Vanessa L. Cook
Adhesions
Intraabdominal adhesions develop as a result of peritoneal inflammation and are likely to develop in many horses after exploratory laparotomy. However, the true incidence of adhesions is difficult to assess because some horses may have adhesions without showing clinical signs.
Adhesions usually manifest as signs of recurrent colic because they form a simple, partial obstruction of the intestinal lumen that restricts the free flow of ingesta through the gastrointestinal tract. Colic signs arise as a result of distension and stretching of intestinal segments orad to the adhesion. The segment of intestine orad to the adhesion usually becomes chronically dilated and hypertrophied. An estimate of the incidence of clinically significant adhesions can be obtained by documentation of their presence in horses that are sent for necropsy subsequent to abdominal surgery, and horses that undergo repeat laparotomy. Data from such studies suggest that approximately 10% to 20% of postoperative colic patients have adhesions. The incidence of adhesions has been found to be increased when an enterotomy or resection is performed, after small intestinal surgery, in horses that have undergone multiple laparotomies, when postoperative ileus develops, and after incisional infection. The common inciting pathway behind all of these risk factors is an increase in peritoneal inflammation. The location of adhesions varies with the site of the initial surgical lesion. After small intestinal surgery, adhesions usually form between the serosal surfaces of adjacent loops of small intestine. They also frequently form between the small intestine and the mesenteric stump, or at the anastomosis site. In horses that have had large colon surgery, adhesions most frequently form between the colon and the small intestine, the spleen and the ventral body wall, or the colon and the omentum.
Pathophysiology
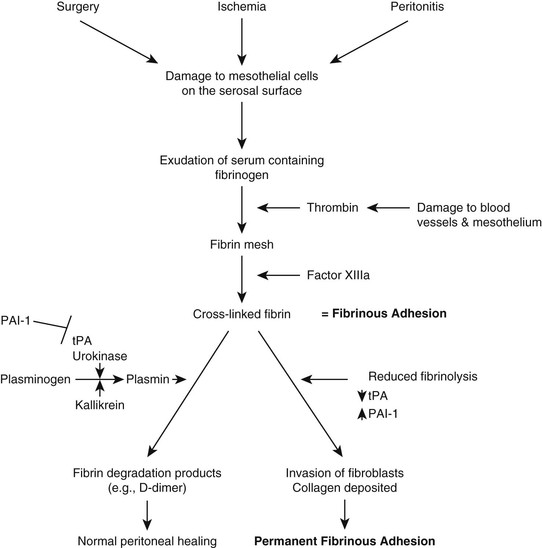
Adhesions develop when the peritoneal mesothelial cells are inflamed. Inciting causes of inflammation include surgery, ischemia, and septic peritonitis. This results in the exudation of serum, which contains fibrinogen, onto the surface of the intestine. Fibrinogen is converted to soluble fibrin by thrombin, and then to insoluble cross-linked fibrin by clotting factor XIIIa (fibrin stabilizing factor) to form a fibrinous adhesion (Figure 78-1). With a fibrinous adhesion, the intestine becomes adherent to another structure, but it can be physically pulled apart. Under normal conditions, these fibrinous adhesions are removed by fibrinolysis. The key to fibrinolysis is activation of the protease plasmin. The inactive precursor of plasmin is plasminogen, which is a component of plasma protein. Several mediators activate plasminogen, including tissue plasminogen activator (tPA), which is released from damaged tissue; urokinase; kallikrein; and clotting factors XIa and XIIa. After plasminogen is activated to plasmin, it cleaves fibrin into fibrin degradation products such as D-dimer. Thus most fibrinous adhesions are resolved by this mechanism. The system is kept in check by mediators that inhibit fibrinolysis. Plasminogen activator inhibitor-1 (PAI1) is an endothelium-derived protein that inhibits tPA and urokinase and therefore inhibits fibrinolysis. α2-Antiplasmin and α2-macroglobulin inhibit fibrinolysis by forming a complex with plasmin to remove it from the circulation. Problems arise when there is disruption in the balance of these mediators. This has been documented with sepsis and inflammation, in which tPA, plasminogen, and urokinase decrease, and PAI1 increases, all of which results in a decrease in fibrinolysis. For this reason, fibrinous adhesions tend to persist instead of being broken down. An influx of fibroblasts occurs, and collagen is deposited in the fibrinous adhesion, resulting in formation of a permanent fibrous adhesion.
Foals have a higher risk for forming adhesions than do adult horses, with adhesion rates as high as 33%. The reason for this is unclear. A recent study by Watts and colleagues measured fibrinogen concentration, plasminogen activity, antiplasmin activity, and D-dimer concentration in blood and peritoneal fluid in foals (≤6 months old) and adult horses (≥5 years old) with colic and compared them with samples from foals and horses without colic. The authors hypothesized that in samples from foals, fibrinolysis would be impaired, indicated by low plasminogen activity and D-dimer concentration, with or without increased fibrin deposition, indicated by high antiplasmin activity and fibrinogen concentration. Samples from colic cases did have significant differences in all four variables that would indicate impaired fibrinolysis and increased fibrin deposition; however, there were no significant differences between adults and foals. Therefore differences between adult horses and foals in fibrinolytic activity can be ruled out as a reason for increased adhesion formation in foals.
Prevention of Adhesions
Minimize Peritoneal Inflammation
Celiotomy is one of the main initiators of peritoneal inflammation and subsequent adhesion formation. Surgical correction of adhesions after they have formed usually results in formation of further adhesions. Therefore the goal must be to try to prevent their formation in the first place. Reducing peritoneal and serosal inflammation is critically important because this reduces the exudation of fibrinogen. One of the most important steps in ensuring that Halstead’s principles are followed during surgery is that the bowel should be handled carefully and kept moist at all times with a physiologically balanced electrolyte solution. When enterotomy is performed, or resection and anastomosis, steps should be taken to minimize contamination and ensure that the mucosa is inverted. Perioperative treatment with antimicrobials and nonsteroidal antiinflammatory drugs for 72 hours is a critical part of reducing adhesion formation. In one study, treatment with flunixin meglumine, penicillin, and gentamicin resulted in no adhesions forming in an ischemia-induced foal adhesion model. In that same study, administration of a low dose of DMSO (20 mg/kg, IV, every 12 hours) also prevented adhesion formation. This dose is substantially lower than the 1-g/kg dose that is frequently used for the treatment of increased intracranial pressure. This high dose has been found to exacerbate ischemia-reperfusion injury, whereas the lower dose may have antiinflammatory effects.
Disagreement exists among surgeons over the use of carboxymethylcellulose during exploratory celiotomy. Some believe that the application of 1% carboxymethylcellulose to the surface of the intestine helps reduce serosal trauma during handling of the intestine and also prevents ingesta and bacteria from sticking to the intestine when an enterotomy is performed. Carboxymethylcellulose may also have a direct antiinflammatory effect. A study by Fogle and colleagues revealed that intraoperative use of 500 mL to 1 L of carboxymethylcellulose reduced the risk for death, especially in horses that developed postoperative colic or ileus. The authors speculated that this was a result of early reduction in adhesion formation, although neither supporting necropsy nor repeat laparotomy results were presented to support this supposition. Other reports suggest that the commercially available form of carboxymethylcellulose may cause peritoneal inflammation and substantially increase adhesion formation. Taking all the reports into consideration, carboxymethylcellulose may have some beneficial effect in reducing adhesion formation, but excessive volumes appear to cause peritoneal inflammation, so it should be used in moderation.
Enhance Fibrinolysis
Another important step in preventing the formation of clinically significant adhesions is to promote fibrinolysis so that fibrinous adhesions that do form are resolved. The treatment most commonly used to reduce fibrin formation is heparin. Heparin is a cofactor for antithrombin III (ATIII), which reduces thrombin concentrations by binding it in thrombin–antithrombin complexes. Heparin changes the conformation of ATIII, which improves its ability to bind thrombin. Thrombin usually converts fibrinogen to fibrin, and that step is thus potentially reduced when heparin is present, subsequently decreasing the number of fibrous adhesions that can form. For heparin to be effective there must be adequate circulating levels of ATIII, which is frequently reduced in postoperative colic patients. Intravenous administration of fresh frozen plasma will provide a source of ATIII, and if possible, its serum concentration should be measured. Heparin is most frequently administered at a dose of 40 IU/kg, subcutaneously, every 8 hours. However, intraperitoneal heparin (30,000 IU in 1 L of saline, poured into the abdomen at the end of surgery) may be more effective and in one study significantly decreased the adhesion rate to 2.8%, compared with 17% in horses that were not treated with intraperitoneal heparin.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree