CHAPTER 5 AMPHIBIANS
COMMON SPECIES KEPT IN CAPTIVITY
The class Amphibia is divided into three orders: Anura (frogs and toads), Caudata (salamanders, newts, and sirens), and Gymnophiona (caecilians). There are over 4000 extant species, but very few (one tenth) are ever held in captivity.1
The frogs and toads are the most represented of these orders in captivity. The distinction between frogs and toads, according to most veterinarians, is whether the animals are found in or around water (frogs) or not (toads). Common species in captivity include the firebelly toads (Bombina spp.) (Figure 5-1), African clawed frogs (Xenopus laevis), dwarf clawed frogs (Hymenochirus spp.), ornate horned frogs (Ceratophrys spp.), true toads (Bufo spp.), poison dart frogs (Dendrobates spp., Phyllobates spp., Epipidobates spp.), true frogs (Rana spp.), and tree frogs (Hyla spp.).
Newts and salamanders (grouped as caudates) have gained in popularity as vivarium pets in the past decade. The defining characteristic of a newt versus a salamander is that a newt maintains an aquatic lifestyle throughout both its larval and adult stages.1 The redbelly or firebelly newt (Cynops pyrrhogaster) (Figure 5-2), water dog or tiger salamander (Ambystoma tigrinum), axolotl (Ambystoma mexicanum), mudpuppy (Necturus maculosus), rough-skinned newt (Taricha granulosa), red-spotted newt (Notophthalmus viridescens), and members of the Triturus genus are common to the pet trade. Sirens are a lesser known subset of this order and are not typically held in captivity except in some zoological facilities.
Caecilians are infrequently kept in amphibian collections. When observed in zoos or aquaria, the most common species found are the aquatic Typhlonectes natans (Figure 5-3) and the fossorial Mexican caecilian (Dermophis mexicanus) and Varagua caecilian (Gymnopis multiplacata).1
BIOLOGY
Amphibian body plans are consistent with those of other vertebrates. However, as with any exotic animal species, knowledge regarding the nuances of species variation for some anatomical and physiological traits can be helpful when developing a diagnostic or treatment plan for an amphibian patient. For example, amphibians do not have distinct thoracic and abdominal cavities. Instead, they have a single coelomic cavity. This information is important when considering surgery (e.g., no loss of negative pressure as would be the case with mammals). The following comments on anatomy and physiology will focus on salient clinical features. See other amphibian texts for greater detail.2,3
Unique Anatomy of Frogs and Toads3,4
MUSCULOSKELETAL
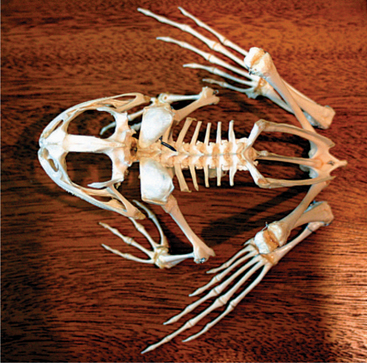
Four limbs are present, but there may be variable hindlimb lengths. The length depends on species locomotory modes; longer hindlegs occur in animals that jump (Figure 5-4). The forelimbs possess a fused radius and ulna (radioulna), whereas the hindlimbs have a fused tibia and fibula (tibiofibula). The forefeet possess four phalanges on each foot, whereas the hindfeet have five phalanges. The hyoid apparatus in some species is adapted to eject the tongue for prey capture. Vertebrae are separated into three fused regions: presacral, sacral, and postsacral (note that a sacrum is not present). The pelvic girdle is fused with the last presacral vertebra, and a fused urostyle (or coccyx) is found caudal to the girdle. Some animals possess pigments that result in a blue coloration of the bones (Phrynohyas resinifictrix). Amphibians do not have diaphragms.
RESPIRATORY SYSTEM
Three modes of respiration occur in anurans: buccopharyngeal, pulmonic, and cutaneous; the mode used depends on species variability and environment. Anurans have a short trachea, thus impacting clinical procedures such as tracheal intubation and washes. Two equally sized lungs are present. The lungs of anurans are thin, sac-like structures. Care should be taken when positive pressure ventilating these animals to prevent pulmonary rupture.
GASTROINTESTINAL SYSTEM
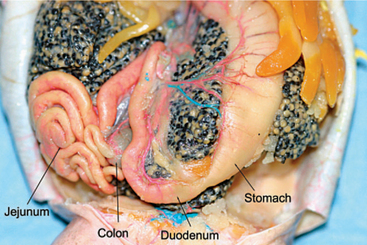
Teeth are absent in some frogs; others lack tongues (e.g., Pipa pipa, which uses negative pressure to create a vacuum to catch prey). Occasionally, frogs evert their stomachs in order to wipe noxious substances from their stomach mucosa. It is not unusual to see undigested insect skeletons, bones, or plant fibers in the feces of anurans. All amphibians (note: within this text, “all” amphibians will include caudates and caecilians) have a simple, short alimentary system that empties into a cloaca (Figure 5-5). A large bilobed liver (which can fill up to 50% of the coelom) encases the pericardium. There are typically two lobes, though a third will occur in some species. The color and shade varies from pale gray to brown to black. Melanomacrophages are present and can result in the dark coloration of the liver. In the larval amphibian, the liver is the primary hematopoietic organ, switching to bone marrow after metamorphosis. Anurans have a large gall bladder.
Unique Anatomy of Newts and Salamanders3,4
INTEGUMENT SYSTEM
In some species of aquatic salamander, there is no keratin cell layer. The dermis is firmly attached to muscle, and therefore there is no subcutaneous layer. Glandular secretions may contain toxic compounds. Folding of the skin and similar anatomical modifications may increase the available surface layer for respiratory purposes.
GASTROINTESTINAL SYSTEM
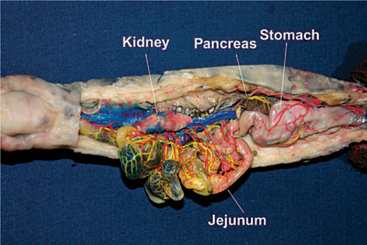
The liver is elongate and single lobed; it may have scalloped edges. The intestinal anatomy of newts and salamanders has no relevant clinical differences from other amphibians (Figure 5-6). The oral cavity hinges such that the maxilla is mobile while the mandible is static.
Unique Anatomy of Caecilians2–5
REPRODUCTIVE SYSTEM
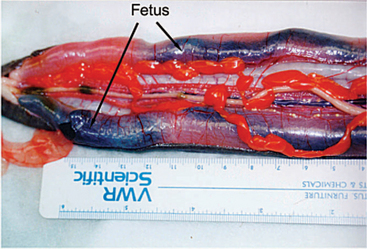
Male caecilians possess a phallodeum (intromittent organ), enabling them to practice internal fertilization. Caecilian ovaries are paired and are closely connected with the kidneys. Viviparous animals have an oviductal lining where embryos feed off the lining (Figure 5-7). Oviparity occurs in most caecilians, with some species-specific parental care occurring until hatching.
Basic Premetamorphic Anatomy2–5
CAECILIANS
Aquatic caecilians are viviparous. Larval animals are born fairly precocious, although they do have gills (Figure 5-8) present for the first day after birth. Oviparous caecilian larvae also have external gills that are quickly resorbed, but they also possess larval fins, which disappear shortly before hatching. The larval caecilian has a pronephric kidney that changes to a mesonephric kidney during metamorphosis. This change alters the kidney’s function from a filtration system to a more complex excretory and water resorptive system.
HUSBANDRY
Illness in captive amphibians is largely due to poor husbandry practices. It is outside the scope of this chapter to discuss the intimate details of building a successful vivarium for all the diverse species. Rather, the emphasis will be on general considerations for housing to educate the clinician in basic requirements and for designing hospital tanks. For information on captive amphibian husbandry, see Wright and Whitaker.6,7 Additionally, examining natural habitat can be exceptionally useful in determining captive husbandry standards.
Environmental Considerations4–8
AQUATIC
Animals that live in ponds versus streams have very different tolerance levels for water quality. Stream-dwelling animals generally have a greater need for more dissolved oxygen and higher quality water. In general, water quality should be maintained as for fish. Table 5-1 provides basic parameters to observe with most amphibians.
Creating a Vivarium
TEMPERATURE
The environmental temperature range provided an amphibian should be based on its natural habitat (Box 5-1).6,7 There should always be a gradient with a range of 5° to 8° C. When changing water, make sure the temperature matches the enclosure. Basking spots are necessary, and ceramic heaters (which produce no light) or spotlights can usually provide the necessary temperatures. Care and diligence are required for appropriate wattage and distance. Spot heaters are best placed outside of the enclosure to avoid trauma via thermal burns. Do not use heat rocks for amphibians.
BOX 5-1 Environmental Temperature Ranges for Amphibians Based on Natural Habitat
| Tropical lowland | 24°–30° C |
| Tropical montane (moist, cool, coniferous strata) | 18°–24° C |
| Subtropical | 21°–27° C |
| Temperate, summer | 18°–24° C |
| Hibernation | 10°–16° C |
| Aquatic, tropical lowland | 24°–30° C |
| Aquatic, tropical montane | 18°–24° C |
| Aquatic, subtropical | 21°–27° C |
| Aquatic, temperate stream (summer) | 16°–21° C |
| Aquatic, temperate pond (summer) | 18°–24° C |
Aquarium heaters may cause thermal burns for caecilians if they wrap around these devices. To prevent this, place mesh or PVC piping around the heater to prevent contact. It is ideal to use a thermometer that dually measures the warmest and coolest spot; the more advanced thermography units allow detection of temperature gradients as well.9 Hibernation is important in some species for stimulating breeding, but it is unknown whether it is necessary for long-term health and success in captivity.6 Many salamander species prefer cooler temperatures,10 and some vivaria might require chillers.
HUMIDITY
A fine mist can be sprayed into the vivarium several times daily (manually or via misting systems). When doing so, distilled or aged water should be used. An air stone in a bowl of water or live plants can also be used to help maintain the environmental humidity. Relative humidity above 70% will suit most amphibians. Tree frogs have a natural behavior to adduct their heads and press against surfaces to retain water—if they do this exclusively, the environmental humidity is not adequate. Conversely, waxy tree frogs may develop dermatitis with excessive environmental humidity.6
LIGHTING
Full-spectrum lights are generally recommended for amphibians, but most do not provide radiation in the wavelengths consistent with vitamin D3 synthesis. Ultraviolet B (UVB) (290-320nm) radiation may be important for vitamin D3 metabolism in amphibians, but the role of UVB radiation has not been fully examined. I prefer to be conservative and recommend the use of full-spectrum lights. Replacement of the bulbs is recommended every 9 months to ensure that the appropriate wavelength is emitted. UVB does not transmit through glass or plastic, and the depth at which it transmits is typically no greater than 9 to 18 inches away from the source. Care must be taken that the lighting does not adversely affect the animals. Low-level nocturnal lighting (moonlight simulation) may be useful for nocturnal species to ensure that the animals are not startled when the lights are turned on or off. Dimming lights gradually is ideal.
SUBSTRATE
Substrate selection is an important consideration for a vivarium. When considering different types of substrates, it is important to try to mimic an amphibian’s natural habitat. Smooth or small pebble gravel may be used as a substrate, but it can become a gastrointestinal foreign body if it is ingested. This can be prevented by using large pieces of gravel (e.g., bigger than can fit in the oral cavity) or feeding animals away from such substrates. Soil substrates should be organically rich and pH balanced (neutral). For burrowing caecilians, soil depth should be 3 to 10 cm.5 For fossorial species, there should be a moisture gradient and the soil should be loose enough to allow for tunnel formation. Soil should be replaced every 2 to 3 months. Substrates that need to be sterilized should be baked. Soil and leaf litter substrates should be sterilized to prevent arthropod and helminth infestation. To sterilize soil, bake at 200° F (95° C) in a thin layer (<2 cm) or place in direct sunlight for several hours. In general, soil substrates are very difficult to maintain. If sand is used, horticultural silver sand is recommended. Sphagnum moss is a useful substrate because it retains moisture, is soft and pliable, and is easily discarded. Sphagnum moss should be changed every 2 to 4 weeks to avoid compacting the substrate and breeding undesirable organisms. Living moss does not usually do well in amphibian vivaria because the water needs of moss cannot be met while maintaining a healthy amphibian. Certain substrates should be avoided. Rotting wood can be a good living substrate, but the risks of introducing disease may outweigh the benefits.6 Vermiculite is not recommended because it can cause a gastrointestinal impaction. Peat moss and manure are not recommended because they are acidic and irritating to the skin. Mulch can be used, but aromatic woods (cedar, pine) should be avoided as they can be toxic and irritating.
ACCESSORIES
It is important to take time and consider what accessories are needed in a vivarium, as these items can be used to mimic an animal’s natural habitat and reduce stress. Overplanting and overfurnishing a vivarium may make prey capture difficult for the animal. Plastic plants can be used to landscape a vivarium and are easily disinfected. Live plants can also be used and have the benefit of increasing environmental humidity. It is important to only use live plants that are known to be nontoxic, as prey being offered to amphibians may eat the live plants and transfer any toxin to the amphibian.6 If live plants are chosen, the pet owner should check to see if harmful pesticides or fertilizers have been used. Live plants may be contaminated with parasitic ova (e.g., from Florida, where tree frogs live in the plants). Recently acquired live plants should be cleaned and their soil should be replaced to reduce the likelihood of contaminating the vivarium with unwanted pathogens. Aquatic plants can harbor snails, which can introduce parasites to a vivarium as they serve as intermediate hosts for a number of parasites.
“Furniture” considerations for a vivarium may include large rocks, branches, shelters, and waterfalls. Traumatic injuries may occur with such items if they are not secured, and all furnishings should have smooth edges. These items should also be conducive to sterilization or disinfection. Bear in mind that porous objects may retain disinfectants. Proper rinsing is critical. Shelter type will depend on the natural camouflage of the animal and should be provided. Such accessories are easily created (Figure 5-9).
NUTRITION
Feeding Mechanisms
Larval anurans are filter feeders.11 Water is pumped through the mouth, and then planktonic algae are extracted and entrapped by a mucus-covered filter in the pharynx and then directed into the esophagus. Adult, terrestrial amphibians use their tongues to capture prey. These animals have glands on their tongues, which produce a sticky substance that helps apprehend prey until it is securely placed in the mouth. Some anurans can shape their tongue into a tube to assist in feeding on ants, termites, and worms. Aquatic salamanders direct prey to their mouths by creating an inward flow of water into the oral cavity. Pipa pipa direct food and water into their mouth with hypobrachial pumping movements. Some aquatic amphibians use their long phalanges to direct the food into their oral cavity.
Feeding and Diet
The list of possible invertebrates to offer amphibians is extensive.12 In general, the most common food items offered to terrestrial amphibians include earthworms, red worms, mealworm larvae, waxworm larvae, crickets, fruit flies, other larvae, occasionally small reptiles (e.g., anoles), and amphibian eggs and larvae. For aquatic species, these same foods are offered, but bloodworms, blackworms, and crustaceans, such as crayfish and brine shrimp, may be offered as well. Some animals have very specific diets, like the termite-eating caecilian (Boulengerula spp.), and can be difficult to maintain in captivity.5 Avoid brightly colored insects,6 as they may be naturally toxic. Finally, although wild-caught invertebrates would provide enrichment and balance for an amphibian, caution must be exercised not to feed out organisms that have been subject to pesticides or other such toxins.
Feeding pelleted diets intended for fish or reptiles may not be appropriate for amphibians, as these diets may be intended for herbivores or omnivores and will not meet the fat, protein, and vitamin requirements of amphibians.12 Rodents can be offered on occasion, but vitamin A levels are often too high in these prey items, and secondary nutritional hyperparathyroidism can occur without supplementation of vitamin D3. On the other hand, oversupplementation with vitamin D3 may cause mineralization of organs. Unfortunately our knowledge regarding vitamin D3 metabolism in amphibians is limited.
Supplementing amphibian diets with vitamins and minerals is a questionable but necessary practice. With a truly well-balanced diet, supplementation is unnecessary and may even cause problems (e.g., hypervitaminosis A and D). Unfortunately, with the limited number of commercially available food items, providing a well-balanced diet is difficult. Based on this limitation, it is best to offer the largest variety (e.g., multiple prey species) of food types to provide the best nutrition. Most supplements target calcium and vitamin D3 in an attempt to balance the Ca:P ratio (1.5 : 1 or 2 : 1). Contrary to that target, most invertebrates, with the exception of earthworms, have an inverse Ca:P ratio.12 Invertebrates must therefore be “gut-loaded” with vitamins and minerals by feeding them a diet that is high in Ca and low in P (available commercially) in the 24 hours before offering that prey item. Some of these diets can result in death of the prey item if fed longer than 24 hours. Additionally, these diets are not balanced for invertebrates and should not be used long term. Another product used for increasing the nutritional value of invertebrates is to “dust” them with a supplement. Placing the supplement and invertebrates in a brown bag and gently shaking the bag will result in the invertebrate being covered with the supplement. Dusting can be used with small invertebrates where gut loading is not practical or lethal, and this should be done immediately before feeding.12 Dusting larger invertebrates with these supplements does not successfully achieve the calcium levels that gut loading does. When feeding frozen fish, it is necessary to supplement the diet with thiamine as the fish produce natural thiaminases that destroy the thiamine in the tissues.
PREVENTIVE MEDICINE
Disinfection and Sanitation
Utensils, accessories, enclosures previously occupied by amphibians, and enclosures with known disease entities, should be disinfected to prevent the spread of disease. Removal of all debris is necessary before application of a disinfectant, as some products are deactivated in the presence of proteins and other organics. Warm water and surfactants can be used to help break down organics during the cleaning process. Numerous classes of disinfectant are available, each with variable effectiveness against certain organisms (Table 5-2). Some products absorb the chemicals and can leach out while the amphibian is present (e.g., iodine products). Ammonia and chlorine bleach are good general disinfectants. With all chemical products, thorough and repeat rinsing is mandatory. For most disinfectants, a minimum contact time of 10 minutes is recommended. For more fastidious organisms, longer contact times are recommended. Porous objects should be discarded rather than reused, not only because they harbor pathogens but also because it is difficult to thoroughly rinse and remove the chemicals from these materials. Humidifiers should be disinfected weekly, as should any aerosolizing tools. Povidone iodine, chlorhexidine, isopropyl alcohol, and quaternary ammonium compounds are known to cause skin lesions and to be toxic in amphibians at high concentrations, and thus should be used with caution.13
RESTRAINT
When handling amphibians, moistened, wet gloves should be worn to minimize the likelihood of damaging the animal’s skin. If gloves are not worn, then hands should be rinsed with aged or dechlorinated water. As an indictment to the delicacy of amphibians’ skin, smokers who handle smaller frogs can cause them to die, due to the nicotine residue on the smokers’ skin.14 Preparing water to use with amphibians is easily accomplished by retaining tap water in a bucket of water and aerating it with an air stone for an hour. The water can also be passed through charcoal to remove contaminants. Alternately, water can be dechlorinated by allowing it to degas in a nonaerated reservoir for 48 to 72 hours. After handling amphibians, clinicians should thoroughly wash their hands, as many species produce toxins that can irritate human skin and mucosa. Gloves should be changed between handling animals to decrease disease transmission and toxin transmission.
Manual Restraint15
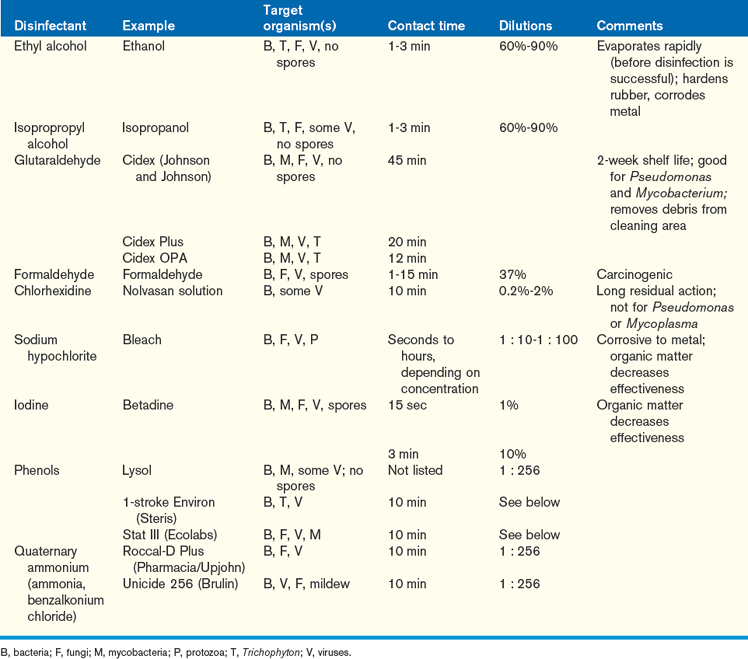
Large frogs should be handled by firmly grabbing the animal around the inguinal region just cranial to the hindlimbs (Figure 5-10, A). Smaller frogs can be cupped into the palm of the hand for limited access or, while in this grip, gently restrained by the hindlimbs and caudal coelom using the opposing hand. When handling an animal in this way, it is important to take care not to squeeze or traumatize the animal (Figure 5-10, B). Expect anurans to urinate when handled, and be prepared to collect the sample if desired. Additional behavioral manifestations identified after handling may include breath holding or air sac insufflation, color change, and vocalizing.
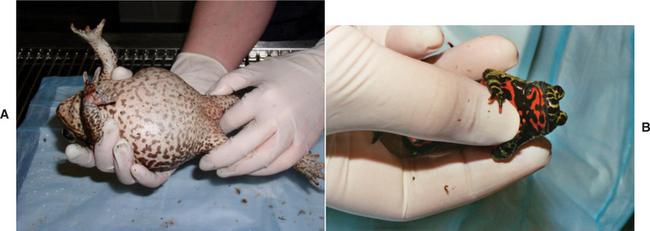
Salamanders and newts should be handled at the pectoral girdle initially, then the hindlimbs (Figure 5-11). Tail autotomy can occur in these animals, and care should be taken to not hold the animal by its tail.
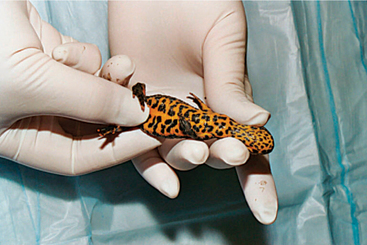
Caecilians are very difficult to manage with manual restraint. Clear tubing or a plastic bag can limit the mobility of these animals and facilitate examination (Figure 5-12). For a more thorough examination, the animal should be sedated.
Chemical Restraint
The most common general anesthetic agent used for amphibians is tricaine methane sulfonate (MS-222) (Finquel, Argent Chemical Laboratories, Redmond, WA). The drug comes in a powdered form and is usually reconstituted in distilled water. MS-222 acts by reverting transient sodium ion permeability and decreasing excitability and blocking conduction of nerve impulses.16 This compound is very acidic and should be buffered with an equal volume of baking soda to stabilize the pH. Unbuffered MS-222 can be highly irritating to the amphibian, causing excitability and erythema. Unbuffered MS-222 can also result in a more rapid induction because uptake across the skin is more rapid in the ionized form. Although buffering the agent will result in slower uptake, it is far safer for the animal (e.g., hyperglycemia and tachycardia have been reported with unbuffered MS-222).17 Induction at 100 to 200 mg/L will achieve steady state levels in most amphibians, but induction may be prolonged for more than 25 minutes. Higher doses (1-5 g/L) may induce anesthesia faster, but these levels are considered potentially lethal. Higher dosing levels can also prolong recovery. At induction, animals may try to jump out of the solution, as it can be irritating. Anesthetic deaths are rare with MS-222, but they are possible. Under MS-222 anesthesia, pulmonary ventilation is diminished or absent. Gas exchange across the skin is usually adequate to prevent hypoxia at low (<74° F) environmental temperatures. Once anesthetized, the animal’s nares and oral cavity must be kept out of the water to avoid drowning. Keeping the animal at room temperature (74°–78° F) usually results in acidosis and hypercarbia. Oxygen can be aerated into the immersion solution, and this usually helps the animal compensate. MS-222 has been used intramuscularly (IM) and intracoelomically (ICe); however, I have not used these routes. When using these alternative routes, expect prolonged recoveries.
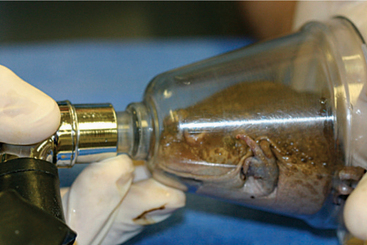
Gas anesthesia has been used in a variety of forms.16–18 Using the vaporized form results in direct exposure to the inhalant, as with a face mask (Figure 5-13), and can induce hypothermia and/or dehydrate the animal. Therefore, when using this form of anesthesia, it is important to monitor the animal’s hydration status and body temperature. Using direct exposure appears to be more successful in toad species than frog species. Dosing anurans percutaneously with topical isoflurane and halothane has also been successfully done, but it can induce a very deep plane of anesthesia and prolonged recoveries. Although some clinicians report that aerating an inhalant directly into water is useful, I have not had success with this method. Another potential complication with aerating the anesthetic into the water is that scavenging excess gas may not be possible, posing a human health risk. Finally, a gel mixture of a sterile KY jelly lubricant with isoflurane or sevoflurane and water mixed in a 3 : 3:1 ratio can be utilized. The mixture may be applied directly on the animal or via a gauze (Figure 5-14). This method results in rapid induction and recovery.

Figure 5-14 Anesthetic induction of a frog using a gauze soaked in isoflurane and water-soluble gel.
Clove oil (eugenol) has been used to anesthetize leopard frogs (Rana pipiens) and tiger salamanders (Ambystoma tigrinum).19,20 The dose required to induce surgical anesthesia in the salamanders (0.23 ml of 85% clove oil/500 ml dechlorinated water) is much higher than in the frogs (333 mg/L). Propofol was also evaluated in both species. Delivery in the frogs via the lingual vessels was difficult and produced only mild effects. When Propofol was administered intracoelomically in the tiger salamanders (35 mg/kg), surgical anesthesia was achieved in the majority (73%, 8/11) of test subjects.20
Monitoring the Anesthetized Patient
The amphibian heart rate can be monitored using a pulse oximeter, Doppler, or ultrasound. Respirations can be difficult to monitor. The best methods to use are to evaluate the excursion of the rib cage or the gular area. Oxygen supplementation is important, as hypercarbia and hypoxia are common during anesthesia. However, too much oxygen may limit respirations, based on the theory that these animals are stimulated to breathe based on low oxygen levels and not high carbon dioxide levels. Based on this, it is generally preferred to recover these animals on room oxygen (21%). Readings of 95% oxygen saturation or greater are expected in a normoxic patient.15 If bradycardia occurs, reduce or end the anesthetic event. Atropine (IM or IV) can be used to increase the heart rate. If asystole occurs, begin cardiac compressions. Cardiac compressions can be used to re-stimulate the cardiac function. There are two methods that may be used: (1) Place the animal in dorsal recumbency. Then place an index finger on the cranioventral rib cage (e.g., level of the pectoral girdle) and compress downward. This will press the heart against the spine and act as a stimulus to restart the heart. (2) Grasp the lateral rib cage at the level of the pectoral girdle and squeeze inward. Epinephrine can be given intratracheally, intracardiacally, or intravenously to initiate cardiac function.
When monitoring the depth of anesthesia in amphibians, it is important to follow a consistent pattern and record all measurements. The following measurements are generally used: righting reflex, escape response, palpebral reflex, superficial pain response (e.g., pinch skin over dorsum of foot), and deep pain response (e.g., apply pressure to one of the rear digits). It is important to evaluate those reflexes before starting the anesthetic event, to provide a baseline for that animal. The generally accepted method for characterizing these reflexes is based on an ordinal scale: 0-present, 1-mild loss, 2-loss. Based on this scale, surgical anesthesia is achieved when all of the reflexes are recorded as a 2 (loss).
PERFORMING A PHYSICAL EXAMINATION
Before handling an amphibian, it is important to consider the role of handling on the production and removal of lactate. If an animal is repeatedly handled, lactate may accumulate, and it can take a long time for this compound to be removed from the amphibian body.3 In severe cases, an amphibian with excess lactate levels may collapse or develop a myopathy. To prevent this, restrict handling times, and be prepared to perform an examination and collect diagnostics at the same time.
An ocular examination can be performed using an ophthalmoscope and slit lamp to examine the cornea, iris, and anterior and posterior chambers.21
An oral exam can be accomplished using a variety of tools as potential specula (Figure 5-15). Such tools may include radiograph film that is cut and rounded at the edge, standard metal bird specula, guitar picks, a rubber spatula, and credit cards.22 Animals may bite, and therefore caution must be exercised when examining these animals. Salamanders use their maxilla to hinge upward rather than downward at the mandible, which is the opposite of what is observed in most amphibians. Overmanipulation of an amphibian’s jaws can lead to fractures, so care should be exercised. The oral cavity of an amphibian should be moist and free of thick, ropy mucus. Excessive mucus often suggests dehydration. Look closely in the oral cavity for fractured teeth and oral abscesses.
Coelomic palpation can provide significant information regarding an animal’s condition. Some animals will “puff up” when in a defensive mode, which can make palpation challenging, but patience will often result in success. After palpating the coelomic cavity, muscle tone and body condition should be assessed. With poor body condition, the clinician must examine the environment and the animal’s caloric intake and pursue diagnostics for chronic infectious/parasitic diseases (recurrent bacteremia/septicemia, mycobacteriosis, and parasitism). Skeletal confirmation should be evaluated and the long bones and rostrum palpated to assess for metabolic bone disease (e.g., secondary nutritional hyperparathyroidism).
Gender determination can be challenging, but it should be done at the time of physical examination. Sexual dimorphism can occur, but it is not present in most of the species found in captivity. Transillumination of the abdomen may reveal eggs (Figure 5-16). In male salamanders, the base of the tail may become enlarged.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree