Chapter 19 Systemic Bacterial Infectious Diseases
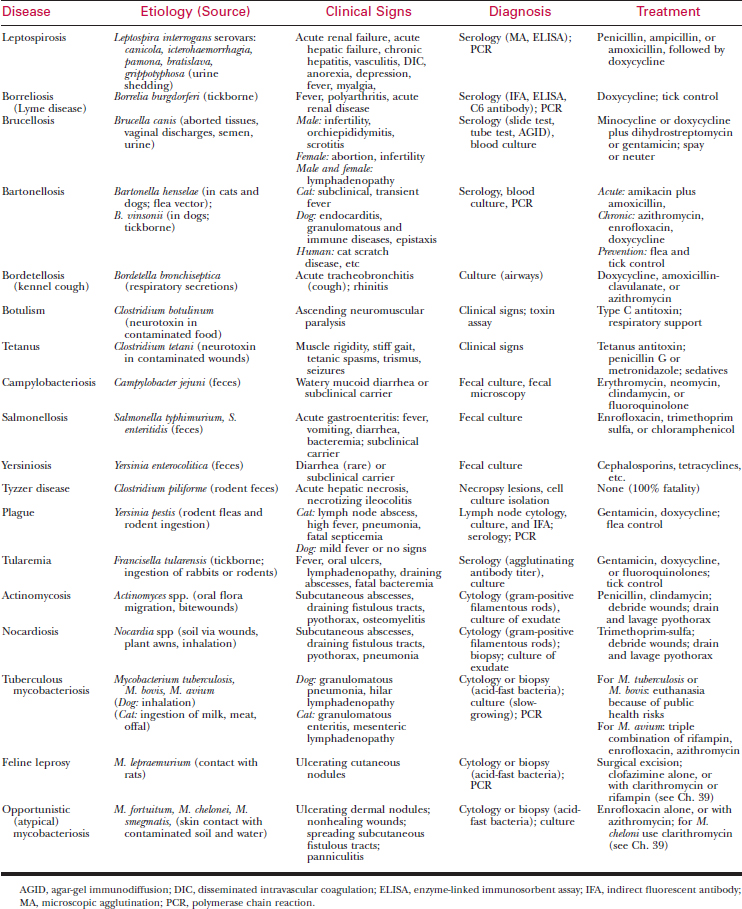
A large number of bacteria can infect dogs and cats. This chapter focuses on leptospirosis, brucellosis, and bartonellosis. Chapter 18 is devoted to Lyme borreliosis. Other infectious diseases caused by bacteria are summarized in Table 19-1, and most are described in the respective organ-system chapters. Bordetella bronchiseptica is associated with the canine infectious tracheobronchitis (see Chapter 12).Mycoplasma haemofelis and Mycoplasma haemominutum are hemotropic infections (see Chapter 22). Mycobacterial infections are associated with chronic skin disease (see Chapter 39). Actinomycosis and nocardiosis are causes of pyothorax (see Chapter 164). Tetanus and botulism cause severe neuromuscular dysfunction (see Chapters 128 and 130, respectively). Salmonellosis, campylobacteriosis, yersiniosis, and Clostridium perfringens and Cl. difficile primarily involve the intestinal tract (see Chapter 69).