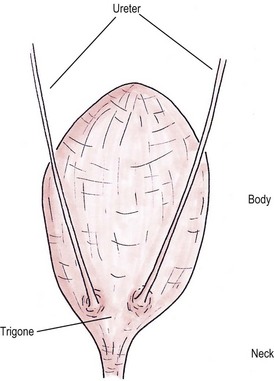
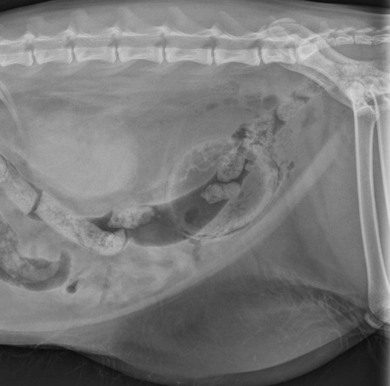
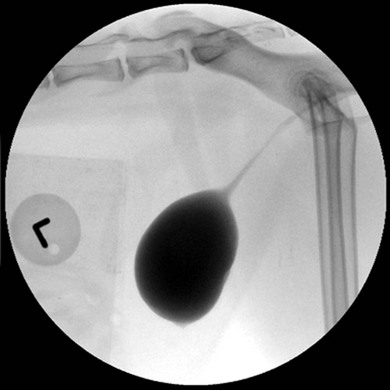
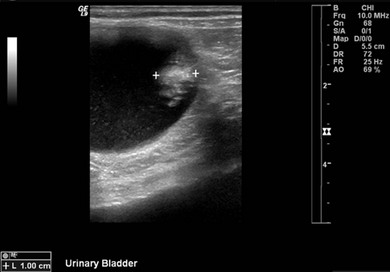
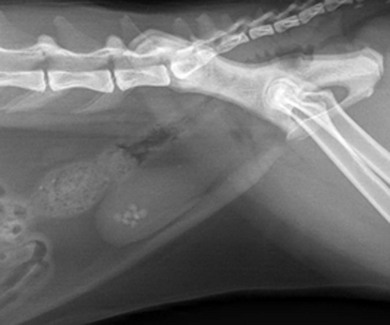
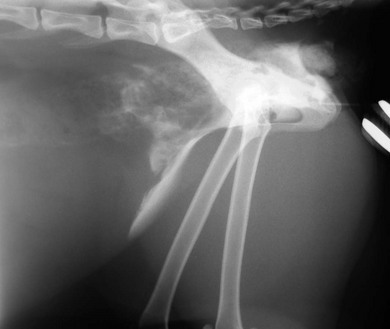
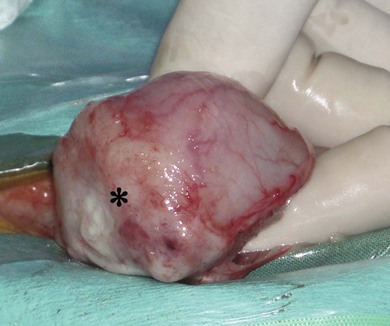
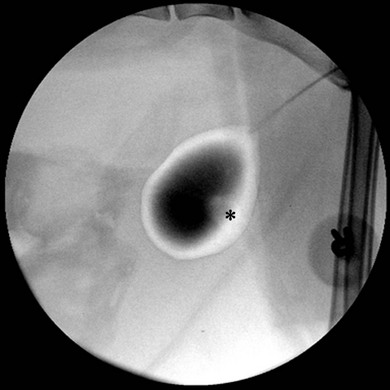
Chapter 37 Unlike the canine bladder, the feline bladder remains consistently craniad to the pubis regardless of its degree of distension.1 The bladder is located ventral to the descending colon, with the ductus deferens or uterine body contacting the dorsal surface in intact males and females, respectively. The bladder is attached to the caudal abdominal body wall by three serosal folds. In the female, two lateral vesical folds are continuous with the uterine broad ligaments and contain the round ligaments of the bladder, which are fetal remnants of the umbilical arteries. A third median vesical fold arises from the ventral abdominal wall and contains the vestigial fetal urachus. The bladder consists of two distinct anatomic areas, the neck and the body. The body comprises the majority of the bladder, and its most cranial aspect is the apex. The neck contains the trigone, which is a triangular area on the dorsal aspect of the bladder bound by the urethral orifice caudally and the ureteral openings cranially (Fig. 37-1). The main vascular supply arises from the caudal vesicle artery which originates from the vaginal or prostatic artery, which are branches of the internal pudendal artery. The paired caudal vesicle arteries enter the bladder at the neck and course both ventrally and dorsally to anastomose with the contralateral vessels. The nervous supply to the bladder consists of both autonomic and somatic innervation. Autonomic control is mediated via the sympathetic (hypogastric nerve) and parasympathetic (pelvic nerve) nervous systems and the somatic control is mediated via the pudendal nerves. The lymphatics of the feline bladder drain into the hypogastric and lumbar lymph nodes. Survey radiography of the bladder is useful for general evaluation of the size, shape, presence of radiopaque calculi, or other abnormalities of the bladder (Fig. 37-2). If additional information is required following survey radiographs, contrast procedures may be indicated. If contrast radiography is performed on an elective basis, overnight fasting and a cleansing enema should be performed to aid in the detection of subtle urinary tract abnormalities by limiting the amount of ingesta and gas in the small intestine and colon that may obscure the bladder and urethra. The following contrast procedures complement each other, and they should be done in the order outlined below if it is likely that multiple procedures will be performed as part of the diagnostic workup. However, all of these contrast procedures are rarely indicated for a single case. Pneumocystography can be useful for evaluating the bladder for wall thickness, radiopaque calculi, and diverticula. Pneumocystography can be performed with infusion of air or CO2 into the bladder via a urethral catheter after the bladder has been emptied to prevent any residual urine from creating the appearance of an artificially thickened bladder wall. Full distension of the feline bladder is generally achieved with 15–45 mL, and can be detected with digital palpation. In many cases, partial distension of the bladder is sufficient for evaluation and may be more useful for demonstrating small urachal diverticula that are not apparent with full distension.2 Double contrast cystography aids in detection of bladder wall lesions and radiolucent calculi, which appear as intraluminal filling defects when outlined by the contrast material (Fig. 37-3). It is performed by infusing 1 mL of aqueous organic iodinated contrast solution through a urethral catheter, followed by infusion of air or CO2 to further distend the bladder. If a positive contrast cystogram is to be performed, the bladder should be emptied before moderately distending it with contrast solution. Positive contrast cystography is useful for detecting bladder location, rupture or leaks, and urachal diverticula (Fig. 37-4). Figure 37-3 Lateral fluoroscopic image of a double contrast cystogram of a cat. Note the subtle filling defect on the ventral aspect of the bladder lumen as indicated by the asterisk. Ultrasonography is a valuable tool for diagnosing various diseases or conditions of the bladder including wall thickening or inflammation, masses or neoplasia, and cystoliths (Fig. 37-5). Both radiopaque and radiolucent calculi as small as 2 mm in diameter can routinely be diagnosed with ultrasound.3 The ability to detect calculi or assess the size and location of a mass is influenced by the degree of bladder distension, and accuracy can be improved by infusing physiologic saline into the bladder to separate the walls and better isolate specific lesions. However, ultrasonography is not as sensitive as survey radiography for determining the exact size, number, and possible composition of radiopaque calculi. Cystoscopy is an excellent diagnostic tool that allows for direct visualization of various lesions, as well as sampling, using a biopsy instrument or basket (see Chapter 9). Transurethral cystoscopy can be performed in male and female cats using small diameter flexible or rigid cystoscopes, respectively.4 Examination of the lumen of the urethra and bladder can be accomplished for diagnosis of lesions such as tumors, polyps, inflammation, mucosal tears, calculi, and ectopic ureters. In many cats, it can be difficult or impossible to pass a biopsy instrument simultaneously with the cystoscope and, therefore, if samples of the bladder are desired, the cystoscope can be used to guide percutaneous fine needle aspiration of specific lesions.5 The composition of feline uroliths has evolved during the past thirty years (Fig. 37-6). Struvite uroliths were very common in the early 1980s, while calcium oxalate was much more rare, comprising only 2% of uroliths submitted to the University of Minnesota Urolith Center.6 Since that time, reformulation of commercial diets is thought to have altered the distribution of urolith composition and by 2002 calcium oxalate comprised 55% of feline uroliths, whereas struvite made up 33%.6 Since 2002, struvite uroliths have increased again and are now more common than calcium oxalate in the US, with the more recent decrease in calcium oxalate being most likely due to further reformulation of commercial diets.6 In Canada, analyses of over 11 000 cystoliths submitted from 1998–2008 revealed a different distribution, with calcium oxalate being slightly more prevalent than struvite.7 Overall, struvite and calcium oxalate comprised 92% of feline cystolith submissions. Male domestic cats were 1.4 times more likely to develop calcium oxalate versus struvite cystoliths, whereas females were 1.3 times more likely to develop struvite versus calcium oxalate cystoliths.7 Other cystoliths that have been reported in cats include calcium phosphate, xanthine, silica, cysteine, sodium pyrophosphate, and dried solidified blood calculi. Interestingly, 85% of cystoliths from Egyptian Mau cats were urate.7 While there is currently no effective method to dissolve calcium oxalate cystoliths, dietary management is instrumental in preventing or reducing recurrence. Administering oral citrate and feeding a non-acidifying, high-moisture diet formulated to avoid excessive protein, calcium, oxalate, and sodium minimizes cystolith formation.8 In contrast, medical dissolution of struvite cystoliths in cats can be very effective. Most sterile struvite cystoliths will dissolve within four weeks when feeding an acidifying, high-moisture diet with reduced magnesium.9 Because sterile struvite cystoliths can be successfully managed with medical therapy, it is important to determine cystolith composition prior to instituting therapy. Urine sediment examination for crystalluria, urine pH, and radiographic appearance of cystoliths provides valuable information for predicting the composition. Dissolution diets should be fed for one month past radiographic resolution of cystoliths. Infected struvite cystoliths require substantially longer dietary and antimicrobial therapy for dissolution. Once dissolution is achieved, cats should remain on a urolith preventative diet for life. Physical removal of cystoliths leads to a rapid resolution of clinical signs, definitive diagnosis of cystolith composition, and reduced risk of urethral obstruction. Removal options include voiding hydropulsion, cystoscopic basket retrieval, laser lithotripsy, cystotomy (see Box 37-1), laparoscopic-assisted cystotomy, and laparoscopic-assisted cystoscopy. Regardless of the removal method, postoperative imaging should be performed to document removal of all cystoliths. Voiding urohydropulsion is generally effective for stones with diameters <3 mm in females and <1 mm in males.10 It is also more likely to be effective for smooth, rather than irregularly marginated stones. General anesthesia is recommended for hydropulsion, with the main contraindications being stones that are too large to pass through the urethra, recent bladder surgery, and active urinary tract infections. If infection is present, the increased pressure on the bladder wall can potentially lead to vesicoureteral reflux of urine and pyelonephritis. For this reason, prophylactic antibiotics are recommended for five days following the procedure. Cystoscopic basket retrieval can be used to remove cystoliths smaller than the diameter of the distended urethra. This technique is generally limited to females, or males with a perineal urethrostomy. When cystoliths larger than the diameter of the urethra are present, cystoscopic laser lithotripsy with a holmium:yttrium-aluminum-garnet laser can be used to fragment the cystoliths.11 Laser lithotripsy has been shown to be safe and effective in cats;11 however, when large numbers or sizes of cystoliths are present, the time necessary to remove all cystoliths makes surgical intervention a more efficient method of removal. Complications with cystotomy are rare, and several minimally invasive approaches to surgical removal are available and are discussed later. Bladder rupture most commonly occurs following blunt trauma, but can also result from other types of penetrating trauma, unalleviated urethral obstruction, or iatrogenically from catheterization or bladder expression.12 Traumatic ruptures occur most frequently at the apex and may be associated with pelvic fractures, although the condition is not recognized to the same extent as in dogs. A ruptured bladder does not necessarily prevent normal urination, thus making it difficult to diagnose based on clinical signs alone. Similarly, the presence of a palpable bladder does not eliminate the possibility of rupture since 20% of cats with a ruptured bladder had a palpable bladder on abdominal palpation.12 Peritoneal fluid from cats with a ruptured bladder is usually serosanguinous, and does not always resemble urine. Consequently, biochemical analysis of fluid for creatinine and potassium levels, in addition to routine cytology, protein level, and cell counts, are essential. In a study of 26 cats with uroperitoneum, the mean serum-to-abdominal fluid ratio for creatinine was 1 : 2 (range, 1 : 1.1 to 1 : 4.1). Similarly, the mean serum-to-abdominal fluid ratio for potassium was 1 : 1.9 (range, 1 : 1.2 to 1 : 2.4).12 While the serum-to-abdominal fluid creatinine and potassium ratios may be lower in cats than dogs, they remain clinically useful for diagnosis of uroperitoneum. The location of a suspected urinary tract rupture should be confirmed with contrast imaging such as an excretory urogram, positive contrast cystourethrogram, or cystoscopy (Fig. 37-7). Surgical correction of bladder rupture is rarely an emergency, and care should be taken to stabilize concurrent injuries and correct any azotemia or hyperkalemia prior to anesthesia. For small tears or punctures, maintaining the bladder in a decompressed state with an indwelling catheter for seven to ten days is generally sufficient time for small defects in the bladder wall to heal and has been used successfully in both cats and humans.13,14 However, most tears require visual assessment and debridement, and surgical exploration and repair is indicated if any doubt exists as to the extent of the injury. Neoplasia of the bladder is relatively rare in cats and is nearly always primary in origin. Males are markedly overrepresented, accounting for 65–75% of all cases,15,16 and a predisposition for older cats exists, with a median age of 15.2 years in a retrospective study of 20 cats.15 Hematuria is the most common clinical sign associated with bladder neoplasia, although any lower urinary tract sign may be encountered. Between 57 and 75% of cats have positive urine cultures at the time of diagnosis;15,16 therefore, urine culture should be performed in cats suspected of having bladder neoplasia, and therapy instituted based on antimicrobial susceptibility testing. No correlation has been shown between feline bladder cancer and positive feline leukemia status.16 Definitive diagnosis can occasionally be made from cytologic examination of samples obtained from urine sediment, traumatic catheterization, or percutaneous fine needle aspirates. However, cytologic diagnosis is often complicated by the presence of reactive transitional epithelial cells, which can be impossible to differentiate from neoplastic cells, making cytologic examination of urine sediment diagnostic only 30% of the time.17 Percutaneous aspiration of bladder masses is controversial and should be done with caution due to the risk of seeding the abdomen or body wall with neoplastic cells. Other diagnostic techniques should be attempted first, since implantation of neoplastic cells following needle aspirate of the bladder has been reported in the cat. In humans, it is reported to occur in 1 in 10 000 cases.15,18 Approximately 20% of cats have evidence of metastatic disease at the time of diagnosis of transitional cell carcinoma.15 Therefore, staging for both local and distant metastasis with ultrasound, thoracic radiography, or CT is important prior to surgical intervention. Rectal examination should also be performed to detect evidence of urethral extension or sublumbar lymph node involvement. To obtain a definitive diagnosis, histologic examination of cystoscopic or surgical biopsy samples of the tumor (and regional lymph nodes) is recommended. Carcinoma is the most common urinary bladder cancer. In a review of 27 cats with bladder neoplasia, 15 were diagnosed as carcinoma, five were sarcoma, five were benign leiomyomas or fibromas, and two were lymphoma. Transitional cell carcinoma accounted for 30% of all neoplasms.16 In a study of 23 cats with bladder neoplasia examined at two American universities between 1990 and 2004, 20 were diagnosed with transitional cell carcinoma.15 Over 50% of these tumors were located in an area other than the trigone (Fig. 37-8). Similarly, in another study, tumors were more commonly located on the ventral aspect or apex.19
Bladder
Surgical anatomy
General considerations
Diagnostic procedures
Radiography
Ultrasonography
Cystoscopy
Surgical diseases
Calculi
Trauma
Neoplasia
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Bladder