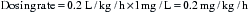CHAPTER 3 Update on Common Antimicrobials
β-LACTAM ANTIMICROBIALS
Oral Administration
The aminobenzyl penicillins (ampicillin and amoxicillin) and first-generation cephalosporins are poorly absorbed in adult horses; however, they have reasonable to good bioavailability in foals. Cefadroxil (30 mg/kg, administered orally every 12 hours), a first-generation cephalosporin, has 99% bioavailability in 2-week-old foals, but this decreases to 14% by 5 months of age. Amoxicillin, administered orally at 13 to 20 mg/kg, every 8 to 12 hours, reaches serum concentrations effective for treatment of streptococcal and non-β-lactamase–producing staphylococcal infections in foals. Cephalexin can also be orally administered in foals at a dosage of 25 mg/kg every 6 hours.
Cephalosporins: New Drugs and Novel Administration
Cefquinome is a new fourth-generation cephalosporin licensed in the United Kingdom for use in foals with septicemia and horses with respiratory tract disease. The recommended dose is 1 mg/kg, IV or IM administration, every 12 hours (for foals with septicemia) or every 24 hours (in adults with respiratory tract disease).