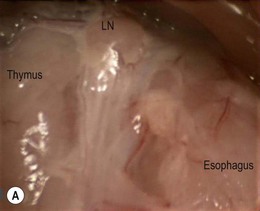
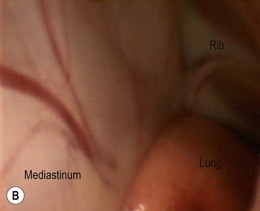
Chapter 42 There are limited descriptions of the use of thoracoscopy in cats for exploration, aspiration, and biopsy of intrathoracic tissues.1–3 Cats are included in general texts based mainly on canine and human literature,4–6 but specific procedural information is very sparse. Until recently however, the same was true for feline laparoscopic procedures (see Chapter 24), and there is no convincing reason not to assume that similar advantages of minimally invasive thoracic procedures apply to cats as they do to humans (including neonates of < 1 kg body weight7) and dogs. This chapter covers the principles of thoracoscopy in cats and describes selected surgical procedures. Minimally invasive access to the thoracic cavity for thoracoscopic exploration and observation of intrathoracic tissues, biopsy of abnormal structures, or intrathoracic surgical procedures, also referred to as video-assisted thoracic surgery (VATS), has several advantages over conventional open thoracic procedures. Thoracotomy is associated with considerable intra- and postoperative pain, discomfort, and convalescence period, which are clearly reduced in humans when using a thoracoscopic approach8,9 and there is also evidence to suggest the same applies to dogs.10 Other advantages are the magnified and detailed view of intrathoracic tissues and the increased access to deep regions, which actually allows a more extensive and complete exposure of the thoracic cavity compared to open approaches. The number of applications of thoracoscopic surgery in dogs is rapidly increasing, and procedures such as the creation of a pericardial window or partial pericardectomy,11,12 or thoracic duct ligation13,14 are becoming the standard of care nowadays in several specialized institutions. In contrast, data on thoracoscopic surgery in cats is lacking. The number of possible thoracoscopic procedures in both dogs and cats is merely related to the surgeon’s experience and skills and the willingness and creativity to convert these into an expanding range of VATS procedures. Companion animal minimally invasive surgery lags behind its human counterpart, which is primarily related to the associated costs of investment in equipment and surgical training. Nevertheless, considering the recent but revolutionary history of minimally invasive surgery in humans, both canine and feline minimally invasive surgery is likely to be increasingly applied in the near future. Moreover, the majority of advantages of minimally invasive surgery that have been described for humans are increasingly reported in veterinary patients. As with laparoscopy (see Chapter 24), the smaller average body size of cats compared to dogs requires dedicated equipment and skills, which may more often result in veterinary surgeons initially investing in minimally invasive surgery for canines rather than felines. This applies in particular to thoracoscopy, because the thorax does not expand as much as the abdominal wall does upon insufflation for laparoscopy, making the average feline thorax considerably smaller than an average canine thorax. The basic surgical principles of laparoscopic surgery also apply to thoracoscopic surgery (see Chapter 24). Anesthesia for thoracoscopy is essentially the same as for thoracotomy procedures, implying general anesthesia and mechanical intermittent positive pressure ventilation (IPPV). Extensive monitoring of physiologic parameters during thoracoscopy should be mandatory. In contrast to laparoscopy, insufflation of the thoracic cavity is usually not needed and not recommended because it carries the risk of significant hemodynamic compromise. Left hemithorax CO2-insufflation using 2–5 mmHg intrathoracic pressures resulted in significant hemodynamic compromise in dogs15,16 and is therefore advised to be used with great caution and with as low as possible pressure for as short as possible intervals. In cats, such measurements have not been performed and intrathoracic insufflation should probably be discouraged. If established pneumothorax (after trocar placement) and altered ventilation settings (e.g., increasing ventilation rate and decreasing tidal volume) do not provide sufficient space for thoracoscopy, selective single-lung ventilation may improve the working space in one hemithorax, with less severe hemodynamic changes compared to intrathoracic insufflation.17 One-lung ventilation is commonly applied in human thoracoscopic surgery and is considered a safe technique in dogs.18 Again, such techniques have not been investigated in cats and caution is indicated. Single-lung ventilation can be established through selective endobronchial intubation or selective endobronchial blockade. Double lumen endobronchial tubes are too big for use in cats. Because of the relatively short distance between the branches of lobar bronchi in cats,19 any cuff or balloon placed in a main stem bronchus may occlude branching bronchi, preventing ventilation of the cranial lung region using an endobronchial tube, especially on the right side.20 Endobronchial blockade combined with ventilation through an endotracheal tube is commonly used in human pediatric thoracoscopy and may provide a more reliable solution to one-lung ventilation in cats.21 However, selective endobronchial intubation or blockade is difficult to perform in cats, usually requiring endoscopic guidance using a flexible pediatric endoscope, and is not without risks. Fortunately, diagnostic thoracoscopy and most thoracoscopic procedures can be performed without single-lung ventilation.4,7,11,13,22 At the authors’ institution, thoracoscopic procedures in cats (and dogs) are performed commonly without single-lung ventilation. When required, surgical space can be increased by decreasing tidal volume in combination with an increase in ventilation rate, manual retraction of lung lobes away from the location of interest, and occasionally alternating brief periods of apnea in combination with periods of increased ventilation. Close cooperation and communication between the anesthetic and surgery teams is of major importance for thoracoscopic procedures. The equipment for thoracoscopic surgery is essentially the same as for laparoscopic surgery and is described in Chapter 24. It is even more essential in feline thoracoscopy than laparoscopy to use pediatric equipment. Trocars for thoracoscopy do not need valves or rubber washers because air should be able to enter and exit during ventilatory movements of the lungs. Dedicated thoracoscopic trocars (rigid or flexible) may be used, with blunt obturators. The same set of laparoscopic instruments can, however, also be used for thoracoscopy. The authors prefer to use threaded cannulas (most commonly the threaded laparoscopic cannulas) because of their controlled insertion, blunt tip, and firm grip in the thoracic wall (see Chapter 24). The part containing the internal valve and washer can be disconnected and removed, which also reduces cannula weight. Threaded cannulas are also very useful for initial entry into the thorax. The standard thoracoscopic entry point in lateral recumbency is in the 6th–8th intercostal space (ICS), midway between the costochondral junction and the epaxial muscles.23 For a specific lesion, however, an entry point is selected which is a short distance away from it, to allow triangulation of the instruments and camera to the location of interest. For specific procedures, portal locations may differ depending on the size of the cat, to enable a triangulated approach. Several techniques can be used to establish pneumothorax and insert the primary trocar (Box 42-1). Thoracoscopy can be performed as a diagnostic technique or surgical approach. It is indicated in any case in which it can obviate the need for a thoracotomy or enhance a diagnostic or therapeutic procedure, provided that the surgeon has an adequate level of VATS skills. In the case of thoracic duct ligation combined with subtotal pericardectomy for idiopathic chylothorax, for instance, a thoracoscopic approach may even prevent a second thoracotomy.24 Furthermore, VATS includes both complete thoracoscopic and thoracoscopic-assisted procedures. Thoracoscopy is contraindicated in patients with severe cardiorespiratory problems that cannot compensate for the respiratory changes inflicted by pneumothorax, such as severe cardiomyopathy or generalized lung disease requiring full utilization of tidal volume. Thoracoscopic exploration can be performed for visualization and biopsy of tissues, such as lymph nodes, mediastinum, lungs,10 pericardium, and mass lesions9 (Fig. 42-2); for identification of underlying causes of pleural effusion8 or spontaneous pneumothorax; and to evaluate or diagnose abnormalities found with diagnostic imaging techniques, such as CT, ultrasound, or MRI (Fig. 42-3). Furthermore, thoracoscopic-assisted procedures can prevent more invasive surgery, such as thoracoscopic exploration in cases of thoracic trauma, avoiding the need for additional thoracotomies or contralateral thoracotomy (Fig. 42-4). For exploration of both thoracic compartments, a paraxiphoid camera portal with the patient in dorsal recumbency is most convenient. For exploration of one specific hemithorax, access to thoracic structures is enhanced with the patient in lateral recumbency. Biopsies of mass lesions, lymph nodes, and other non-respiratory tissues can be performed using biopsy cup forceps (Fig. 42-5; see also Chapter 24). Figure 42-2 Images of thoracoscopic exploration of the left hemithorax of a cat with a persistent right aortic arch (PRAA). (A) Cranial view of the mediastinum with thymus, enlarged mediastinal lymph node (LN), and dilated esophagus. (B) Dorsal view of the aberrant mediastinum, caused by a dilated esophagus due to PRAA.
Thoracoscopy
Background
Anesthesia
Equipment, entry technique and closure
Indications
Thoracoscopic procedures
Exploration and biopsy of intrathoracic tissues
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Thoracoscopy