Chapter 33 Virology of Nonavian Reptiles
An Update
Reptile virology is a rapidly changing field, with many ongoing exciting discoveries. We are still learning about the diversity of infectious agents in reptiles and the clinical significance of these agents. This chapter is intended solely as an update on recent developments regarding viruses associated with the most significant pathology, and not as an overview. For overviews, the reader is directed to earlier reviews.11,27
What is a Reptile?
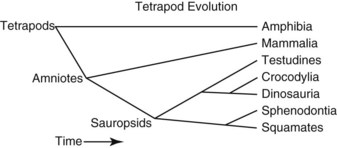
When the evolution of the terrestrial vertebrates (tetrapods) is examined, the fossil record and far more complete and convincing nucleic acid sequence phylogeny analyses are in agreement on relationships.9 The earliest divergence among the tetrapods is between the amphibians and amniotes, who are fully terrestrial (Fig. 33-1). The amniotes are then further divided into mammals and sauropsids. Within the sauropsids, the next group to diverge is the squamate (lizards and snakes)–sphenodontid (tuatara) clade. Following this, the Testudines (turtles) diverged, and the last two major sauropsid groups to diverge were the crocodilians and dinosaurs. The only surviving group of dinosaurs is the birds. This is supported by the fossil record and by sequence data.23 Although birds themselves constitute a monophyletic group, if birds are not considered as part of the reptiles, then reptiles are not a monophyletic group. What we really mean by reptiles is sauropsids, and birds are a group of reptiles. Crocodilians and birds share a number of medically relevant similarities, including a four-chambered heart with the right aortic arch being the major outflow for oxygenated blood, and lungs with unidirectional rather than tidal air flow.5 However, the significant differences between birds and crocodilians are also obvious and the clinician needs to be careful not to overextrapolate. The obvious differences between a caiman and sparrow underscore the more cryptic but greater differences between a caiman and monitor lizard.
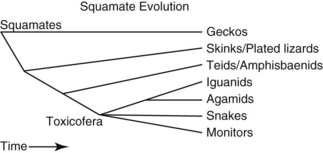
Another common error is the idea of lizards as a group distinct from snakes. In squamate evolution, the earliest divergence is the geckos, followed by the divergence of the skinks, night lizards, plated lizards, and girdled lizards (Fig. 33-2). The next groups to branch off were the teiids, lacertids, and amphisbaenids and the remaining group, containing snakes, iguanids, agamids, chameleons, monitors, helodermatids, and anguids, is known collectively as the Toxicofera, named for the commonality of the presence of venom glands.6 Snakes diverge in the middle of the squamates and, if snakes are removed, then lizards are not a monophyletic group. Snakes are a group of lizards and a ball python is a better model for an iguana than a leopard gecko.
Novel Viruses
Because reptile virology is still a nascent field, it is not uncommon to find previously unknown viruses. Assessing risk and responding appropriately to the identification of a novel virus requires understanding of context, viral ecology, and evolution. In an ideal situation, we would have information including the results of experimental infection with the virus in a set of animals from the species of concern. This is rarely the case. However, even if it is, it is important to recognize that Koch’s postulates represent a concept that should not be overinterpreted. No infectious agent is either a pathogen or not a pathogen. There have been many asymptomatic human Ebola virus infections, and people have died of septicemia caused by Lactobacillus acidophilus.4,14 It is important to correlate infection with clinical patient evaluation and histology before infectious disease is considered as a significant differential. All clinical infectious diseases have multiple contributing factors, including those from the infectious agent, the host, and the environment.
Another important viral factor with clinical implications is genome organization; some viruses have segmented genomes. Throughout biology, hybridization is a factor allowing rapid nondetrimental change, allowing species to invade novel habitats.20 This is considered the main reason for the evolution of sex. Segmentation of viruses, allowing reassortment, is essentially the viral equivalent of sex, providing a hybrid advantage for crossing host species. This has been best studied in the Orthomyxoviridae.
Because of lack of proofreading by their polymerases and constant selection by the host immune system, RNA viruses have the fastest mutating genomes found in nature, and tracking evolution of these viruses is highly clinically significant. One recent meta-analysis has found that of the 20 virus families infecting humans, four RNA virus families—Reoviridae, Bunyaviridae, Flaviviridae, and Togaviridae—accounted for more than 50% of emerging and reemerging viruses.31 Emerging disease is also frequently associated with host switches. The human disease meta-analysis found that 816 of 1407 (58%) are zoonotic and, of human diseases, zoonotic diseases are significantly more likely to be emerging. The simpler nature of smaller RNA viruses and their ability to change rapidly makes them more adaptable to new hosts. This ability to move successfully and rapidly to new hosts removes the selective pressure not to harm the host, and RNA viruses generally tend to result in more explosive epidemiology.
There is generally little that a clinician may do about host factors when dealing with novel viruses, other than to advise avoiding mixing species and resultant aberrant host infections. The areas that the clinician can affect most are environmental factors. Many factors, including but not limited to coinfections, stress, nutritional status, temperature, reproductive status, age, and noninfectious disease processes, all play significant roles in viral disease. Of these, temperature is the area that differs most in nonavian reptiles from what is seen in mammals. Poikilothermy provides the patient with the ability to tolerate a wider range of body temperatures; viruses often tolerate a narrower temperature range, or at least disease may only manifest in a narrower temperature range. Patients with a wide thermal gradient may choose a febrile or other thermal response.17 Providing appropriate thermal opportunities is often a highly effective therapy.
Adenoviruses
The genus Atadenovirus is endemic in squamates, although some species have undergone host switches at some point into birds and mammals.28 These viruses are fairly host-specific and infection is often only seen in the endemic host, in which clinical disease most commonly manifests as hepatitis and enteritis and is most often seen in young animals. Asymptomatic infection is not uncommon. Bearded dragons and king snakes are commonly recognized as clinically affected species.8 A growing diversity of squamate atadenoviruses is being recognized,18 and it is likely that most squamate species may have associated atadenovirus species. Persistence appears to be common and, given the environmental stability of adenoviruses, infection tends to become widespread within collections unless highly rigorous biosecurity is practiced. Given the tendency of reptile breeders to have large collections at high density, acquire reptiles through venues such as trade shows, and practice poor biosecurity, atadenoviruses are ideally suited to become widespread in captive populations. A study of typing of Agamid adenovirus 1 in North American collections has found that different genotypes are widespread across geographically separate collections.19
The genus Siadenovirus has been associated with the most dramatic pathology of any known virus in tortoises. Sulawesi tortoise adenovirus 1 was associated with necrosis and intranuclear inclusions in the gut, bone marrow, liver, kidneys, spleen, pancreas, gonads, adrenals, and blood vessels, as well as rhinitis and choroid plexitis, in a group of 105 Sulawesi tortoises (Indotestudo forsteni).21 A group mortality of over 90% was seen. A breach of this virus, with resultant disease, was seen from the initial group into additional tortoise species in the home collection of one caretaker, confirming both pathogenicity and transmissibility of this agent. The high pathogenicity of this agent is not typical of how a large DNA virus behaves in an endemic host, and it is expected that these tortoises were aberrant rather than endemic hosts. Other members of the genus Siadenovirus have been found in birds and frogs; bird siadenoviruses tend to be highly pathogenic, and disease has not been studied in frogs. It is not clear which taxa are endemic hosts of the siadenoviruses.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree