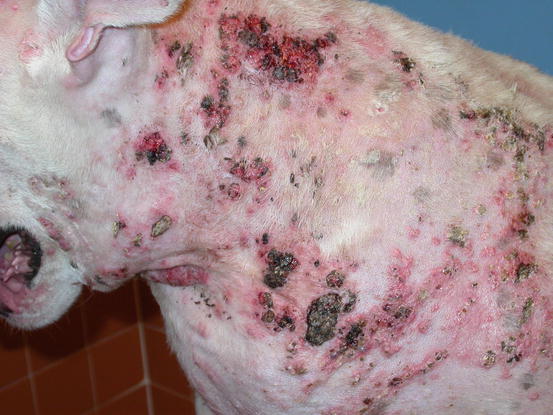

Fig. 3.51
Multiple erythematous nodular papules on the abdomen of a dog with calcinosis cutis secondary to iatrogenic hypercortisolism

Fig. 3.53
Calcinosis cutis: multiple nodular papules, some of them confluent, which form small plaques in a dog with spontaneous pituitary-dependent hyperadrenocorticism. Note the presence of comedones

Fig. 3.54
Calcinosis cutis: multiple nodular papules, plaques and haematic crusts in a bull terrier with iatrogenic hypercortisolism
Cytological Findings
Cytological specimens are characterised by very different aspects, which differ based on the inflammatory reaction developed around the mineral salts and by the potential for secondary bacterial infections. In cases of lesions with an intact epidermidis, histopathology is characterised by multifocal deposits of mineral salts surrounded by a variable inflammatory response, usually composed of epithelioid macrophages and multinucleated giant cells (Figs. 3.55 and 3.56).
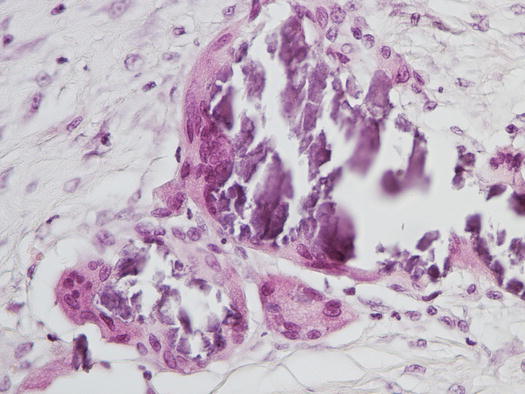
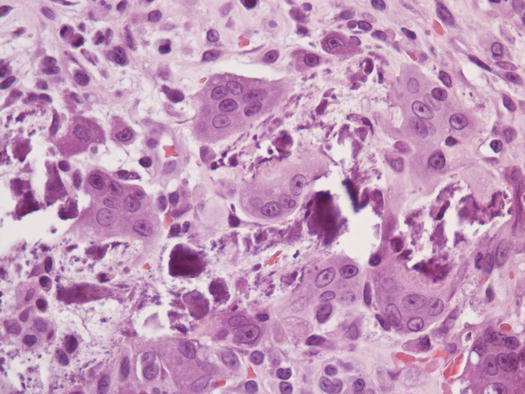

Fig. 3.55
Histopathology of calcinosis cutis: multifocal mineralisation of the dermis

Fig. 3.56
Histopathology of calcinosis cutis: multinucleated giant cells and reactive fibroblasts surrounding mineral salts
Cytology shows the same findings as those observed in histopathology, with many giant cells surrounding the calcium salts. In many cases, a severe fibroblastic reaction, cytologically represented by many spindle cells with some atypia, is detected. To an inexpert cytologist, especially when mineral salts are not evident, this cytological appearance could be confused with an anaplastic soft tissue sarcoma with giant cells; fortunately, the clinical aspects of calcinosis cannot be confused with a malignant neoplasia (Figs. 3.57, 3.58, and 3.59).
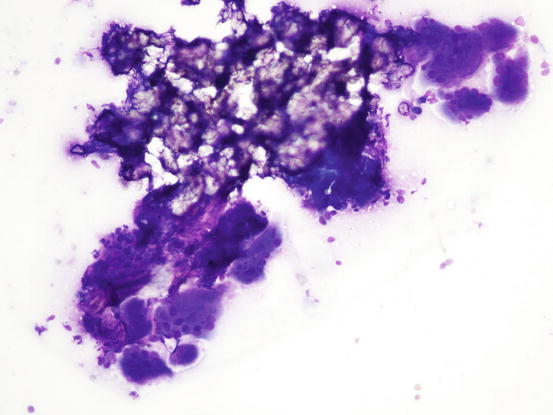
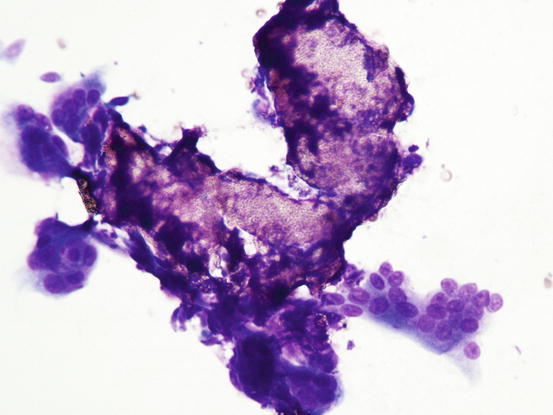
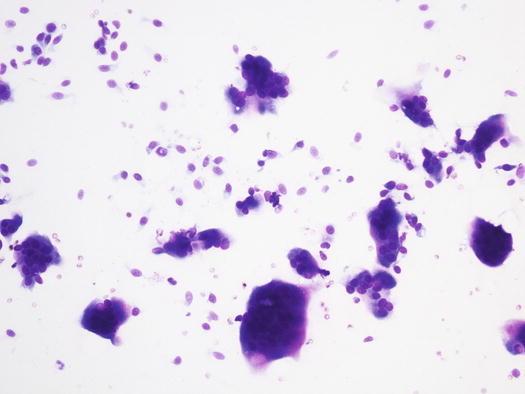

Fig. 3.57
Cytology of calcinosis cutis: many multinucleated giant cells surrounding mineral salts

Fig. 3.58
Cytology of calcinosis cutis: many giant cells attacking calcium salts

Fig. 3.59
Cytology of calcinosis cutis: many spindle fibroblasts and giant cells. Mineral salts are not present on this image
When the lesions ulcerate, many neutrophils infiltrate the dermis. In these cases, slides show neutrophilic or pyogranulomatous inflammation (Fig. 3.60).
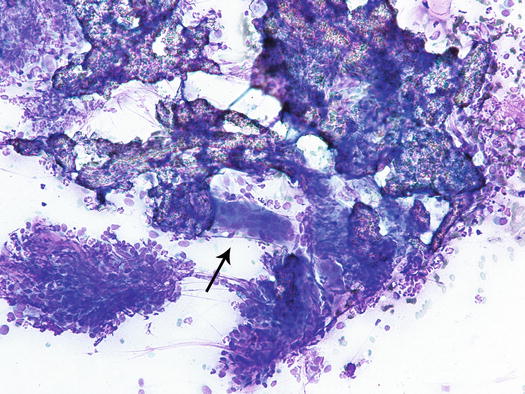

Fig. 3.60
Cytology of calcinosis cutis: inflammatory pyogranulomatous inflammation surrounding mineral salts. Note the giant cell (arrow)
In cytology, the morphology of calcium salts can be very different, ranging from basophilic pointed or coarse granulations to more typical formations with an amorphous, glassy, transparent and sometimes rhomboidal or needle-like appearance.
In some cases, minerals salts may hardly be detectable in hypercellular inflammatory specimens; in such cases, to better demonstrate the calcium salts, it is possible to stain some slides with Von Kossa, which colours in black the area were calcium salts were present.
When a needle is introduced into a mineralised lesion, it is possible to perceive a crunching sensation between the fingers, which helps to support the suspicion of the presence of mineral salts in the tissue. In chronic cases, calcium salts can transform into bone tissue (osseous metaplasia) and lesions turn from calcinosis cutis to osteoma cutis; in these cases, the suspicion may be generated when even large needles cannot penetrate the firm nodular papules (Frazier et al. 1998). Histology is the only examination that allows the diagnosis of osteoma cutis, as the clinical lesions cannot be differentiated from calcinosis cutis (Fig. 3.61).
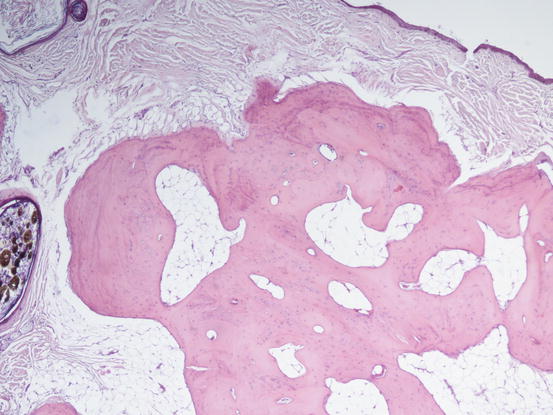

Fig. 3.61
Histopathology of osteoma cutis: osseous metaplasia secondary to chronic deposit of calcium salts in the dermis
3.3.6 Xanthomatosis
Cutaneous xanthomatosis is a rare skin disease of cats and dogs in which cholesterol, triglycerides and phospholipids accumulate in the dermis. In cats, xanthomas are mostly due to diabetes mellitus, both spontaneous and iatrogenic, the latter usually secondary to chronic corticosteroid therapy administered to manage pruritic diseases (especially allergic dermatitis). Less commonly, feline xanthomatosis develops in cats fed with a high-fat diet, in young patients suffering from idiopathic hyperlipidaemia or in patients with pancreatitis (Vitale et al. 1998; Chanut et al. 2005).
In dogs, xanthomatosis can be idiopathic or, as in cats, secondary to an excessively fatty diet or to diabetes mellitus (Banajee et al. 2011). In any case, skin lesions are represented by papular nodules and little plaque and nodules, usually multiple and located in the dermis, alopecic and white–yellow in colour, from which greasy material is drained (Figs. 3.62, 3.63, 3.64, and 3.65) (Gross et al. 2005). Single nodular lesions are rarely reported both in normolipaemic dog and cats (Ravens et al. 2013).
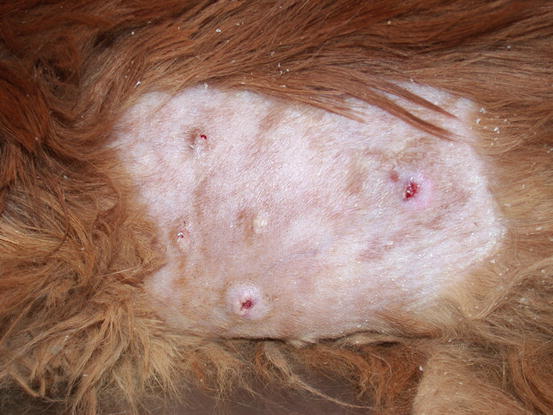
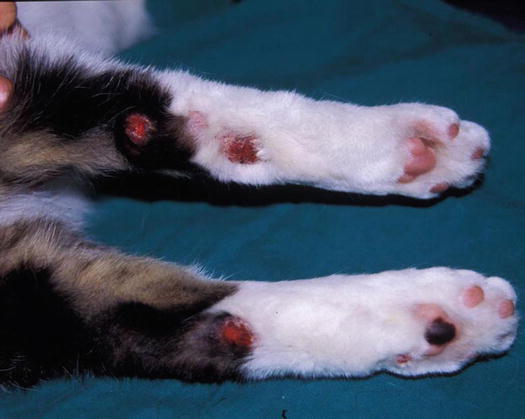
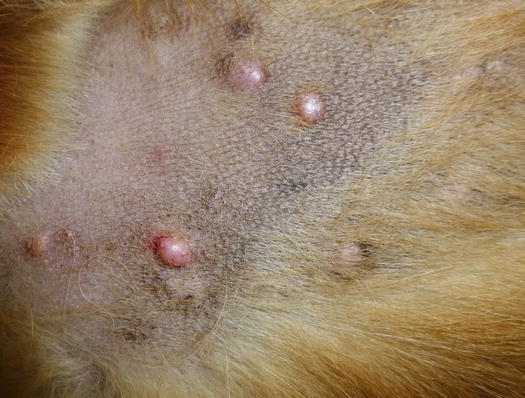

Fig. 3.62
Multiple ulcerated xanthomas on the chest of a cat with diabetes mellitus

Fig. 3.64
Multiple and ulcerated nodules on the rear legs of a kitten affected by idiopathic hyperlipaemia (Courtesy of Dr. C. Noli, Italy)

Fig. 3.65
Multiple papular nodules on the chest of a dog with idiopathic xanthomatosis
Cytological Findings
Cytologically, xanthomatous nodules are composed almost exclusively of histiocytic cells represented by large macrophages with cytoplasm rich in vacuoles, containing lipids that give the cell a characteristic foamy appearance (Figs. 3.66, 3.67, and 3.68). Neutrophils are few or absent, whereas numerous giant cells, with haphazardly distributed nuclei, are commonly observed. The multinucleated cells with nuclei arranged in a crown shape, similar to the Touton giant cells reported in human xanthomatosis, are rarely observed in our pets. Some macrophages and giant cells can show a central homogeneous and eosinophilic cytoplasmic area, representing a pre-xanthomisation stage (Fig. 3.69). To better show the lipid content of the vacuoles, a oil-red-O stain can be performed; lipids stain bright red (Fig. 3.70).
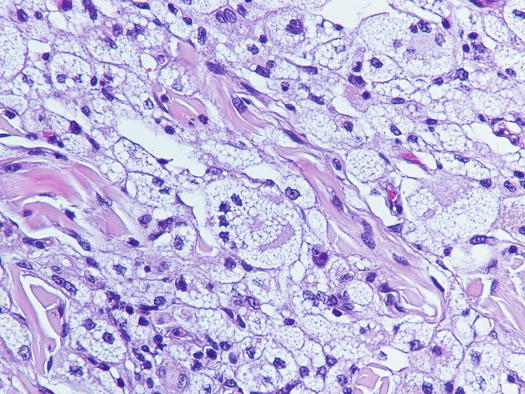
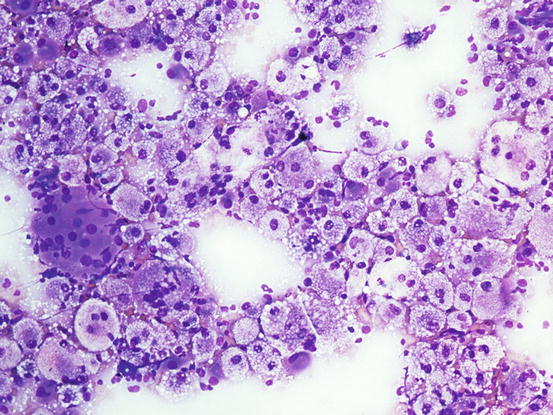
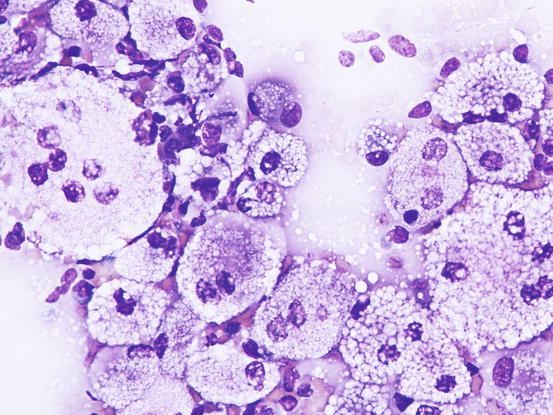
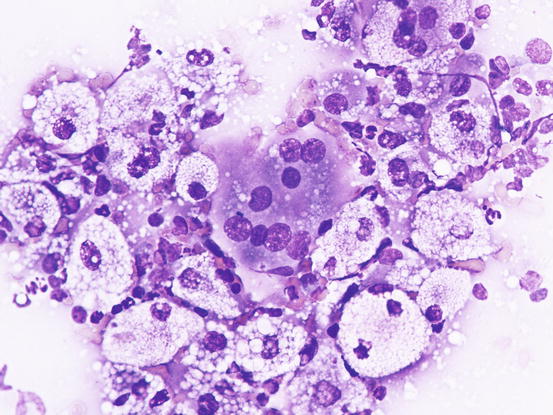
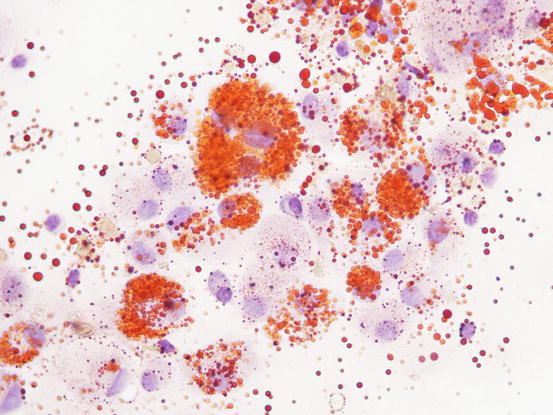

Fig. 3.66
Histopathology of xanthomatosis: many foamy macrophages and giant cells infiltrating the dermis

Fig. 3.67
Cytology of xanthomatosis: many foamy macrophages and giant cells

Fig. 3.68
Cytology of xanthomatosis: at high magnifications the foamy macrophages and giant cells are more evident

Fig. 3.69
Cytology of xanthomatosis: note the incompletely xanthomised giant cell

Fig. 3.70
Cytology of xanthomatosis: the lipid content of vacuoles is stained red with oil-red-O stain
3.4 Pustules
Specimens collected from pustules are always very useful and many often provide a diagnosis.
The gross recognition of follicular and non-follicular pustules is very useful in interpreting the origin of the lesions.
3.4.1 Pustular Diseases in Dogs
3.4.1.1 Diseases Characterised by Follicular Pustules
Follicular pustules indicate an infection localised in the follicular lumen and, when observed, an intrafollicular disease such as bacterial folliculitis, demodicosis or, exceptionally, dermatophytic folliculitis, must be suspected.
Bacterial Folliculitis
The most common cause of follicular pustules in dogs is bacterial folliculitis. Pyoderma is a very common disease in dogs and the predisposing causes are numerous and of different origin. Microorganisms, parasites, allergic diseases and endocrine disorders can frequently cause an injury to the skin barrier, allowing bacteria such as Staphylococcus pseudintermedius to cause infection (Miller et al. 2013).
Clinically, pustules are mainly follicular, but because they are delicate lesions, they rapidly dehydrate and form crusts and epidermal collarettes (Fig. 3.71) (Irhke 1996; Gross et al. 2005; Miller et al. 2013).
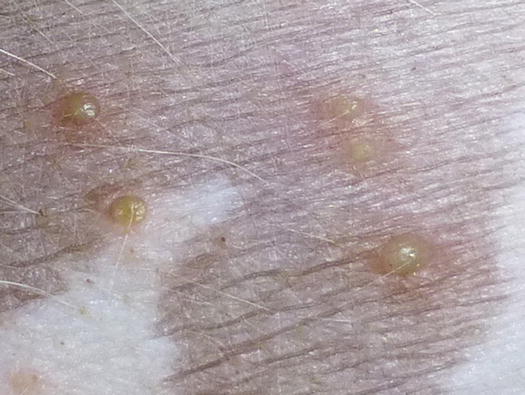

Fig. 3.71
Multiple follicular pustules: note that a hair emerges from the centre of the pustules
Cytological Findings
Slides from an intact pustule contain a large number of neutrophils characterised by nuclear degeneration represented by swollen and hypochromatic nuclei with a loss of the classical multilobed silhouette, due to severe cell membrane damage that permits water to penetrate into the cell. As mentioned in Chap. 1, this very common type of nuclear degeneration is known as karyolysis.
In the cytoplasm of neutrophils many coccus bacteria are usually present, arranged singly, in pairs or in tetrads. Many bacteria are also extracellular, but this is linked to the rupture of severely degenerated neutrophils that inevitably occurs during the preparation of the slides (Fig. 3.72).
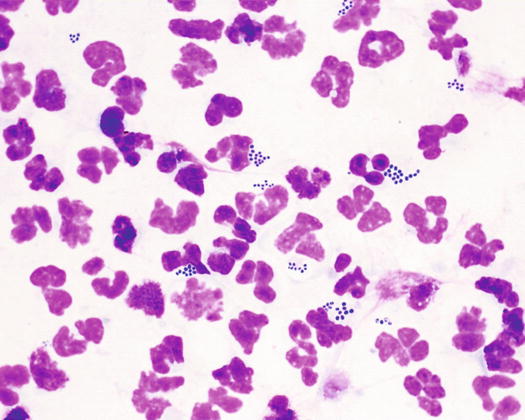

Fig. 3.72
Cytology of pyoderma: karyolytic neutrophils with many intracytoplasmic cocci
Karyolytic neutrophils are very delicate and once broken give rise to streaks of nuclear material. Together with neutrophils, it is common to find a small number of corneocytes, which are the cells comprising the roof of the pustule that exfoliate into pus. Sometimes, in specimens sampled from bacterial pustules, it is possible to detect a few nucleated keratinocytes of the spinous or granular layers (acantholytic cells); these epithelial cells separate from other keratinocytes because of the proteolytic enzymes released by bacteria (acantholysis). Rarely, in superficial pyoderma, there is a very high number of acantholytic keratinocytes (AKs) (Fig. 3.73).
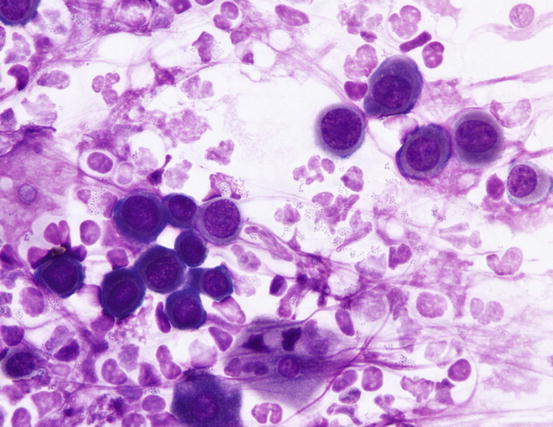

Fig. 3.73
Cytology of pyoderma: severe karyolysis of neutrophils with many cocci and acantholytic keratinocytes
Demodicosis
In dogs with demodicosis and secondary pyoderma, pustules are very common lesions and are usually associated with comedones, which contribute to the suspicion that the problem involves the follicles (Figs. 3.74 and 3.75) (Gross et al. 2005; Mueller et al. 2011; Miller et al. 2013).
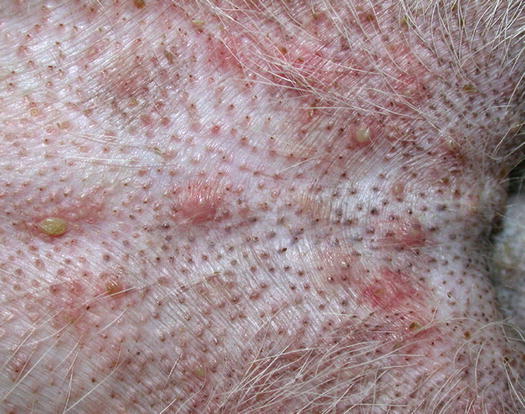
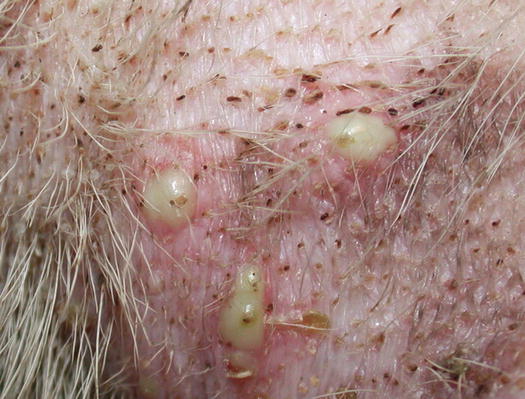

Fig. 3.74
Follicular pustules and comedones in a puppy with demodicosis

Fig. 3.75
Follicular pustules: note that a hair emerges from the centre of the pustule
Cytological Findings
Cytology specimens from pustules are similar to those described in pyoderma, because although the cause of the lesions is Demodex mites, pustules are caused by secondary bacterial intrafollicular infection (Figs. 3.76 and 3.77). In addition to karyolytic neutrophils that phagocytose cocci, a variable number of adult and immature mites and their eggs are usually observed (Figs. 3.78 and 3.79). As mentioned for pyoderma, it is also possible to observe a variable number of acantholytic cells.
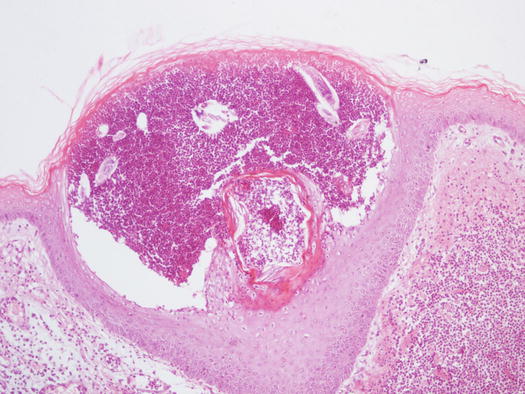
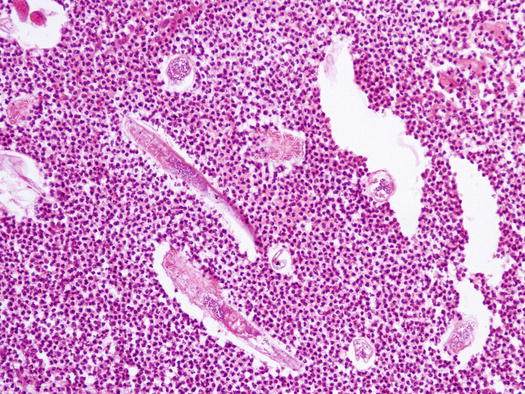
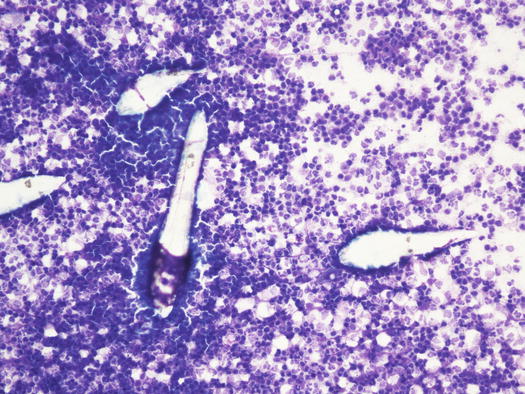
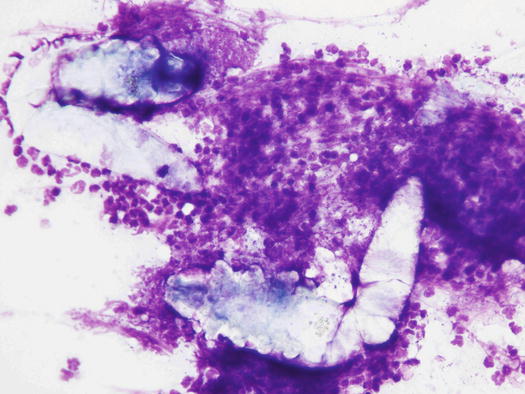

Fig. 3.76
Histology of pustular demodicosis: follicular pustule containing many Demodex mites

Fig. 3.77
Histology of pustular demodicosis: at high magnifications mites are clearly recognisable

Fig. 3.78
Cytology of pustular demodicosis: many Demodex canis immersed in a neutrophilic exudate

Fig. 3.79
Cytology of pustular demodicosis: two adult Demodex canis and karyolytic neutrophils, which phagocytose many cocci
Pustular Dermatophytosis
In dogs with dermatophytosis intrafollicularly infected hair shafts is a common finding; nevertheless, it is less frequent for fungi to attract neutrophils in the follicular lumen in such quantities as to be grossly evident as follicular pustules.
3.4.1.2 Diseases Characterised by Non-follicular Pustules
Unlike from follicular pustules that are almost exclusively linked to bacterial infections, numerous infectious and sterile diseases cause non-follicular pustules. As mentioned, non-follicular pustules may vary in size according to their cause and extension into the epidermal layers.
Pustular Non-follicular Staphylococcal Infection (Impetigo)
In superficial pyoderma it is possible to observe intra-epidermal subcorneal pustules that are not localised in the follicular lumen (Fig. 3.80) (Gross et al. 2005). Impetigo is a form of surface pyoderma mostly reported in puppies and young dogs, mainly hunting breeds, clinically characterised by small to medium-sized non-follicular pustules, localised on the abdomen, groin and axilla (Ihrke 1996). Pustules are of varying shapes, from round to irregular and with a tendency to merge (Figs. 3.81 and 3.82). The causes of bacterial infection are not always ascertained, but, because the lesions are observed in young animals and do not recur once recovered, stressful events or local trauma associated with poor hygiene have been speculated to be possible causes. Rarely, an infectious predisposing disease such as in puppies suffering from distemper can cause impetigo. In adults or old dogs affected by several immunosuppressive systemic diseases such as spontaneous or iatrogenic hypercortisolism, it is common to observe large and often coalescent non-follicular pustules. Because of their large size, this form of pyoderma is named bullous impetigo (Figs. 3.83 and 3.84). In this pustular pyoderma, exotoxins produced by Staphylococcus pseudintermedius bind to the extracellular portion of the desmoglein-1 (Dsg-1) and, in association with the development of proteolytic enzymes, are the cause of the keratinocytes splitting and of the consequent development of pustules (Terauchi et al. 2003; Nishifuji et al. 2008; Amagai 2009).
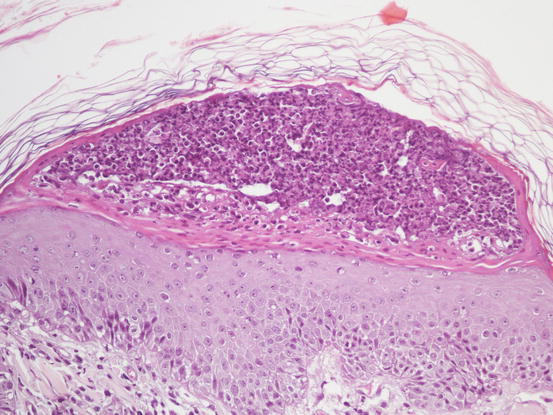
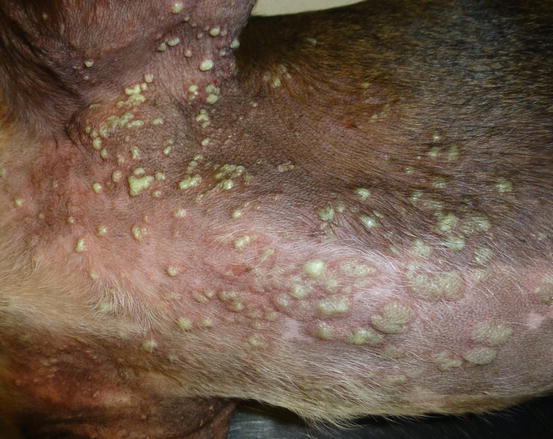
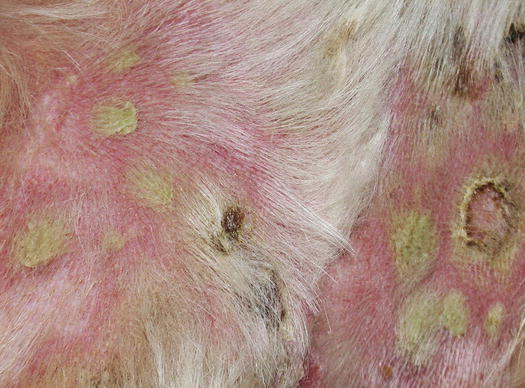
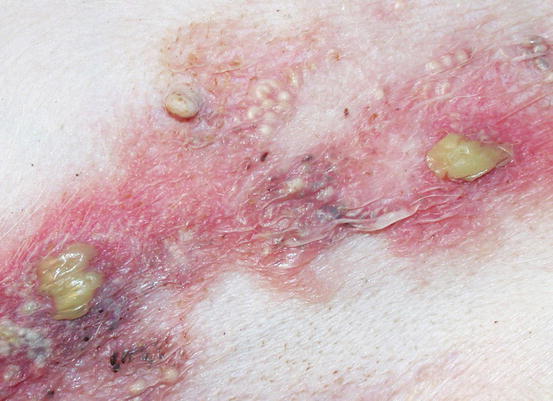

Fig. 3.80
Histology of impetigo: large non-follicular pustule

Fig. 3.81
Multiple large non-follicular pustules on the sternum and chest of a dog with juvenile impetigo

Fig. 3.83
Bullous impetigo: large non-follicular pustules in a dog treated for a long time with corticosteroids

Fig. 3.84
Bullous impetigo: erythema, comedones, skin atrophy and multiple small follicular cysts in a dog with spontaneous hypercortisolism are present together with large non-follicular pustules
Cytological Findings
The cytology of an impetiginous pustule is very similar to that described for a follicular pustule. In many cases of impetigo, especially in the bullous variant, the karyolytic nuclear changes are so severe as to make it difficult for an inexperienced cytologist to recognise those cells as degenerated neutrophils (Fig. 3.85). Another characteristic is the high number of bacteria detected compared with those observed in typical follicular pustules. This difference could be explained because bullous impetigo is usually secondary to severe and systemic immunosuppressive diseases that can impair the phagocytic ability of neutrophils, allowing them to proliferate massively.
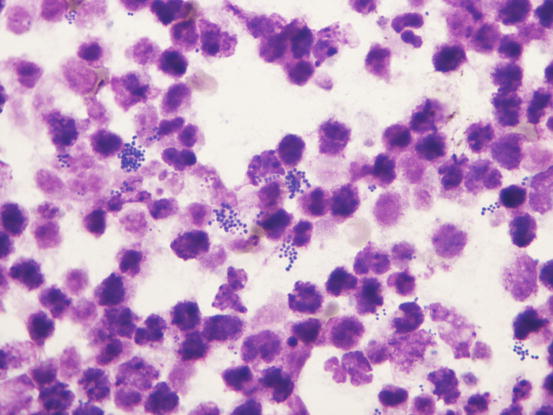

Fig. 3.85
Cytology of bullous impetigo: severe nuclear degeneration of neutrophils (karyolysis) characterised by unlobulated faded nuclei. Many cocci are immersed in the pus
Pemphigus Foliaceus
Among the sterile diseases characterised by non-follicular pustules, pemphigus foliaceus (PF) is definitely the most common. PF is an autoimmune disease histologically characterised by intra-epidermal pustules, which can be localised in the spinous, granular or corneal layer (Gross et al. 2005; Vaughan et al. 2010). The pathogenic mechanism of the pustular formation is acantholysis during which the splitting of keratinocytes following the breakage of the intercellular junctions (desmosomes) occurs. Autoantibodies of the PF target the extracellular component of some cadherins (adhesion molecules) that make up desmosomes (desmocollin-1 and to a lesser extent desmoglein-1 and plakoglobin) (Olivry 2006; Olivry and Linder 2009). Following the attack to desmosomes operated by autoantibodies, neutrophils that fill the intra-epidermal cleft created by acantholysis are recruited and give origin to the pustules.
Pemphigus foliaceus is a pustular disease that is clinically characterised by large non-follicular pustules (Fig. 3.86). Pustules rapidly dehydrate, creating a yellowish crusty dermatitis, which involves mainly the skin of the abdomen, face, pinna, muzzle and bridge of the nose (Figs. 3.87 and 3.88). The nose often shows an inflammatory depigmentation with crusts over the surface (Fig. 3.89). Paws, scrotum and mucocutaneous junctions are other sites where pustules are often observed and, in generalised cases, lesions can be present all over the body (Figs. 3.90 and 3.91) (Gross et al. 2005; Olivry 2006; Miller et al. 2013).
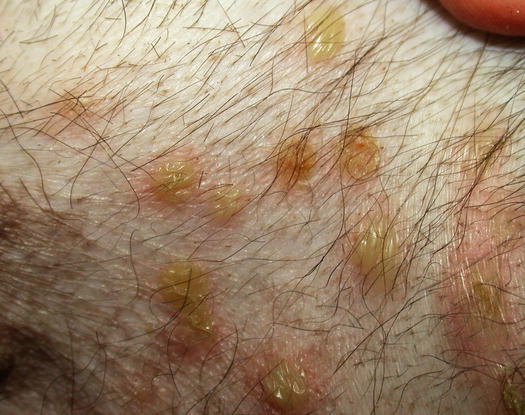
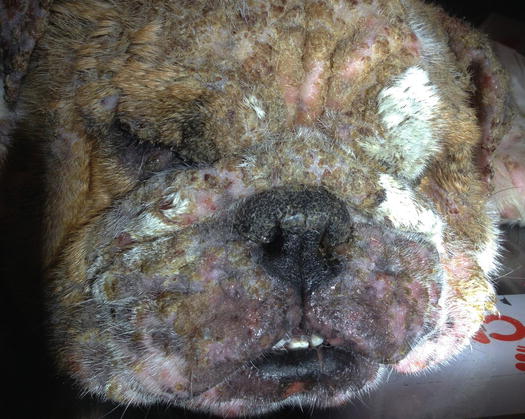
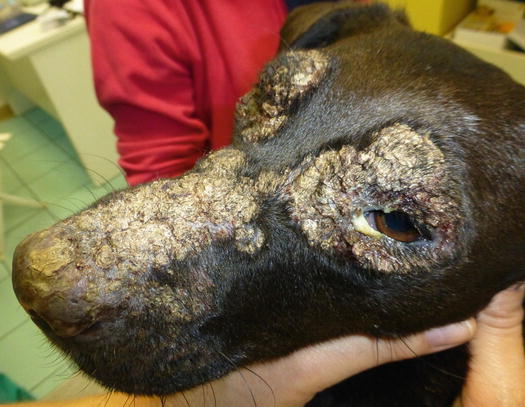
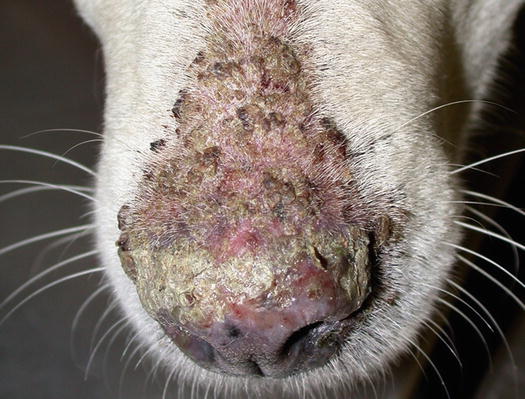
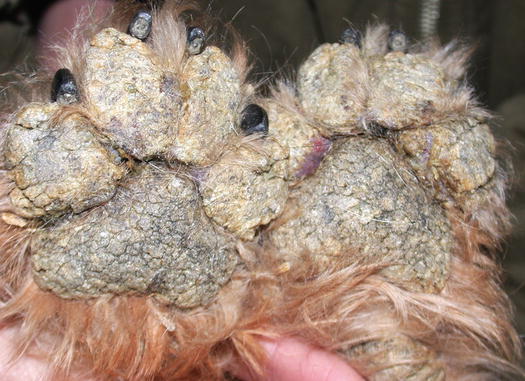

Fig. 3.86
Large non-follicular pustules in a dog affected by pemphigus foliaceus (PF)

Fig. 3.87
Yellowish crusts spread all over the face of an English bulldog with PF

Fig. 3.88
Pemphigus foliaceus: symmetrical yellowish crusts on the bridge of the nose and on the periocular area

Fig. 3.89
Inflammatory depigmentation of the nose and yellowish crusts in a dog with PF

Fig. 3.90
Diffuse crusts on the paws of a dog affected by PF

Fig. 3.91
Yellowish crusts on the scrotum of a dog with PF
Cytological Findings
As mentioned above, the histopathology of lesions in PF is characterised by intra-epidermal pustules that should contain many AKs (Fig. 3.92). Because PF is a sterile disease, samples obtained by intact pustules are typically characterised by many non-degenerated neutrophils with segmented nuclei in which nuclear lobes are recognisable. In intact pustules that have been present for several days, neutrophils can manifest morphological alterations indicative of cellular aging, such as poorly evident nuclear lobes and hyperchromatic nuclei. Together with neutrophils, a large number of keratinocytes characterised by a round/oval to polygonal shape and deep basophilic cytoplasm is observed. These cells represent the result of acantholysis and are thus named acantholytic cells or AKs (Figs. 3.93 and 3.94).
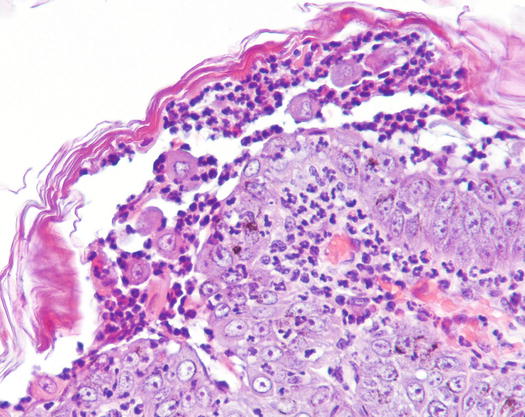

Fig. 3.92
Histology of PF: subcorneal non-follicular pustule filled with many acantholytic keratinocytes

Fig. 3.93
Cytology of PF: at low magnifications acantholytic cells are not clearly recognisable

Fig. 3.94
Cytology of PF: many segmented neutrophils and acantholytic cells indicative of PF
Acantholytic keratinocytes come from both the spinous and granular layers, depending on which part of the epidermis the acantholysis occurs. AKs have round or polygonal shape and the cytoplasm is often so dark (deep blue) that it can mask the nuclear profile when viewing the slides at low magnifications (Fig. 3.95). The nucleus is round and central, with a single nucleolus often evident. AKs can be released into the pustules, both singly and in small clusters (Fig. 3.96).
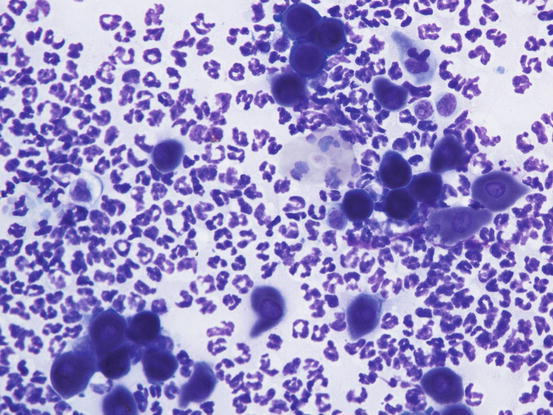
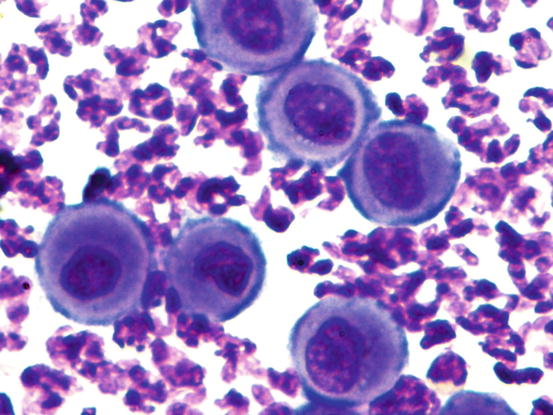

Fig. 3.95
Cytology of PF: deep blue round and polygonal acantholytic cells. Note that in many acantholytic keratinocytes the nucleus is not visible

Fig. 3.96
Cytology of PF: round-shaped acantholytic keratinocytes
Although they are the typical cells of PF, AKs are not pathognomonic of that disease, as it is possible to observe them in other infectious (bacteria and dermatophytes) and sterile pustular diseases as well. In any case, when many AKs are collected from an animal with typical clinical lesions, they are highly indicative of PF and histology must be performed to confirm this.
In many specimens it is possible to observe many single AKs surrounded by a single line of granulocytes arranged in a wagon wheel or flower petal arrangement; when AKs with these cytological findings are very numerous, PF should be suspected (Fig. 3.97). In about half of dogs with PF, cytology of pustules can show, along with neutrophils, a significant number of eosinophils, but the latter are rarely more numerous (Figs. 3.98 and 3.99) (Vaughan et al. 2010).
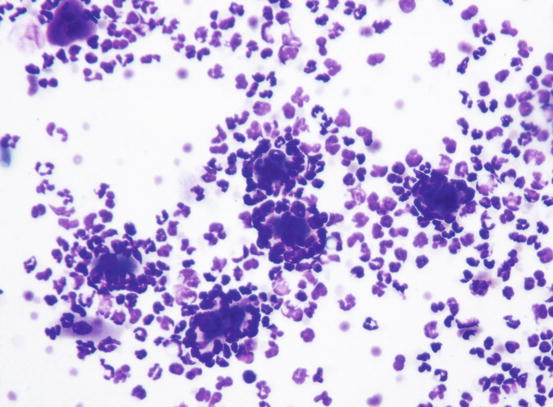
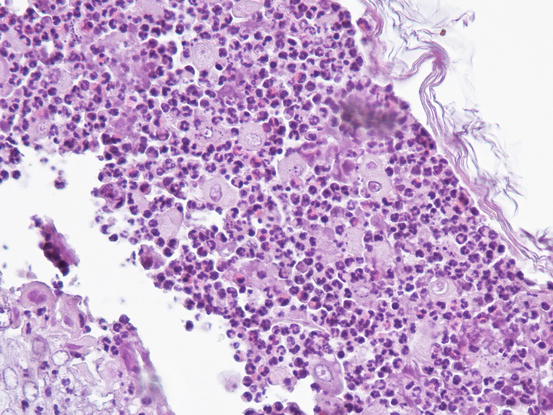
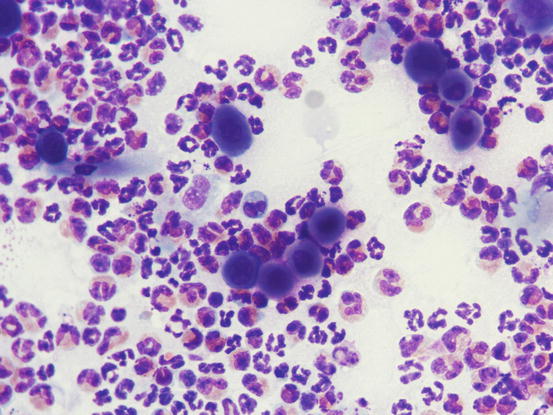

Fig. 3.97
Cytology of PF: many neutrophils arranged around single acantholytic cells

Fig. 3.98
Histology of PF: non-follicular pustule containing neutrophils, acantholytic cells and many eosinophils

Fig. 3.99
Cytology PF: neutrophils, acantholytic cells and many eosinophils
Dermatophytosis
Pustular dermatitis in dogs affected by dermatophytes belonging to the Trichophyton mentagrophytes complex, are rarely reported (Scott 1994; Parker and Yager 1997; Poisson et al. 1998).
This form of dermatophytosis is histopathologically characterised by superficial epidermal, non-follicular pustules. In some dogs, pustules may contain a variable number of AKs and histopathological findings can be very similar to those observed in PF (Fairley 2001; Gross et al. 2005; Peters et al. 2007). For a definitive diagnosis fungal hyphae should be discovered in the stratum corneum, sometimes detectable only using special stains such as PAS or Grocott’s. The mechanisms by which dermatophytes cause acantholysis is still unknown, but presumably it occurs because of proteases secreted by the fungi (Poisson et al. 1998; Olivry and Linder 2009).
The clinical lesions are characterised by superficial pustules and yellowish crusts with a distribution resembling that of PF (Fig. 3.100).
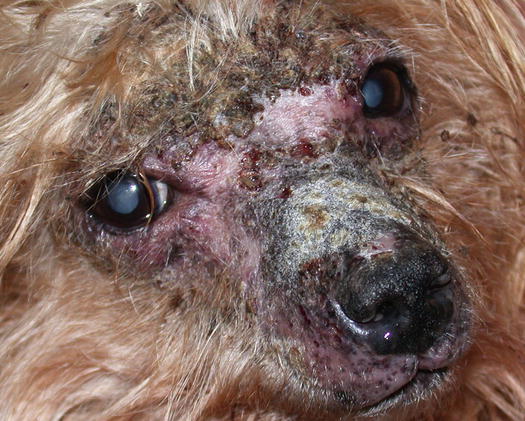

Fig. 3.100
Pustular dermatophytosis: yellowish crusts on the face and on the bridge of the nose (Courtesy of Dr. C. Caporali, Italy)
Cytological Findings
As mentioned, cytological specimens from intact pustules may be very similar to those described in PF, with many AKs and segmented neutrophils. If there is the clinical suspicion of dermatophytic pustulosis, to detect fungi, the surface of the corneocytes dispersed on the specimens must be accurately checked. In more successful cases, septate hyphae are detected on the surface of the keratinocytes (Figs. 3.101 and 3.102). Special stains such as PAS or Grocott’s can also be useful in identifying hyphae on cytological specimens.
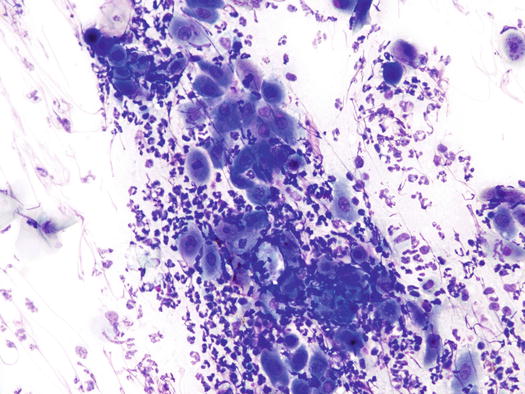
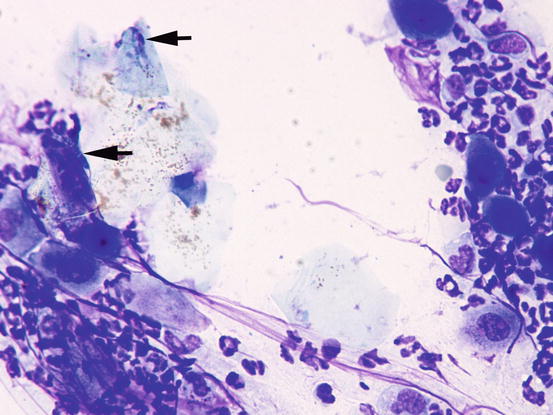

Fig. 3.101
Cytology of pustular dermatophytosis: segmented neutrophils and many acantholytic cells. Note that the cytological features are similar to those observed in PF

Fig. 3.102
Cytology of pustular dermatophytosis: some septate hyphae are detected on the surface of the corneocytes (arrows)
Idiopathic and Drug-Related Sterile Pustular Diseases
Apart from PF, other immune-mediated sterile pustular diseases, such as subcorneal pustular dermatosis with IgA deposits (Kalaher and Scott 1990; Gross et al. 2005), superficial acantholytic pustulosis due to a drug reaction (Oberkirchner et al. 2011; White et al. 2002; Bizikova et al. 2014, 2015) and idiopathic pustular dermatitis, are rarely reported in dogs. All these diseases are clinically characterised by non-follicular pustules spread all over the body (Figs. 3.103 and 3.104) (Miller et al. 2013).
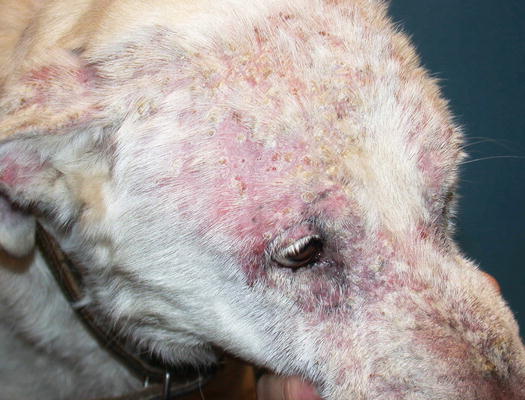

Fig. 3.103
Yellowish crusts secondary to pustule dehydration in a dog with sterile neutrophilic subcorneal pustulosis
Cytological Findings
Cytological specimens are not diagnostic, as they are composed of segmented neutrophils and a variable number, usually low number, of acantholytic cells (Fig. 3.105).
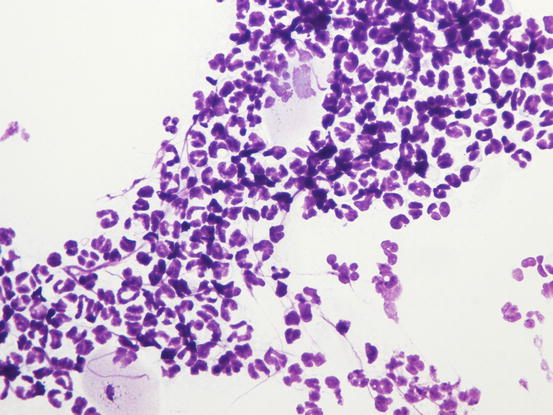

Fig. 3.105
Cytology of sterile subcorneal pustulosis: many segmented neutrophils and no acantholytic cells
Non Follicular Pustulosis in Dogs With Canine Leishmaniasis
In veterinary literature, there are only a few articles describing neutrophilic pustular lesions in dogs affected by leishmaniasis in which superficial pustules are reported (Ferrer et al. 1988; Saridomichelakis and Koutinas 2014). Unfortunately, a clear description of the characteristics of pustules and their localisation is lacking. Histopathological examination describes subcorneal neutrophilic pustules with few or no AKs (Bardagì 2012).
In a recent paper, dogs suffering from visceral leishmaniasis associated with pustular dermatitis have been reported (Colombo et al. 2016). Cultures of the purulent exudate are negative and many dogs had not responded to previous systemic antibiotic therapy. The author has visited many leishmaniotic dogs with this clinical presentation that have not regressed with anti-protozoal therapy, but required immunosuppressive drugs. The healing of the lesions with immunomodulatory drugs suggests that an immune-mediated mechanism, probably triggered by leishmaniasis, might have occurred.
As canine leishmaniasis is endemic in many countries, it is not possible to exclude the possibility that two different diseases might affect animals simultaneously. A small criticism of this evidence is that in practice sterile pustular diseases are very rare in dogs and it seems strange that most dogs with this clinical presentation are simultaneously affected by leishmaniasis. However, to date, it has not been possible to demonstrate the real role of Leishmania in the development of pustular eruptions. Pustules have an aspect and distribution similar to those described for PF, and as occurs in this autoimmune disease, systemic symptoms such as fever and malaise are sometimes observed (Figs. 3.106 and 3.107).
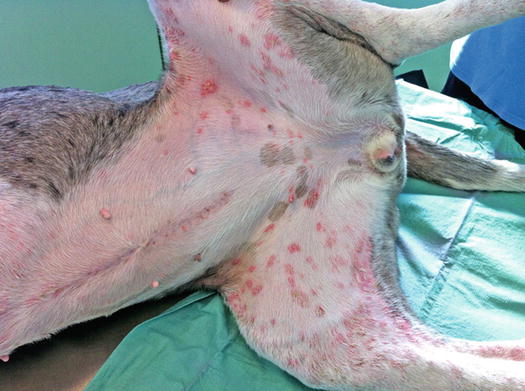

Fig. 3.106
Large non-follicular pustules with an erythematous halo in a dog affected by sterile pustulosis associated with leishmaniasis
Cytological Findings
Cytological and histopathological findings are not compatible with PF and in most cases AKs are absent. When they are present, they are less numerous than in PF. Histopathologically, pustules are non-follicular and in many dogs, macrophages filled with amastigotes can be detected in the subepidermal dermis (Figs. 3.108 and 3.109). In some dogs, to detect a few amastigotes in the dermis, immunohistochemical staining must be performed (Fig. 3.110). Cytology of pustules is composed of segmented neutrophils, whereas AKs are usually scarce or totally absent (Fig. 3.111).
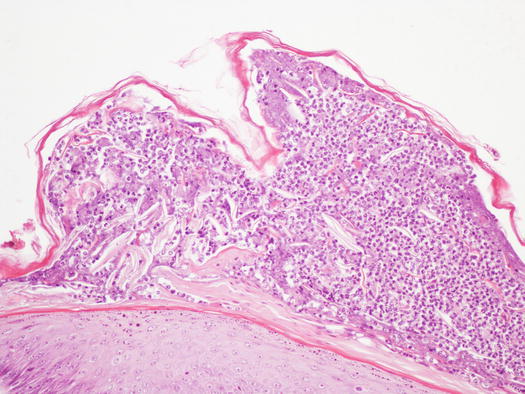
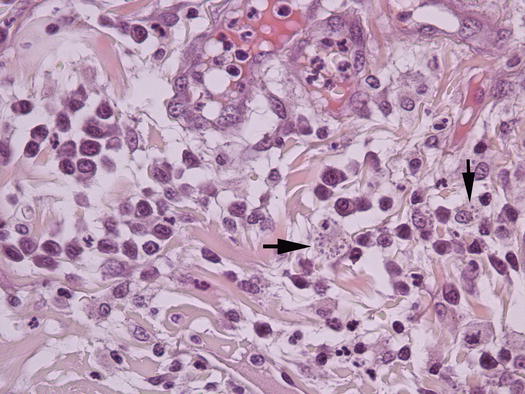
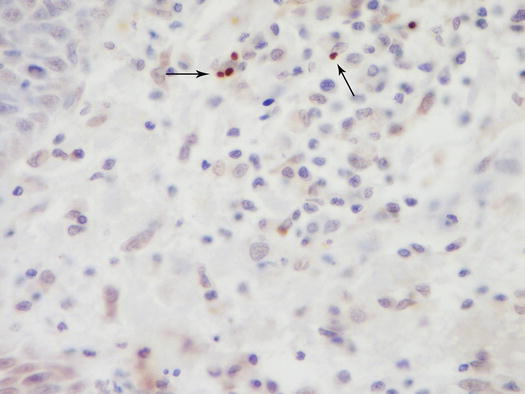
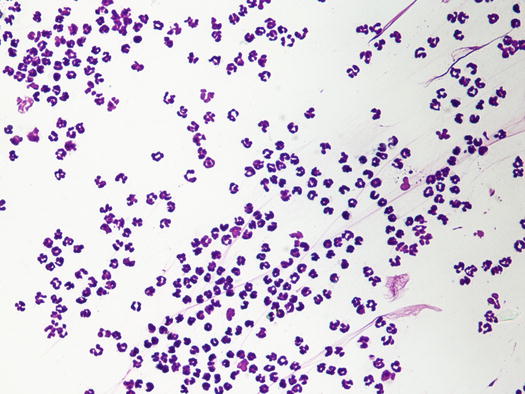

Fig. 3.108
Histology of sterile pustules in a leishmaniotic dog: very large subcorneal non-follicular pustule with no acantholytic keratinocytes

Fig. 3.109
Histology of leishmaniasis: many amastigotes of leishmania infantum in the cytoplasm of macrophages (arrows)

Fig. 3.110
Immunohistochemistry: few amastigotes of leishmania infantum are highlighted by immunohistochemical staining (arrows) (Courtesy of Prof. F. Abramo, Italy)

Fig. 3.111
Cytology of a sterile pustule in a dog with leishmaniasis: many segmented neutrophils and no acantholytic cells
Eosinophilic Pustulosis
Sterile eosinophilic pustulosis is a rare idiopathic disease of dogs characterised by the development of intra-epidermal or subcorneal pustules, predominantly localised on the trunk and associated with systemic symptoms such as fever, malaise, lymphadenopathy and eosinophilia (Scott 1987; Carlotti et al. 1989; Gross et al. 2005). The cause of skin lesions is unknown, but the response to immunomodulatory drugs suggests an immune-mediated pathogenesis.
Intra-epidermal eosinophilic pustules are a common finding in histopathological samples from animals affected by fleabite allergic dermatitis (Gross et al. 2005). The pustules are microscopic and not clinically appreciable. Although the primary clinical lesions most commonly found in dogs with fleabite allergy are papules or crusted papules on the rump, backs of the thighs and abdomen, sometimes it is possible to observe small non-follicular pustules on the groins, abdomen and ventral area of the thighs (Fig. 3.112) (Miller et al. 2013). As these eosinophilic pustules are clinically indistinguishable from those observed in canine pyoderma, a cytological investigation could rapidly differentiate between them. The cytological findings and the concomitant presence of adult fleas on the skin, allow the cause of the pustules to be ascribed to fleabite hypersensitivity.
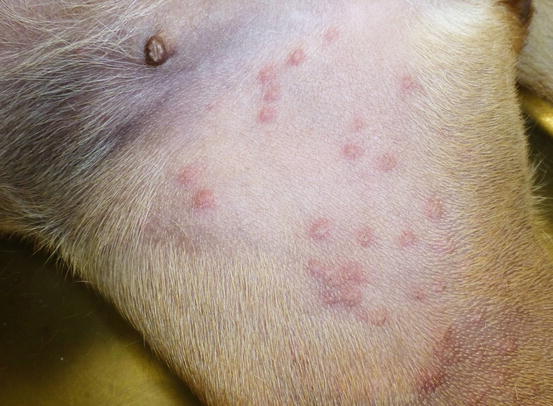

Fig. 3.112
Multiple small and non-follicular pustules on the inner surface of the legs in a dog with fleabite dermatitis
Cytological Findings
Cytology is characterised by numerous eosinophils, a variable number of neutrophils and rare or no AKs (Fig. 3.113).
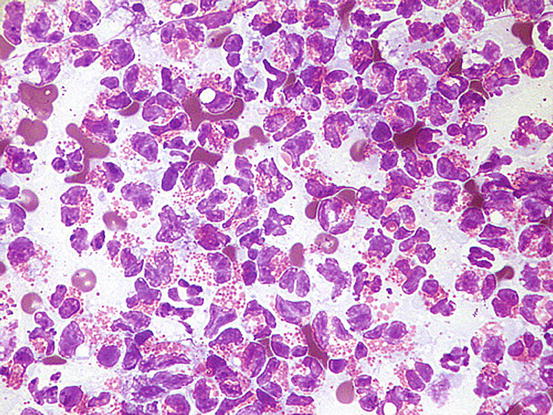

Fig. 3.113
Cytology of eosinophilic pustulosis: pure eosinophilic inflammation
3.4.2 Pustular Diseases in Cats
Pustular diseases are very rare in cats; thus, the chances of detecting intact pustules on the skin of cats are extremely low.
3.4.2.1 Bacterial Infection
Apart from abscesses and other rare bacterial infections (mycobacteriosis), pyoderma in cats is usually related to the complication of self-trauma in patients with pruritic diseases, and the pathogenic role of bacteria in these cases has not been proven. However, clinical presentations in cat pyoderma are very different from those seen in dogs.
An uncommon form of localised pyoderma that rarely shows non-follicular pustules is observed in feline acne. Feline acne is considered an idiopathic disease associated with a defect of follicular keratinisation. Skin lesions vary depending on the chronicity and the development of furunculosis; sometimes together with comedones, papules and papular–nodular lesions, a few non-follicular pustules can be observed (Fig. 3.114). Exceptionally, non-follicular pustules may be detected in cats with systemic diseases such as endocrine disorders (Fig. 3.115).
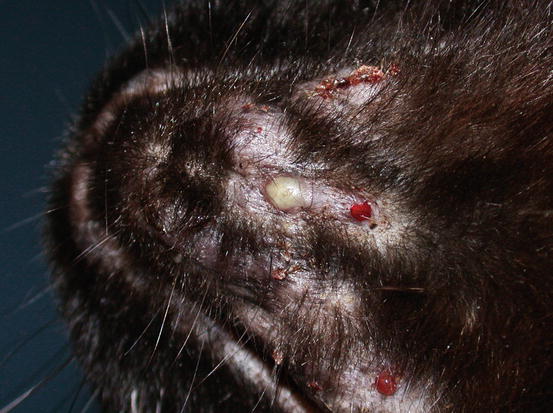
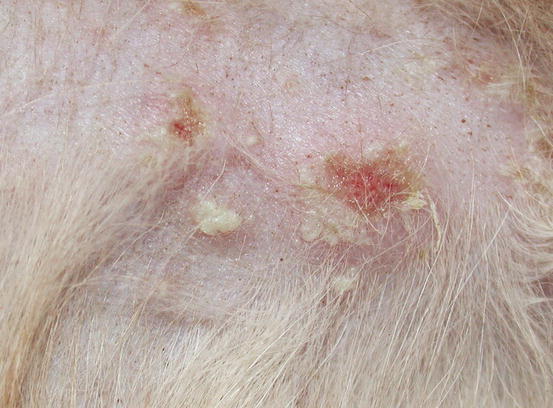

Fig. 3.114
Large non-follicular pustule on the chin of a cat with feline acne

Fig. 3.115
Large non-follicular and confluent pustules on the abdomen of a cat affected by diabetes mellitus
Cytological Findings
As in dogs, cytology of bacterial pustules in cats consists of karyolytic neutrophils with intracytoplasmic cocci (Fig. 3.116).
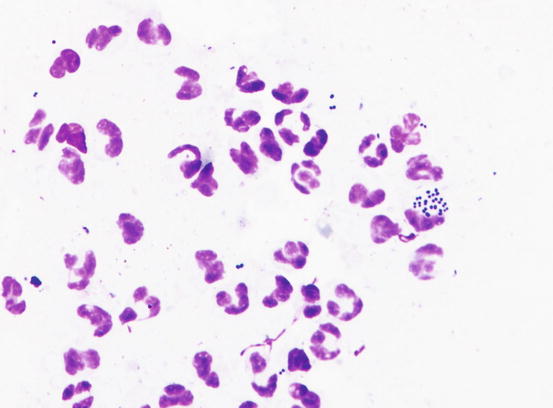

Fig. 3.116
Cytology of pyoderma: karyolytic neutrophils that phagocytose many cocci
3.4.2.2 Pemphigus Foliaceus
Even in cats, PF is a pustular disease. Unlike the dog, the skin of the cats is thinner and the possibility of finding intact pustules is very rare. Pustules rapidly dehydrate and give rise to yellowish crusts. Pustules or crusts located on the pinna, face, nose and around the nipples are highly indicative of feline PF. In association, multidigital paronychia with the nail fold covered by dehydrated pus with a characteristic cheesy appearance, is commonly present (Figs. 3.117, 3.118, and 3.119; Preziosi et al. 2003; Miller et al. 2013).
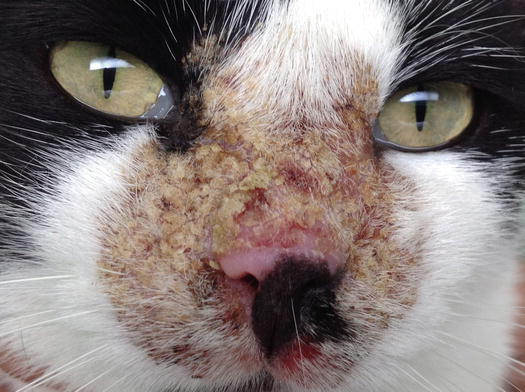
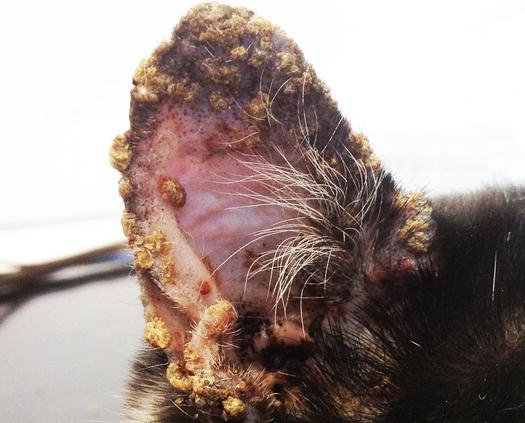
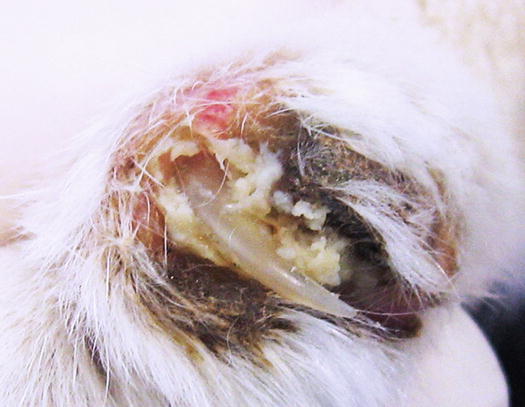

Fig. 3.117
Yellowish crusts around the nose of a cat with PF

Fig. 3.118
Yellowish crusts from dehydrated pustules on the pinna of a cat with PF

Fig. 3.119
Paronychia with dried pus with a cheesy appearance on the nail fold
Cytological Findings
Histopathology and cytology of feline PF are similar to those for PF in dogs, with a population of segmented neutrophils and a high number of acantholytic cells (Fig. 3.120) (Gross et al. 2005). In most cats is very hard to find intact pustules; thus, cytological sampling must be performed from the inner surface of the crusts and histopathological diagnosis of PF is often based on the detection of AKs in the crusts (Figs. 3.121 and 3.122). Cytological examination of the pus sampled from the nail folds is rarely diagnostic because it is usually characterised by numerous karyolytic neutrophils, owing to the innumerable bacteria that infect the lesions. Furthermore, AKs are rare to absent in many specimens collected from nail folds.
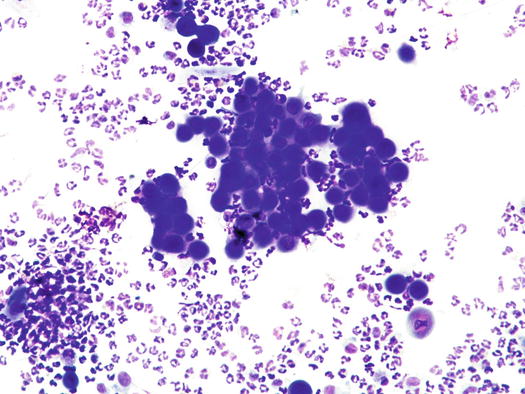
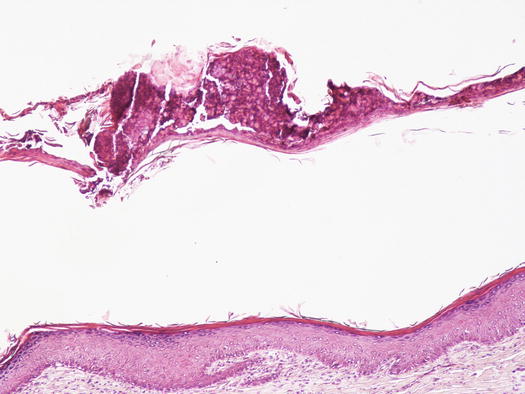
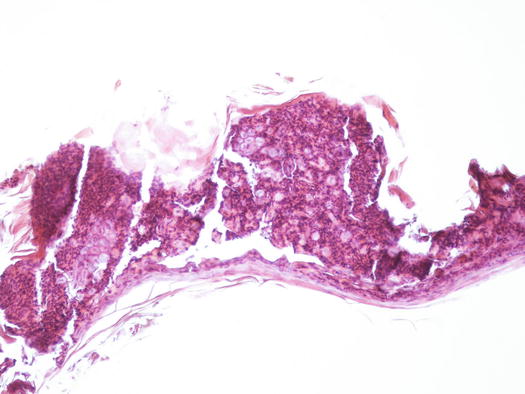

Fig. 3.120
Cytology of PF: segmented neutrophils and many acantholytic keratinocytes arranged both singly and in large clusters

Fig. 3.121
Histology of PF: neutrophilic crust detached from the superficial epidermidis

Fig. 3.122
Histology of PF: at high magnifications, many acantholytic keratinocytes are evident in the context of the crust
3.5 Scales
In many dogs and cats, collection using the impression smear technique from skin with an intact epidermal surface covered in dry or oily/waxy scales can provide useful diagnostic results.
3.5.1 Scaling Diseases in Dogs and Cats
3.5.1.1 Bacterial Overgrowth Syndrome
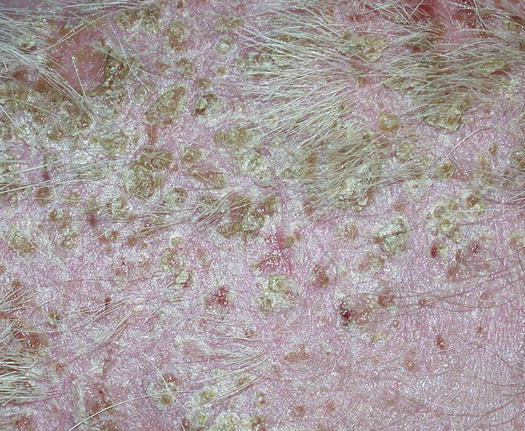
Bacterial overgrowth syndrome (BOGS) is a dermatological disorder observed in dogs characterised by an overgrowth of bacteria on the skin surface in the absence of true neutrophilic inflammation (Jasmin et al. 2001; Pin et al. 2006). An underlying disease usually affects dogs with BOGS, most frequently atopic dermatitis, as it causes injury to the cutaneous barrier and consequently impairs the defensive mechanisms favouring bacterial overgrowth and toxin overproduction. From the above, to resolve BOGS, the underlying predisposing disease must be diagnosed and treated. Skin lesions are characterised by erythema, pruritus, with skin usually covered in a shiny, sticky film and frequently associated with chronic lesions such as lichenification and hyperpigmentation, especially on the groin and axilla (Fig. 3.123).
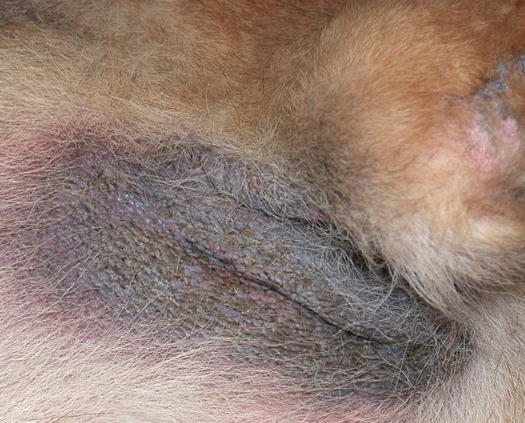

Fig. 3.123
Lichenification and skin hyperpigmentation of the axilla in a dog with bacterial overgrowth syndrome (BOGS)
Cytological Findings
Cytology is characterised by a large number of corneocytes with many cocci on their surface, but with no inflammatory cells. Anecdotally, although the number of bacteria is usually much higher, it is believed that to diagnose BOGS, more than five bacteria per immersion field (×100) must be detected (Fig. 3.124).
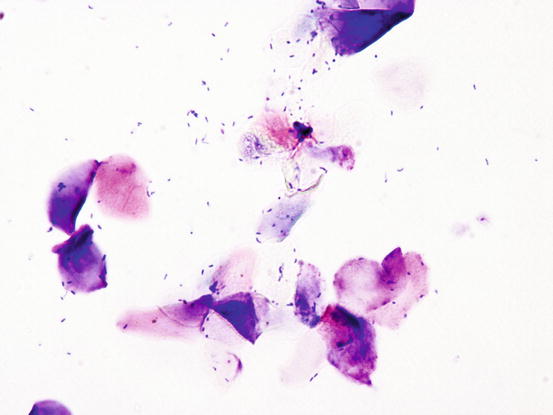

Fig. 3.124
Cytology of BOGS: polygonal corneocytes with many cocci and rod-shaped bacteria on their surface. Note the absence of neutrophils
3.5.1.2 Malassezia Overgrowth
Malassezia pachydermatis is commensal on the skin and mucous membranes of dogs. As discussed for BOGS, there are factors that promote pathogenicity and allow Malassezia overgrowth, such as high temperatures and humidity, excessive secretion of lipids and underlying diseases, e.g. atopic dermatitis (Guaguère et al. 1996; Mauldin et al. 1997, 2002; Chen and Hill 2005; Gross et al. 2005). Malassezia overgrowth usually causes very pruritic dermatitis associated with scaling, erythema and a bad smell (Bond et al. 2004). Typology and topography of lesions are the same as described for BOGS, with which it is often associated.
Malassezia overgrowth/dermatitis is a frequent complication in dogs suffering from allergic diseases (especially atopic dermatitis), even though any disease that affects the skin surface ecosystem can cause excessive multiplication of yeasts. In dogs, clinical lesions are characterised by erythema, greasy scaling, often with a waxy appearance, orange in colour, and with rancid smell, usually associated with lichenification and hyperpigmentation (Figs. 3.125 and 3.126). Such findings are mainly evident in more humid body areas such as the interdigital area, the nail fold, the ventral surface of the neck, axilla and groin, but in some individuals they may be present throughout the body.
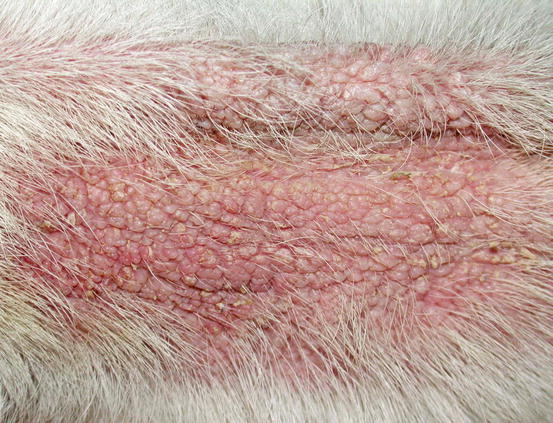
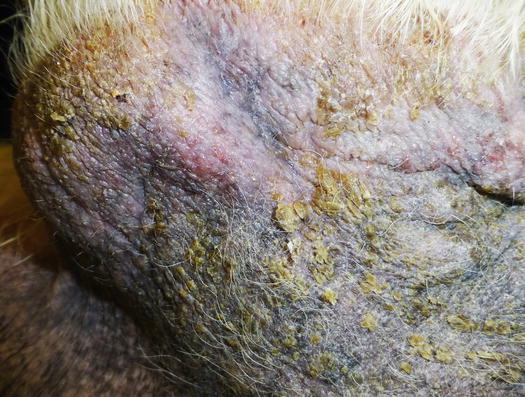

Fig. 3.125
Alopecia, lichenification and waxy scales in a dog with Malassezia overgrowth

Fig. 3.126
Alopecia, erythema, lichenification and waxy scales in a dog with Malassezia dermatitis
Frequently, whether associated with other skin lesions or not, dogs present ceruminous otitis with a yeast overgrowth characterised by yellowish and smelly secretion.
In feline patients Malassezia overgrowth is rarer than in dogs (Miller et al. 2013). Usually, yeasts are observed in cats affected by hypersensitivity or immune-suppressive diesases, such as paraneoplastic alopecia associated with liver or pancreatic neoplasia and exfoliative dermatitis associated with thymoma (Forster-Van Hijfte et al. 1997; Godfrey 1998).
Cytological Findings
As mentioned, Malassezia spp. is a commensal yeast that lives on the skin surface and when there is overgrowth, a high number of yeasts are usually collected. Malassezia is an oval microorganism, measuring 3–8 μm, that assumes a characteristic bilobate appearance during the budding phase (American peanut shape; Fig. 3.127). The number of yeasts present on cytopathological preparations is variable and dependent on the type of injury, the sampling method and the breed. It has been shown that in some feline breeds such as the Devon rex or in canine breeds such as the Basset hound, the number of yeasts on the skin is physiologically higher than in other breeds (Ahman et al. 2007). Anecdotally, is believed that when diagnosing a Malassezia overgrowth more than two yeasts per immersion field (×100) need to be found. Although the number of yeasts usually found is greater than that considered relevant to diagnosing a Malassezia overgrowth, in some dogs with chronic lichenification, it is possible to obtain negative samples. This occurrence is because severe epidermal hyperplasia may not allow the sampling of yeasts hidden between the epidermal folds. To avoid this misdiagnosis it is advisable to carry out repeated sampling from the same area to try to collect more deeply located yeasts. In a severe infection, the number of yeasts is so high that many Malassezia spp. can be easily collected with a piece of transparent acetate tape (Figs. 3.128 and 3.129).
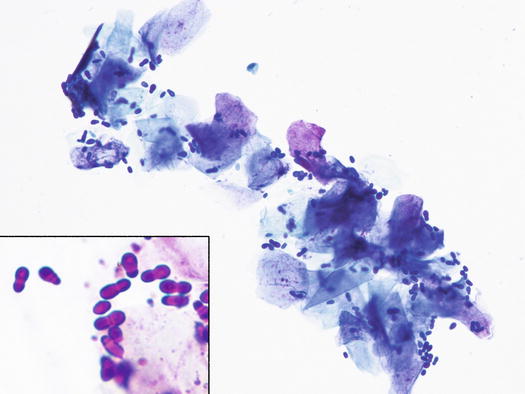
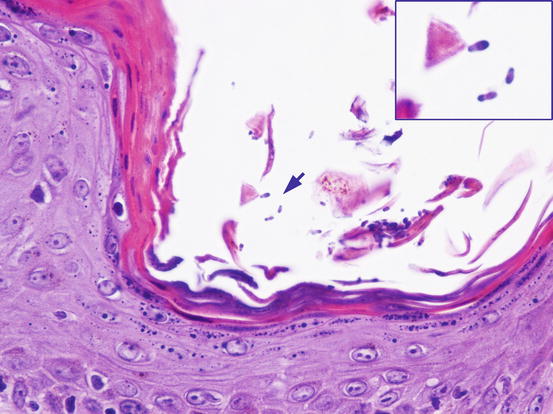
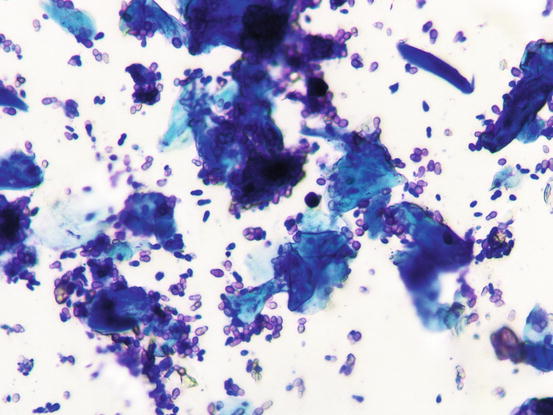

Fig. 3.127
Cytology of Malassezia overgrowth: many yeasts adhering to the surface of the corneocytes. Inset: at high magnifications, the characteristic peanut-shape of yeasts is easily recognisable

Fig. 3.128
Histology of Malassezia overgrowth: many yeasts in the stratum corneum (arrow). Note as the typical bilobed shape is well evident (inset)

Fig. 3.129
Cytology of Malassezia dermatitis: numerous Malassezia pachidermatis collected from the same lesion as in Fig. 3.128
3.5.1.3 Cutaneous Candidiasis
Candida albicans is a commensal mycelial yeast that colonises the mucous membranes of dogs and cats. In addition to C. albicans, other yeasts such as C. parapsilosis and C. guilliermondii have been reported to cause dermatitis in dogs (Carlotti and Pinn 1999; Mueller et al. 2002; Gross et al. 2005). These yeasts can occasionally cause dermatitis, but when it happens they are usually opportunistic infections due to an underlying disease that impairs the immune system, such as metabolic diseases (superficial necrolytic dermatitis/hepatocutaneous syndrome), hormonal disorders (hypercortisolism and diabetes mellitus), visceral neoplasia etc. Usually, the more humid body areas such as mucocutaneous areas, interdigitals, and skin fold areas are more frequently affected and sometimes a smelly whitish film is observed on the skin surface (Figs. 3.130 and 3.131) (Carlotti and Pinn 1999; Greene 2012; Scott et al. 2001; Miller et al. 2013).
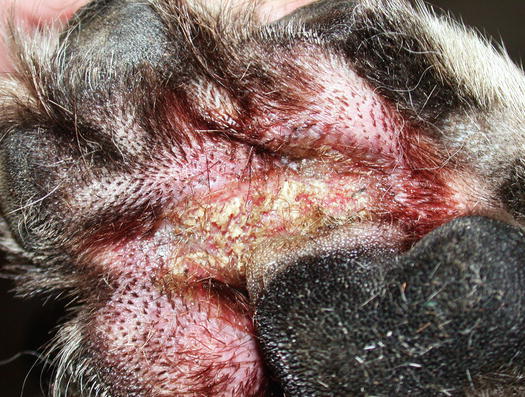
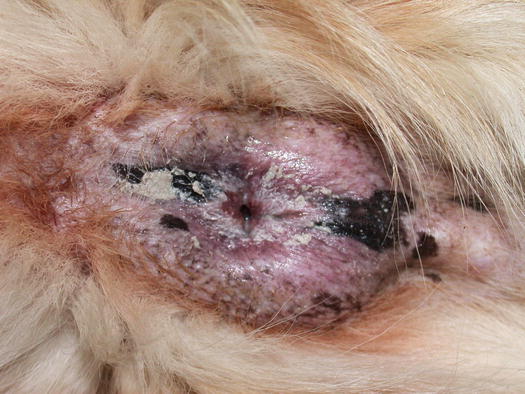

Fig. 3.130
Erythema and whitish patina in the interdigital area of a dog with pododermatitis due to Candida albicans

Fig. 3.131
Erythema and whitish patina on the anal mucocutaneous junction in a dog with cutaneous candidiasis
Like Malassezia, Candida may also be easily sampled using the imprint technique with a slide or a piece of transparent acetate tape.
Cytological Findings
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree