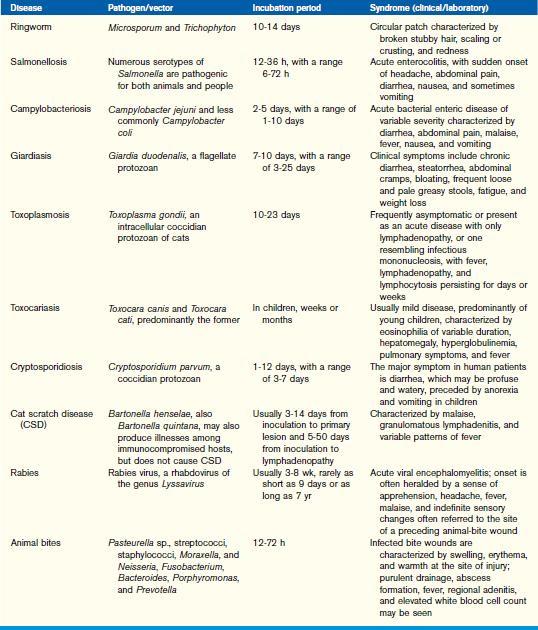
Ringworm (see Chapter 17) is the most common zoonotic disease in the United States. Ringworm (dermatophytosis) is a fungal disease of cats, dogs, sheep, rabbits, cattle, and horses that invades keratinized structures (e.g., hair, horn, nails, feathers, and skin). Although it is self-limiting, clearing without treatment may take several months. Cats, especially young cats, can harbor the fungus without any noticeable clinical signs, thereby serving as an unsuspecting reservoir of infection.
Ringworm is readily transmitted to people by direct contact with infected animals, fomites (e.g., bedding), or contaminated environment such as the soil. The infections are spread mainly by spores, which are very long lived and can persist in the environment for years in blankets, clothing, bedding, combs, and other grooming tools.
Species that cause ringworm belong to the genera Microsporum and Trichophyton. These genera of fungi are somewhat unusual in that they produce asexual spores; sexual spores are produced so infrequently that they have not been observed. The most common source of ringworm in people is Microsporum canis, a species most often found on cats and dogs. When diagnosed, ringworm should be treated to limit the transmission of the fungus to humans.
The classic lesion is a circular patch characterized by broken stubby hair, scaling or crusting, and redness. The lesion will appear to spread outward, often with a central area of healing. Animals may have just a single lesion or many.
Diagnosis is usually made by microscopic examination of hairs from the site of the lesion for the characteristic spores lining the outside of the follicles. For a definitive diagnosis of ringworm, a fungal culture should be done as this is the most reliable diagnostic test for ringworm; however, it may take up to 3 weeks before any growth is seen. Single lesions (mild infection) can be spot treated with topical antifungal creams. Oral medications are available for more severe infections.
Because of its zoonotic potential, the best ringworm prevention is treating infected animals as soon as possible to avoid its spread to other animals or humans and to improve the likelihood of recovery without reinfection. Pet owners (particularly those with young children or immune-compromised individuals in the home) should be advised of the human heath risk and referred to their physician if lesions appear on any humans in the household. Because ringworm spreads easily, it is best to keep infected animals away from others until the infection has cleared.