Chapter 13 Basic Approach to Veterinary Care
Housing
Keep a supply of good-quality feed available for hospitalized rabbits. Rabbits can be finicky eaters, so check with the owner before hospitalizing a rabbit to find out what foods the rabbit prefers. If the diet is of poor quality, it may be necessary to offer the rabbit some of the food to which it is accustomed while gradually introducing a more appropriate diet. A rapid change of diet, even a change from a poor diet to a proper one, may cause gastrointestinal upset and anorexia (see Chapters 14 and 15).3
Fresh water should always be available. Consult the owner to learn what type of watering system the rabbit uses, although rabbits easily learn to drink from sipper bottles. If water crocks are used, they should be made of a heavy ceramic so that they will not easily be tipped over. Bowls with high sides are recommended because rabbits tend to hang their dewlaps in the water when they drink. If the sides of the bowl are too low, this chronic wetting can lead to “wet dewlap” disease, which is an easily preventable moist dermatitis most often associated with colonization by Pseudomonas species. All water containers should be cleaned daily.
If possible, hospitalize rabbits in an area separate from potential or perceived predators. Sounds and smells from hospitalized dog, cat, or ferret patients may stress sensitive rabbits.7 Care should be taken to avoid overheating hospitalized patients. Warm areas appropriate for sick birds or reptiles may cause heat stress in euthermic or hyperthermic small mammal patients.
Handling and Restraint
The rabbit’s skeleton represents only 7% to 8% of its body weight (as opposed to 12%-13% in cats).8 With powerful musculature on the hind limbs and the delicate nature of the skeleton, rabbits are vulnerable to fractures of the back and hind limbs. Rabbits are easily stressed and care should be taken to reduce stress to the patient during examination. Proper handling and housing of patients is necessary to avoid injury.
When a rabbit is being moved, support of the hindquarters is vital to prevent injury. Rabbits used to handling can be carried with one hand under the thorax or by holding the scruff, with the second hand supporting the hindquarters (Fig. 13-1). Fractious rabbits should be carried with the head under the handler’s arm so as to minimize stress by covering the eyes; the hindquarters are supported at the same time (Fig. 13-2). Place a rabbit in a cage with its rear facing the back of the cage while supporting the hindquarters to help reduce chances of injury from the rabbit kicking. In examining a rabbit or placing it in a cage, use a nonslip mat to avoid sliding and injury. Control of the rabbit should be maintained at all times during transport and examination.
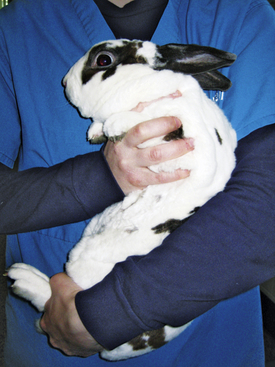
Fig. 13-1 Restrain the rabbit with one hand under its thorax and the other hand supporting its hindquarters.
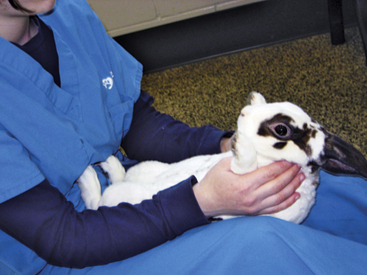
A towel can be used to wrap the patient and cover the head to prevent struggling when the rabbit is transferred from a carrier to a table for examination. In similar fashion, a towel can be wrapped around the rabbit if an assistant is not available to help facilitate physical examination or the administration of medicines (Fig. 13-3). It is important to avoid overheating when a towel is being used to facilitate handling or examination. As an alternative, place your hand over the eyes to calm the rabbit. Rabbits are obligate nasal breathers, so care should be taken to avoid obstructing the nostrils with a towel or hand. Because some rabbits become calm when placed on their backs, the handler can sit on the floor with the rabbit in his or her lap, hindquarters toward the handler’s body. The incisors, abdomen, genitalia, and feet can then be examined (Fig. 13-4). It is preferable for the rabbit to be examined in a room away from “predator” species such as ferrets, dogs, and cats; noises or smells from these animals may stress the rabbit.
Physical Examination
In general, performance of the rabbit’s physical examination is much like that of other species.6 Observe the rabbit at a distance prior to initiating examination. Take note of movement of the rabbit and document any neurologic or musculoskeletal abnormalities. Check for any difficulty breathing and monitor general stress level. Place the rabbit in a quiet, oxygenated cage prior to examination if dyspnea is noted. Thermal support should be provided if the rabbit is hypothermic, taking care to avoid overheating. Although rare, a rabbit may be too stressed to undergo an examination without sedation. A sedative such as midazolam, at a dosage of 0.5 to 1.0 mg/kg IM, is often very effective in calming the animal.4
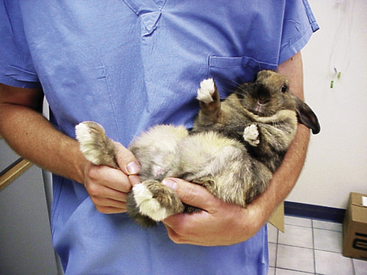
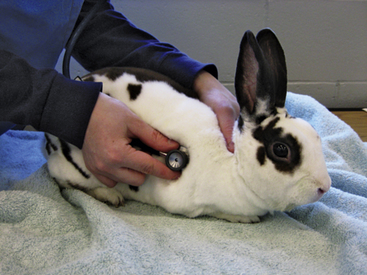
A systematic approach is needed for the examination. Take the body temperature early because the body temperature may increase from the stress of the exam.1 A rabbit can be cradled on its back in a ‘C-shape’ with the hind end supported to allow for examination of the ventrum, trimming of the nails, and taking the animal’s temperature (Fig. 13-5). Avoid using a glass thermometer because it can break if the patient struggles. In examining the rabbit, use a nonslip surface to help keep the animal from slipping and injuring itself (Fig. 13-6). The heart and respiratory rates should be calculated before the rabbit is stressed. It can be challenging though, to differentiate heart and lung sounds on auscultation; respiratory sounds superimposed over the heartbeats may create the false impression of a heart murmur. Note any ocular or nasal discharge in examining the head. The tympanic membrane should be visualized on otic examination. Lymphadenopathy is rare in rabbits, whose lymph nodes are located as in other species. Skin and coat quality should be documented; the plantar surface of the feet should be examined for any signs of pododermatitis.
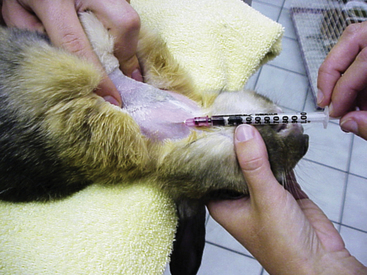
A complete oral examination is an important part of the assessment. Palpate the face and ramus of the mandible for any evidence of swelling or tooth elongation. Although the mouth can be examined without using anesthesia, rabbits are often reluctant to allow access to the oral cavity. Therefore the examination may have to be performed using light sedation or general anesthesia. Lesions may be missed when otoscopic cones are used to examine the mouth of an unanesthetized patient. Nasal or vaginal specula with an attached light source can provide a better field of view than the otoscopic cone. Rabbit mouth gags and cheek dilators are commercially available; however, lesions are occasionally missed in live patients using these instruments. To examine the oral cavity in a sedated patient, place the mouth gag over the incisors to hold the mouth open and insert the cheek dilator. Dental cameras or endoscopy, good lighting, suction, and magnification can further aid in visualization of the oral cavity. Chapter 32 offers more details and sources of instrumentation.
Palpate the abdomen for masses, distention, gas, or other abnormalities. Tensing of abdominal muscles or tooth grinding may be signs of discomfort. A firm, or dough-like stomach, increased gas or fluid within the intestinal tract, and absence of normal intestinal sounds on auscultation may be noted with gastrointestinal disorders.12 Document and investigate any organomegaly.
Sample Collection
Blood Collection
Sites used for venipuncture in rabbits can include the marginal ear veins, central ear artery, jugular vein, cephalic vein, and lateral saphenous vein. Because hematoma formation, bruising, or vessel thrombosis and skin sloughing can result, use of the ear veins and, in some breeds, the central ear artery is not ideal. Use alcohol to part the fur and allow for visualization of the vessel; clipping or plucking of the fur may be useful in some cases. Avoid damaging the delicate skin of the rabbit if clippers are used.
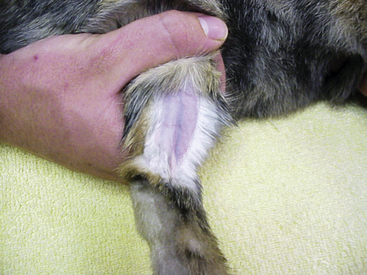
The lateral saphenous vein is an ideal site for venipuncture in the rabbit (Fig. 13-7). If needed, restrain the rabbit in a towel with the head covered while a rear limb is gently extended. The restrainer holds off the vein with pressure across the proximal thigh. The vessel is readily accessible across the lateral surface of the tibia just proximal to the hock. Apply gentle pressure or a pressure wrap briefly over the venipuncture site to prevent hematoma formation after the sample is obtained.
Rabbits have large paired jugular veins, which are often a preferred venipuncture site for very calm or sedated rabbits or when a larger amount of blood (e.g., for blood transfusion) is being collected. However, it is difficult to visualize the jugular vein in overweight rabbits or in females with large dewlaps. For jugular venipuncture, shave the neck over the midtrachea cranial to the thoracic inlet. Position the rabbit with the front legs held over the edge of the table and the head extended up, avoiding overextension of the head. Alternatively, wrap the rabbit in a towel and place it in dorsal recumbency, extending the head and neck to allow for visualization and sampling from the jugular vein (Fig. 13-8).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree