CHAPTER 38 Ovulation Synchronization Strategies in Dairy Cattle Using PGF2α and GnRH
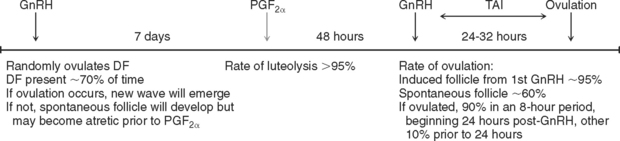
Synchronization of ovulation using timed injections of prostaglandin F2α (PGF2α) and gonadotropinreleasing hormone (GnRH)–the Ovsynch program of ovulation management–improves pregnancy rates and reduces days open in lactating dairy cows1,2 (Fig. 38-1). Inadequate pregnancy rates are one of the primary obstacles to dairy farm profitability and sustainability.3 During the past 50 years, pregnancy rates of lactating dairy cows progressively decreased, primarily because of low and steadily declining conception rates (CRs) and estrus detection rates (EDRs).4,5 CR in cows decreased from approximately 70%6,7 to 35%,8,9 although CR in heifers remained steady at approximately 70%.2,7 Current reports indicate that EDR is approximately 32% in lactating cows4 and 70% in heifers. Poor EDR in cows may be attributed to changes in hormonal concentrations between the two groups,10,11 as well as differences in environment–lactating dairy cows spend more time on surfaces with poor footing12,13 and are more susceptible to heat stress.14 Currently, the effectiveness of Ovsynch in lactating dairy cows is attributable primarily to an increase in EDR, or artificial insemination (AI) service rate, because all cows, regardless of cycling status or stage of estrous cycle, can be inseminated by appointment1,2 without the need for detection of estrus. Thus, Ovsynch can be effectively used to control time to first and subsequent inseminations by AI, thereby maximizing service rate and improving overall EDR.1
Ovsynch is the basis for most timed-AI programs in dairy cows in the United States. Ovsynch was developed in the early 1990s at the University of Wisconsin– Madison by Pursley and Wiltbank.15 The objectives of the original Ovsynch research were to (1) hormonally control the onset of a new follicular wave, (2) control the lifespan of the spontaneous and induced CL, and (3) control the time of ovulation of the dominant follicle (DF).
Three hormonal injections were needed to accomplish these three objectives (see Fig. 38-1). The first GnRH injection caused ovulation, or luteinization, if a functional DF was present in the ovary. Subsequently, if ovulation occurred, a new follicular wave emerged approximately 1.5 to 2 days later.15 GnRH did not cause ovulation if the stage of follicle development was in the first 3 days of a spontaneous follicular wave. The newly induced wave, or a spontaneous wave if ovulation did not occur, was allowed to develop, with selection and dominance of a DF during the following 7 days. At that time, PGF2α was administered to induce luteolysis, thus allowing for further growth and maturation of the DF. Then, 48 hours later, a second GnRH injection induced a preovulatory luteinizing hormone (LH) surge that triggered ovulation approximately 28 hours after treatment with GnRH. Cows treated with Ovsynch demonstrated overall CRs similar to those obtained in cows that had been bred to detected estrus (37% versus 39%, respectively; P > 0.05).2
This chapter discusses how Ovsynch works and how it may be altered in the future to improve the percentage of cows synchronized, and the possibility of improving CR by means of greater control over the ovulatory follicle.
SYNCHRONIZATION RATES WITH OVSYNCH
The main limitation of Ovsynch is the wide variability in synchronization rate (SR), defined in this protocol as regression of a CL and induction of ovulation in response to the final GnRH. Approximately 10% to 30% of cows may not synchronize.9,16–18 It is likely that nonsynchronized cows could receive AI at an inappropriate time relative to ovulation, thereby decreasing their chances of becoming pregnant. Nevertheless, with SRs in this range, it is impressive that CR after Ovsynch is similar to that with AI at the time of detected estrus.
Vasconcelos and associates attributed most of the variation in SRs to the stage of the estrous cycle at which Ovsynch was initiated.18 Cows started on Ovsynch at midcycle (days 5 to 9 of the cycle) had a greater probability of synchronizing and therefore had a greater chance of conception. The key reasons for increased SR in midcycle were the presence of a DF responsive to GnRH during the first follicular wave and a CL that remained functional during the 7-day period between GnRH and PGF2α. Early in the estrous cycle (days 1 to 4), synchronization was impaired by the presence of newly emerging follicles incapable of ovulating in response to the first GnRH dose. These growing follicles had an increased chance of reaching atresia before PGF2α was given. Later in the estrous cycle (day 10 and beyond), the presence of a functional DF at the time of first GnRH dose varied depending on follicular wave pattern of the cow and on time of follicular emergence. Moreover, it was possible to encounter spontaneous luteolysis triggered by endometrial PGF2α during this period. Characterization of the ovarian events that take place when Ovsynch is started at different stages of the estrous cycle indicates that the main causes of synchronization failure were (1) lack of ovulatory response to the first GnRH, (2) atresia of the DF before PGF2α administration, and (3) spontaneous luteolysis between administration of the first dose of GnRH and of PGF2α.18 As previously stated, early atresia of the DF before PGF2α administration was primarily a function of lack of ovulatory response to the first GnRH dose. For example, a 3-day-old follicle at the time of first GnRH dose (which probably would be the oldest not capable of ovulating19) would be 10 days old at the time of PGF2α administration and already undergoing atresia. Therefore, at the time of the second GnRH dose, another follicular wave could be emerging, and the follicle destined to be dominant would be too young to respond to the final GnRH dose with ovulation. Cows in the latter situation probably would be in standing estrus 3 to 5 days after the second GnRH dose. In addition, cows with spontaneous luteolysis during the time from the first GnRH dose to PGF2α administration probably would be in late stages of the estrous cycle and undergoing natural luteolysis before PGF2α was given. Such cows probably would be in estrus from 1 day before to 1 day after PGF2α administration. In either case, cows would not have a synchronized ovulation in response to the second GnRH injection and probably would not conceive with the timed AI.
Because synchronization to Ovsynch depends primarily on the ovulatory response to the first GnRH, followed by emergence of a new follicular wave,18 several studies tried to develop presynchronization strategies to ensure presence of a functional mature DF at the time of the first GnRH of Ovsynch.20–24 The most popular strategies were based on the use of PGF2α at specific times before Ovsynch was started. The absence of consistent synchronization results probably was due to the fact that PGF2α controls only the lifespan of the CL, which has little effect on follicular development. Thus, stage of follicular development at the beginning of Ovsynch probably would have too much variation if it was presynchronized only with PGF2α-based programs.
Heifers are poorly synchronized when treated with Ovsynch (50%–60% SR).2 This is the key reason why heifers treated with Ovsynch have significantly lower CR compared with heifers bred to detected estrus (35% versus 74%, respectively; P < 0.01).2 The physiologic events underlying such poor synchronization can be interpreted as an exacerbation of what happens in some of the cows that do not synchronize. Therefore, the synchronization situation in heifers may be used as a model to illustrate the underlying ovarian physiology of some nonsynchronizing cows.
Newly emerging follicles grow more rapidly15 and for a shorter period10,11 in heifers than they do in lactating cows. Thus, a DF in heifers can be expected to reach atresia sooner. Ovsynch-treated heifers that respond to the first GnRH dose may undergo follicular turnover of the new ovulatory DF before PGF2α administration. If a DF from a new wave becomes atretic before the PGF2α is given, an emerging follicular wave, rather than a mature DF, probably would be present at the time of the final GnRH injection. Consequently, synchronized ovulation to the final GnRH dose of Ovsynch is unlikely–and so is conception.
PROTOCOL COMPONENTS AND STRATEGIES
Interval between First GnRH Dose and PGF2α Administration
Effects of Increasing Time between First GnRH Dose and PGF2α Administration
Treatments based on maintenance of subluteal levels of progesterone (P4) (1–2 ng/ml) for an extended period are known to cause prolonged growth and dominance of the DF. This follicle is described as a persistent DF.25–29 When the persistent DF is allowed to ovulate, fertility is decreased when compared with that of younger ovulatory follicles.27,28,30–32 It was proposed that the negative effect of prolonged growth and dominance of the ovulatory follicle on fertility is associated with the hormonal environment in which the follicle is induced to persist. Subluteal levels of P4 during a prolonged period cause increased frequency of LH pulsatility33,34 and prevents a preovulatory LH surge from occurring.28,32,35,36 As a result, the oocyte resumes meiosis while still contained in the follicle and starts undergoing premature nuclear maturation dissociated from follicular maturation and ovulation. Histologic characteristics of these persistent DFs indicate that the oocyte undergoes early germinal vesicle breakdown and continues a progression through the cell cycle toward metaphase I or II.31,37 By the time of ovulation of the persistent follicle, the oocyte has already matured and aged, resulting in lower fertility, which may be explained by either low fertilization rates38,39 or high early embryonic mortality,40 or both. Also, persistent follicles are known to maintain a high and sustained production of estradiol, which may alter intrafollicular, oviductal, and uterine environments, thereby compromising sperm or oocyte transport and embryonic development.26–28,31,32,37
Follicular wave pattern and fertility.
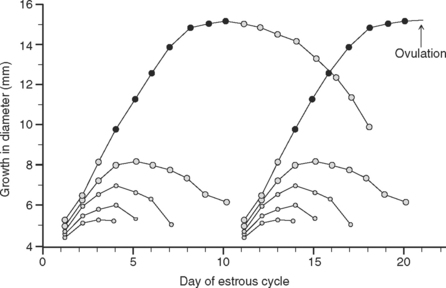
It is widely accepted that ovarian follicles develop primarily in two- or three-wave patterns throughout the estrous cycle of cattle4,42,43 (Fig. 38-2). Lactating dairy cows generally have two waves of follicular growth per cycle.10,11,44–46 In either wave pattern, the DF of the final wave is the one intended to ovulate. The mean interovulatory interval is shorter for two-wave versus three-wave cows (approximately 20 days versus 22–23 days, respectively), and the emergence of the ovulatory wave occurs earlier for two-wave than for three-wave cows (approximately day 11 versus day 16 of the cycle), leading to different lifespans of the ovulatory follicles for each wave pattern. At the time of ovulation, therefore, a difference in age can be observed between the preovulatory follicle coming from a two-wave (9 to 10 days) versus a three-wave (6 to 7 days) pattern of follicle development.41,43 Thus, the two-wave pattern that predominates in lactating dairy cows generally is characterized by a larger and older DF reaching ovulation compared with that in three-wave patterns.1,44 Moreover, second-wave preovulatory follicles were found to be approximately 3 days older than those of a third wave when the interval from emergence to estrus was measured.44 This difference in lifespan between second-wave and third-wave ovulatory follicles was explained by a longer period of follicular dominance, rather than by the interval of follicle growth from emergence to dominance, which remained the same in both cases.44
As previously stated, the ovulatory follicle of a two-wave cycle is older than that of a three-wave cycle. By analogy with the effect of prolonged dominance of a persistent follicle, the older age of a second-wave ovulatory follicle could be expected to have a negative impact on fertility. If most dairy cows have two waves, then a high proportion of the population could be expected to be ovulating an older DF, thus providing a possible explanation of the low CR problem in lactating dairy cows. In applying this concept to Ovsynch, the established 7-day interval may be limiting the number of cows that ovulate an aged follicle. This may be the reason why fertility of cows that synchronized exceeds fertility of cows inseminated after a detected estrus. Decreasing the age of the follicle at the time of the final GnRH so that it is more like a three-wave follicle may offer greater improvements. Only a few studies compared fertility between three- and two-wave cows.44,46,47 There may be significant trends toward greater CR in three- than in two-wave cows. Unfortunately, sufficient numbers of three-wave cows probably were difficult to procure for these studies, because most lactating dairy cows have two follicular waves during a normal estrous cycle.48
Effect of Duration of Dominance of Ovulatory Follicle on Fertility
An even stronger association is likely between fertility and the length of ovulatory follicle lifespan or duration of physiologic dominance, than between fertility and the number of follicular waves. In one study, the interval from emergence of the ovulatory follicle to estrus was approximately 1 day shorter in cows diagnosed as pregnant compared with cows diagnosed as not pregnant (7.8 ±0.2 days versus 8.6 ±0.2 days, respectively; P < 0.01), regardless of whether they had two or three follicular waves.44 A key result from the same study was a significant inverse relationship between duration of dominance and CR. The longer the ovulatory follicle remained in the ovary waiting for the ovulation trigger, the lower the probability of establishing a pregnancy.
Furthermore, in a recent study that compared follicle development between lactating cows and nulliparous heifers,11 heifers had a 1.2-day shorter duration of dominance of the ovulatory follicle when compared with cows. The relevance of this issue becomes clear when the CR difference between these two groups is considered. It may be possible to reduce the number of cows that would ovulate an old follicle. The only possible way to accomplish this, however, is to reduce the time from the first GnRH dose to PGF2α administration.
Effects of Reducing the Time between First GnRH Dose and PGF2α Administration
Altering the time from first GnRH dose to PGF2α administration probably could affect the maturity level of the ovulatory follicle at the time of the final GnRH dose. Follicles that are induced to ovulate before reaching physiologic maturity may be less fertile. Several studies demonstrated the relationship between smaller follicular size at the time of induced ovulation with GnRH and lower CR.9,49,50 In a recent study from our laboratory, cows treated with Ovsynch that ovulated to a GnRH-induced LH surge with ovulatory follicles less than 12 mm in diameter had lower CR compared with ovulatory follicles 12 mm or larger (27% versus 36%; P < 0.01) and comprised nearly 34% of the cows in the study (N = 1424).9 It is likely that these follicles were not allowed to reach peak physiologic maturity.
Compromised oocyte quality and subsequent inadequate luteal function are possible reasons why reduced age, size, or physiologic status of follicles reduces CR. Lower circulating P4 concentrations in the subsequent cycle51 can impair embryo recognition, development, and implantation. Another possibility could be related to a nonsynchronized, and hence incorrect, timing of endocrine signals controlling final follicle and oocyte maturation, leading to ovulation of an immature oocyte.52 Reduced estradiol concentrations due to a younger DF also can alter uterine and oviductal environment, affecting either gamete or embryo survivability.16,29,43,52,53
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree