CHAPTER 2 Cell Types and Criteria of Malignancy
ARE SUFFICIENT NUMBERS OF WELL-STAINED, WELL-PRESERVED, INTACT CELLS PRESENT TO EVALUATE?
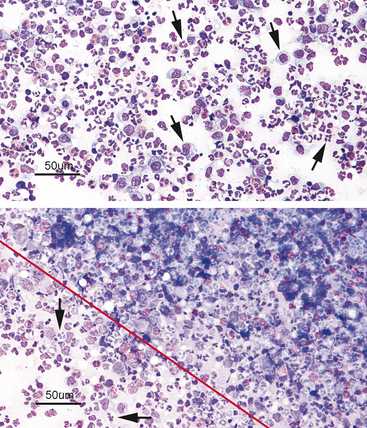
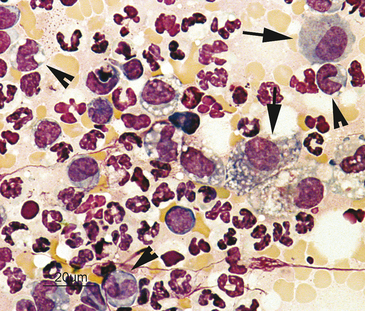
When examined microscopically, the slides should first be scanned using low power (10× to 20×) to assess the cellularity of the slide and find the area(s) containing the highest number of well-stained, well-spread-out, intact cells for evaluation (Figure 2-1). Cells are often unevenly distributed across the slides, especially with impression smears or aspirates of solid tissue lesions. Even with slides made from fluid samples, large cells may all be pulled out to the feathered edge, where they can be easily overlooked if the whole slide is not scanned first (Figure 2-2).
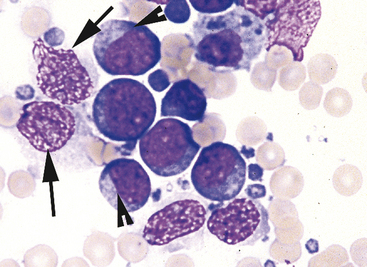
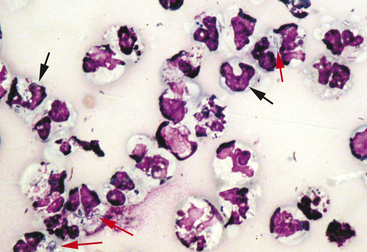
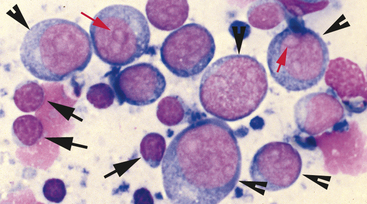
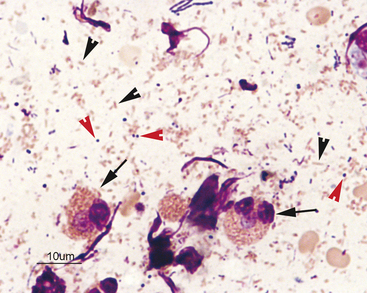
After locating the cellular areas of the slides, it is important to determine whether the cells are sufficiently spread out for evaluation, intact and well stained. This can usually also be done on low power (10× to 20×), which will allow a greater portion of the slide to be evaluated in a short time. Inexperienced cytologists often frustrate, themselves by spending an inordinate amount of time trying to identify cells that cannot be interpreted because they are not spread out, are poorly stained, or are ruptured. Intact, well-stained, well-spread cells should have a clearly evident demarcation between the nucleus and cytoplasm (Figure 2-3). The nucleus may be irregularly shaped (particularly in neoplastic cells), but the nuclear outline should be smooth and distinct. A fuzzy appearance around the outline of the nucleus generally indicates that the cell has been minimally traumatized during sample collection and/or preparation. More significant cell trauma can result in the nucleus appearing fragmented or full of holes (see Figure 2-3). Severely traumatized cells will often appear as strands of light pink nuclear chromatin (Figure 2-4). Some traumatized/ruptured cells will be present in virtually any cytologic specimen. The sample is usually still interpretable if the majority of the cells are intact; however, the traumatized cells are typically not evaluated, particularly when determining criteria of malignancy, because ruptured nuclei may appear enlarged and nucleoli may appear more prominent. If the majority of the cells are traumatized/ruptured, additional samples usually need to be collected.
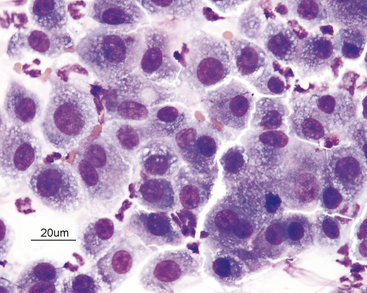
Cells that are not well spread out often stain diffusely dark and it is difficult to distinguish the line of demarcation and color distinction between the cytoplasm and nucleus (Figure 2-5). Also, poor staining (even in well-spread-out cells) can result in a less distinct demarcation between nucleus and cytoplasm.
Figure 2-5 Poorly spread-out cells from a different area of the same slide shown in Figure 2-3. The cells are diffusely dark with the nuclei and cytoplasm staining different shades of blue. Compare this with the purple color of the well-spread-out nuclei in Figure 2-3. Also, it is difficult to discern the demarcation between nucleus and cytoplasm.
ARE ALL OF THE CELLS ON THE SMEAR INFLAMMATORY CELLS?
A good initial decision, particularly for the beginning cytologist, is to determine if the smear is composed entirely of inflammatory cells. In most general practices, inflammatory lesions are probably more commonly sampled than neoplastic lesions. Also, most clinicians initially feel more comfortable recognizing inflammatory cells because they are more familiar with the morphology of these cells, having viewed them many times in peripheral blood smears. Many clinicians choose to screen their cytology specimens, interpreting inflammatory lesions in-house while submitting those composed of tissue cells for outside evaluation. Although inflammatory cells in tissues often look the same as they do in peripheral blood, some may appear different because of morphologic changes induced by being present in a focus of inflammation or simply because they are not well spread out. It is important to remember that one may not be able to identify every cell present on a slide (or even on any given field), and that the interpretation is based on the entire cell population present. If a lesion is found to be composed entirely of inflammatory cells, the relative percentages of the various types of inflammatory cells should be noted, because this may provide clues as to the etiology of the inflammation. Finally, a search for infectious agents should be conducted. A discussion of the various inflammatory patterns and morphology of common infectious agents will be covered more completely in Chapters 3 and 5 of this text. The basic types of inflammatory cells and their morphologic variations are later in this chapter.
Neutrophils
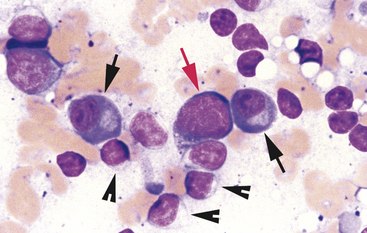
Neutrophils are commonly found in cytologic specimens. Their morphology is often similar to that observed in peripheral blood smears (Figure 2-6). Normal neutrophil nuclei stain dark purple and contain one to multiple distinct segments or lobes. The neutrophil cytoplasm is typically clear. Neutrophils are phagocytic cells and typically are the cells that phagocytize pathogenic bacteria, if present (see Figure 2-6). Neutrophils contain intracytoplasmic granules. However, the neutrophil granules in most domestic animals generally do not stain prominently with cytologic stains. Sometimes, however, these granules will be discernable as elongated, faintly eosinophilic structures, and they must not be confused with bacteria or lightly staining eosinophil granules.
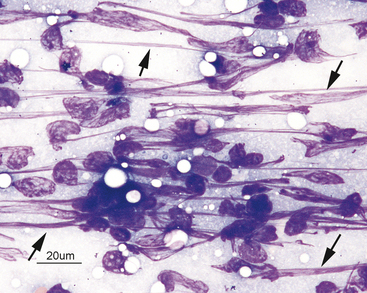
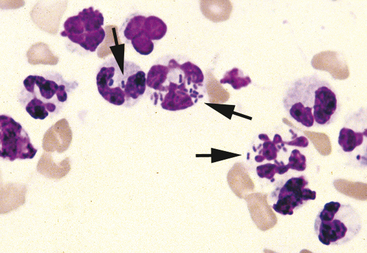
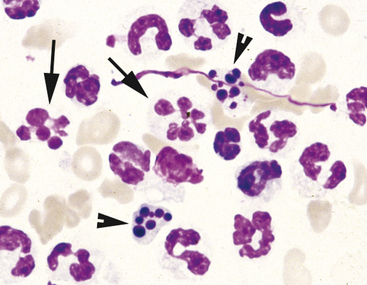
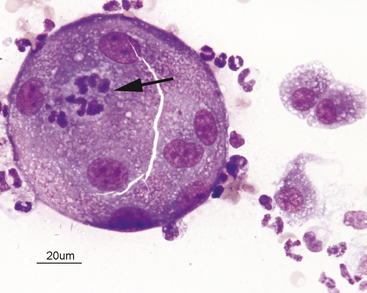
Neutrophils may undergo several morphologic changes in tissues. Aging change is a commonly encountered phenomenon. The initial change seen is hypersegmentation of the nucleus (Figure 2-7). Sometimes, an elongated thin strand of nuclear material (Figure 2-8) connects the nuclear lobes of aged neutrophils. This is especially common in neutrophils from fluid samples and is more commonly seen on cytocentrifuged preparations than in direct smears. The end result of aging change is pyknosis of the nucleus. Pyknosis is condensation of the nuclear chromatin into one or more small discrete, densely staining spheres lacking any nuclear chromatin pattern (see Figure 2-7). Aging artifact simply represents neutrophils dying of “old age” and must not be confused with degenerative change, described subsequently.
Degenerative change occurs when neutrophils are present in an environment that is damaging to the cell. It is commonly seen in neutrophils from lesions in which endotoxin-producing bacteria are present. The presence of many neutrophils with marked degenerative change should prompt a diligent search for bacteria. Degenerative change is an acquired change and is distinct from the toxic changes noted in peripheral blood neutrophils. Degenerative change occurs when the neutrophil is unable to control water homeostasis and undergoes hydropic degeneration. The hallmark of degenerative change is nuclear swelling (Figure 2-9). The nucleus of the cell swells and appears thicker, stains a lighter eosinophilic color, and loses nuclear lobulation. Degenerative neutrophils often resemble large band cells.
Macrophages
Macrophages in inflammatory lesions are derived from peripheral blood monocytes. Many tissues have low numbers of fixed tissue macrophages as normal resident cells (e.g., Kupffer cells in the liver). Macrophages may display extremely variable morphology in tissues, which can be somewhat confusing for the beginning cytologist. Initially, they may resemble peripheral blood monocytes (Figure 2-10). With time, the nucleus becomes round and the cell enlarges as the cytoplasm becomes greatly expanded and, sometimes, extremely vacuolated. Macrophages are also phagocytic cells, typically phagocytizing larger structures like fungal organisms and other cells. Many times, the cytoplasm of macrophages will contain partially phagocytized debris that cannot be identified, but must not be misinterpreted as an infectious agent.
Binucleated or multinucleated macrophages are commonly encountered in long-standing inflammatory lesions (Figure 2-11). Multinucleated macrophages can get very large and are referred to as inflammatory giant cells. In some chronic inflammatory lesions, epithelioid macrophages may be encountered (Figure 2-12). This term is applied to macrophages that are enlarged with expansive cytoplasm that stains uniformly basophilic, giving the cell the look of an epithelial cell. Because these cells often show variation in size and multinucleation (both commonly seen in macrophages) they could potentially be misinterpreted as neoplastic epithelial cells. Extreme caution should be used when diagnosing malignancy in the face of inflammation because of the potential for macrophages to display atypical criteria.
Lymphocytes (Small, Medium, and Large)
Small lymphocytes are smaller than neutrophils with rounded nuclei and scant basophilic cytoplasm (Figure 2-13). The nucleus is generally not perfectly round, but will have a flattened or indented area on one side. The nuclear chromatin has a smudged appearance. Nucleoli are not visible; however, darker areas (heterochromatin) and lighter areas (euchromatin) are often visible. Generally, the cytoplasm does not appear to completely encircle the nucleus, because it is visible for only a portion of its way around the nucleus. Medium sized lymphocytes may be present and are similar to small lymphocytes, but they have moderately increased amounts of cytoplasm and may have nucleoli visible. Large, blastic lymphocytes (lymphoblasts) (Figure 2-14), commonly encountered in aspirates of lymphoid tissue and lymphoid neoplasms, may be present in low numbers in inflammatory lesions. Lymphoblasts are large cells with large nuclei and more abundant cytoplasm, which typically stains basophilic. Nuclei can be variably shaped and have stippled nuclear chromatin, which stains somewhat lighter than that of mature lymphocytes. Distinct nucleoli are often visible, and multiple nucleoli may be observed.
Reactive lymphocytes are lymphocytes that have been antigenically stimulated. They have moderately increased amounts of basophilic cytoplasm. Plasma cells are differentiated B-lymphocytes stimulated to produce antibodies. Plasma cells have a round, eccentrically placed nucleus, moderate amounts of deeply basophilic cytoplasm, and usually a distinct clear area located next to the nucleus (see Figure 2-13). This clear area represents the Golgi apparatus and is often located between the nucleus and the greatest volume of cytoplasm. Plasma cells and lymphoblasts are both larger than small lymphocytes, but in the plasma cells most of the increase in size is due to more abundant cytoplasm, whereas in the lymphoblast the nucleus has enlarged (see Figure 2-13).
Some plasma cells (termed Mott cells) have numerous large clear vacuoles (termed Russell bodies) filling their cytoplasm (Figure 2-15). These vacuoles represent retained immunoglobulin.
Eosinophils
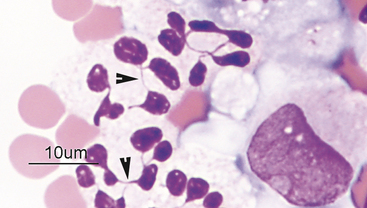
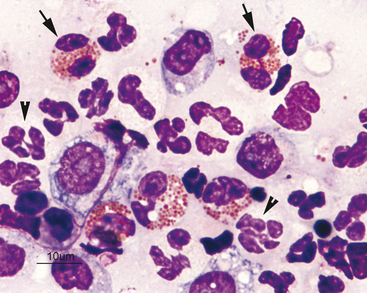
Eosinophils are slightly larger than neutrophils. Their nuclei are segmented, but commonly less lobulated than that of neutrophils and often divided into only two distinct lobes (Figure 2-16). Rarely, eosinophils with perfectly round nuclei will be identified in cytologic specimens. The cytoplasm of eosinophils contains prominent orange to pink granules. In dogs, eosinophil granules are round and vary widely in size and number (Figure 2-17). Eosinophil granules are numerous, small, and rod shaped in cats (Figure 2-18). The delicate, densely packed granules of feline eosinophils are often less obvious than those of the dog, particularly in thick specimens that may not stain well (i.e., transtracheal washes). Also, neutrophils in exudates will occasionally have mild eosinophilic stippling. Care must be taken not to confuse neutrophils and poorly stained eosinophils when trying to differentiate eosinophilic from neutrophilic inflammatory reactions in cats. Eosinophils are slightly larger than neutrophils and their nuclei less lobulated. Often, it is easier to identify feline eosinophils that have been traumatized during slide preparation as their granules spread out and become more obvious. If many eosinophils have been ruptured during sample collection (such as with scraping of feline eosinophilic granuloma complex lesions) high numbers of eosinophil granules will be present throughout the background of the smear and may be identified before intact cells are seen.
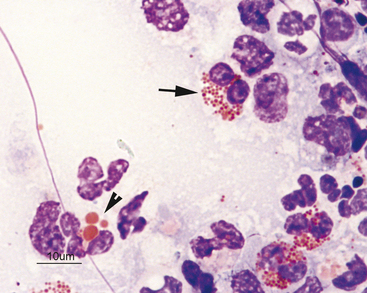
Figure 2-17 Image from the same slide as Figure 2-16. In the dog, eosinophils have numerous small granules (arrow) or just a few large granules (arrowhead).
IF A SMEAR IS COMPOSED OF TISSUE CELLS RATHER THAN INFLAMMATORY CELLS, WHAT TYPE OF CELLS ARE PRESENT?
A wide variety of specific cells may be encountered from the various normal tissues and tumors sampled cytologically. With experience, most of these cells can be easily recognized, particularly with the knowledge of what structure is being sampled. However, even if the cells are not immediately recognizable, they can generally be classified into one of three major categories based on certain common cytologic features (Table 2-1):
Discrete Cells (Round Cells)
General Cytologic Characteristics of Discrete Cell Populations
Because discrete cells are not adhered to other structures within the tissues, they generally exfoliate very readily during fine-needle biopsy (FNB). Hence, the cellularity of the resulting smears is usually very high. In addition, the individual cells are usually evenly spread throughout the smear (Figure 2-19). Cell clusters or aggregates are not present; however, the extremely high cellularity of the smears may result in cells being piled on top of each other in thicker areas of the smears and this may be misinterpreted as cell adhesion (cell clustering). In the thinner areas of the smears, the cells can be seen to be individually oriented.
Specific Discrete Cell Tumors
Mast Cell Tumor
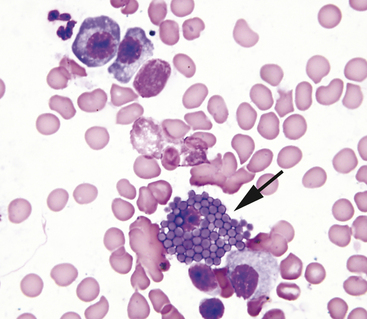
Mast cell tumors are the only lesions that will yield highly cellular smears consisting entirely (or predominantly) of mast cells. Mast cells are recognized by their distinctive small, red-purple intracytoplasmic granules (Figure 2-20). The number of granules in mast cells varies tremendously, even within cells from the same tumor (Figure 2-21). Most mast cell tumors yield cells that contain a sufficient number of granules to be easily recognized as mast cells. Sometimes, the cells are so densely packed with granules that the cytoplasm will appear diffusely dark purple and the individual granules difficult or impossible to discern. In this situation, the granules will be evident in cells that have been ruptured. Since mast cell granules have such a high affinity for most cytologic stains, the nucleus of a heavily granulated mast cell may appear pale or even totally unstained, giving the cell a photographic negative look (dark cytoplasm with pale nucleus) (Figure 2-22). Some of the components of mast cell granules are chemotactic for eosinophils. The number of eosinophils present in smears from a mast cell tumor varies from very few to many. Occasionally, an aspirate from a mast cell tumor will yield predominantly eosinophils with lesser numbers of mast cells (Figure 2-23). In this case, it can be difficult to differentiate a mast cell tumor from a hypersensitivity response. Generally, if there are areas on the slides containing large sheets or aggregates of mast cells, mast cell tumor is most likely.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree